Abstract
Diabetes mellitus (DM) is a cluster of metabolic diseases that exhibits high blood glucose levels accompanied by hyperlipidemia and inflammation. DM is the primary risk factor contributes majorly to cardiovascular disease (CVD) mediated morbidity and mortality. The incidence of dyslipidemia seems to attribute considerably to the initiation of CVDs. The beneficial action of isoquercetin on hyperlipidemia and related signaling pathways are not documented yet, hence we decide to carry out this study. The experimental rats were divided into five groups: Group 1, control rats; group 2, isoquercetin control (40 mg/kg b.w); group 3, diabetic rats (STZ-40 mg/kg b.w); group 4, diabetic + isoquercetin (40 mg/kg b.w); and group 5, diabetic + glibenclamide (600 µg/kg b.w). The animals were sacrificed at the end of the experimental duration of 45 days. Results of our analysis reveal that isoquercetin have a major impact on the tissue lipid profile, isoquercetin strongly regulates the expression of various lipid-metabolizing enzymes, C-reactive protein, expression of various inflammatory genes, SREBP-1C genes and proteins and AMP-activated protein kinase-α (AMPK) signaling pathway genes and proteins. Results recommend that isoquercetin can be effective in mitigating the consequences of hyperlipidemia and DM.
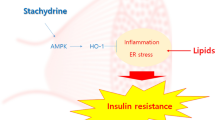
Graphic Abstract





Similar content being viewed by others
Abbreviations
- ACC:
-
Acetyl-CoA carboxylase
- AMPK-α:
-
AMP-activated protein kinase-α
- DM:
-
Diabetes mellitus
- FAS:
-
Fatty acid synthase
- HDL:
-
High-density lipoprotein
- HMG Co-A:
-
3-Hydroxy 3-methyl glutaryl coenzyme A
- LCAT:
-
Lecithin cholesterol acyltransferase
- LDL:
-
Low-density lipoprotein
- LPL:
-
Lipoprotein lipase
- PL:
-
Phospholipids
- STZ:
-
Streptozotocin
- SREBP-1C:
-
Sterol regulatory element-binding protein-1
- TC:
-
Total cholesterol
- T2DM:
-
Type 2 diabetes mellitus
- TG:
-
Triglycerides
References
Leon BM, Maddox TM (2015) Diabetes and cardiovascular disease: epidemiology, biological mechanisms, treatment recommendations and future research. World J Diabetes 6:1246–1258. https://doi.org/10.4239/wjd.v6.i13.1246
Rines AK, Sharabi K, Tavares CDJ, Puigserver P (2016) Targeting hepatic glucose output in the treatment of type 2 diabetes. Nat Rev Drug Discov 15:786–804. https://doi.org/10.1038/nrd.2016.151
Renard CB, Kramer F, Johansson F, Lamharzi N, Tannock LR, Herrath MG, Chait A, Bornfeldt KE (2004) Diabetes and diabetes-associated lipid abnormalities have distinct effects on initiation and progression of atherosclerotic lesions. J Clin Invest 114:659–668
Siasos G, Tousoulis D, Oikonomou E, Zaromitidou M, Stefanadis C, Papavassiliou AG (2011) Inflammatory markers in hyperlipidemia: from experimental models to clinical practice. Curr Pharm Des 17:4132–4146
Ruderman NB, Carling D, Prentki M, Cacicedo JM (2013) AMPK, insulin resistance, and the metabolic syndrome. J Clin Invest 123:2764–2772. https://doi.org/10.1172/JCI67227
Li Y, Xu S, Mihaylova MM, Zheng B, Hou X, Jiang B, Park O, Luo Z, Lefai E, Shyy JY, Gao B, Wierzbicki M, Verbeuren TJ, Shaw RJ, Cohen RA, Zang M (2011) AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell Metab 13(4):376–388
Panche AN, Diwan AD, Chandra SR (2016) Flavonoids: an overview. J Nutr Sci 5:e47. https://doi.org/10.1017/jns.2016.41
Jayachandran M, Wu Z, Ganesan K, Khalid S, Chung SM, Xu B (2019) Isoquercetin upregulates antioxidant genes, suppresses inflammatory cytokines and regulates AMPK pathway in streptozotocin-induced diabetic rats. Chem Biol Interact 303:62–69. https://doi.org/10.1016/j.cbi.2019.02.017
Morikawa K, Nonaka M, Narahara M, Torii I, Kawaguchi K, Yoshikawa T, Kumazawa Y, Morikawa S (2003) Inhibitory effect of quercetin on carrageenan-induced inflammation in rats. Life Sci 74:709–721
Jayachandran M, Zhang T, Ganesan K, Xu B, Chung SSM (2018) Isoquercetin ameliorates hyperglycemia and regulates key enzymes of glucose metabolism via insulin signaling pathway in streptozotocin-induced diabetic rats. Eur J Pharmacol 829:112–120. https://doi.org/10.1016/j.ejphar.2018.04.015
Kim HY, Moon BH, Lee HJ, Choi DH (2004) Flavonoid glycosides from the leaves of Eucommia ulmoides O with glycation inhibitory activity. J Ethnopharmacol 93:227–230
Sheikh BA, Pari L, Rathinam A, Chandramohan R (2015) Trans-anethole, a terpenoid ameliorates hyperglycemia by regulating key enzymes of carbohydrate metabolism in streptozotocin induced diabetic rats. Biochimie 112:57–65. https://doi.org/10.1016/j.biochi.2015.02.008
Folch J, Lees M, Solane SGH (1957) A simple method for isolation and purification of total lipids from animal tissues. J Biol Chem 26:497–509
Zlatkis A, Zak B, Boyle GJ (1953) A simple method for determination of serum cholesterol. J. Clin. Med Res 41:486–492
Fossati P, Prencipe L (1982) Serum triglycerides determined colorimetrically with an enzyme that produces hydrogen peroxide. Clin Chem 28:2077–2080
Falholt K, Lund B, Falholt W (1973) An easy colorimetric micromethod for routine determination of free fatty acids in plasma. Clin Chim Acta 46:105–111
Zilversmit BB, Davis AK (1950) Micro determination of plasma phospholipids by trichloroacetic acid precipitation. J Lab Clin Med 35:155–160
Hitz J, Steinmetz J, Siest G (1983) Plasma lecithin cholesterol acyltransferase – reference values and effects of xenobiotics. Clin Chim Acta 133:85–96
Korn ED (1955) Clearing factor: a heparin activated lipoprotein lipase: Isolation and characterization of enzyme from normal rats. J Biol Chem 215:1–26
Philipp B, Shapiro DJ (1970) Improved methods for the assay and activation of 3-hydroxy-3-methyl glutaryl coenzyme A reductase. J Lipid Res 20:588–593
American Diabetes Association (2009) Diagnosis and classification of diabetes mellitus. Diabetes Care 32:S62–S67. https://doi.org/10.2337/dc10-S062
Ahmad WNHW, Sakri F, Mokhsin A, Rahman T, Nasir NM, Abdul-Razak S, Md Yasin M, Ismail AM, Ismail Z, Nawawi H (2015) Low serum high-density lipoprotein cholesterol concentration is an independent predictor for enhanced inflammation and endothelial activation. PLoS ONE 10:e0116867. https://doi.org/10.1371/journal.pone.0116867
DeFronzo RA, Tripathy D (2009) Skeletal muscle insulin resistance is the primary defect in type 2 diabetes. Diabetes Care 32:S157–S163. https://doi.org/10.2337/dc09-S302
Schofield JD, Liu Y, Rao-Balakrishna P, Malik RA, Soran H (2016) Diabetes dyslipidemia. Diabetes Ther 7:203–219. https://doi.org/10.1007/s13300-016-0167-x
Jayaraman R, Subramani S, Abdullah SHS, Udaiyar M (2018) Antihyperglycemic effect of hesperetin, a citrus flavonoid, extenuates hyperglycemia and exploring the potential role in antioxidant and antihyperlipidemic in streptozotocin-induced diabetic rats. Biomed Pharmacother 97:98–106. https://doi.org/10.1016/j.biopha.2017.10.102
Zodda D, Giammona R, Schifilliti S (2018) Treatment strategy for dyslipidemia in cardiovascular disease prevention: focus on old and new drugs. Pharmacy (Basel) 6(1):E10. https://doi.org/10.3390/pharmacy6010010
Srivastava RAK, Pinkosky SL, Filippov S, Hanselman JC, Cramer CT, Newton RS (2012) AMP-activated protein kinase: an emerging drug target to regulate imbalances in lipid and carbohydrate metabolism to treat cardio-metabolic diseases. J Lipid Res 53:2490–2514. https://doi.org/10.1194/jlr.R025882
Li Y, Xu S, Mihaylova MM, Zheng B, Hou X, Jiang B, Park O, Luo Z, Lefai E, Shyy JY, Gao B, Wierzbicki M, Verbeuren TJ, Shaw RJ, Cohen RA, Zang M (2011) AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell Metab 13:376–388. https://doi.org/10.1016/j.cmet.2011.03.009
Rui L (2014) Energy metabolism in the liver. Compr Physiol 4:177–197. https://doi.org/10.1002/cphy.c130024
Commerford SR, Peng L, Dube JJ, Doherty RM (2004) In vivo regulation of SREBP-1c in skeletal muscle: effects of nutritional status, glucose, insulin, and leptin. Am J Physiol Regul Integr Comp Physiol 287:R218–R227
Badawi A, Klip A, Haddad P, Cole DEC, Bailo BG, El-Sohemy A, Karmali M (2010) Type 2 diabetes mellitus and inflammation: prospects for biomarkers of risk and nutritional intervention. Diabetes Metab Syndr Obes 3:173–186
Acknowledgements
Authors thank Ms. Yinhua Liu in Zhuhai Campus of Zunyi Medical University for her technical assistance on the use of the animal facility.
Funding
The work was jointly supported by two grants R201714 and R201914 from Beijing Normal University-Hong Kong Baptist University United International College, Zhuhai, Guangdong, China.
Author information
Authors and Affiliations
Contributions
BX and SSMC designed, written and proofread the manuscript, MJ designed, carried out the experiments, written and proofread the manuscript, TZ helped in feeding the animals and dissection, ZW helped in carrying out few molecular studies.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jayachandran, M., Zhang, T., Wu, Z. et al. Isoquercetin regulates SREBP-1C via AMPK pathway in skeletal muscle to exert antihyperlipidemic and anti-inflammatory effects in STZ induced diabetic rats. Mol Biol Rep 47, 593–602 (2020). https://doi.org/10.1007/s11033-019-05166-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-019-05166-y