Abstract
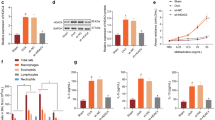
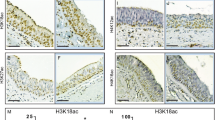
DNMT1 is the most important methyltransferase enzyme, involved in the regulation of gene expression and appropriate histone modification. It interact with proliferating cell nuclear antigen (PCNA), SNF2 family member ATP-dependent chromatin remodeling enzyme, cyclin dependent kinases inhibitor, E2F1 transcription factor and HDACs to form a repressor complex known as HDAC complexes. The interaction of DNMT1 with numerous protein suppressors of promoters suggests that the enzyme is a crucial element of the transcription suppression complex. Since the mechanism behind over expression of Socs3 in Asthma is unclear, we study the Epigenetic mode of overexpression of Socs3 in terms of methylation/acetylation/inactivation of HDACs/activation of HATs enzymes in a mouse model of asthma. The results show that low expression of DNMT1 might indirectly induce the expression of Socs3 and HAT, and inhibit the expression of HDACs family. Furthermore knockdown of DNMT1 by siRNA induced expression of Socs3 while knock down of Socs3 by siRNA has no effect on DNMT1 expression. Our result suggests that the over expression of Socs3 is due to the inhibition of HDACs complex and hyperacetylation of histones molecule along with down regulation of DNMT1 gene. In depth study on DNMT1 might be useful for the development of therapeutic drug against asthma/allergic diseases.










Similar content being viewed by others
References
Robertson KD, Ait-Si-Ali S, Yokochi T, Wade PA, Jones PL, Wolffe AP (2000) DNMT1 forms a complex with rb, E2F1 and HDAC1 and represses transcription from E2F-responsive promoters. Nat Genet 25:338–342
Kulis M, Esteller M (2010) DNA methylation and cancer. Adv Genet 70:27–56
Bedford DC, Kasper LH, Fukuyama T, Brindle PK (2010) Target gene context influences the transcriptional requirement for the KAT3 family of CBP and p300 histone acetyltransferases. Epigenetics 5(1):9–15
Sun Y, Jiang X, Price BD (2010) Tip60: connecting chromatin to DNA damage signaling. Cell Cycle 9(5):930–936
Bao J, Sack MN (2010) Protein deacetylation by sirtuins: delineating a post-translational regulatory program responsive to nutrient and redox stressors. Cell Mol Life Sci 67(18):3073–3087
Moss TJ, Wallrath LL (2007) Connections between epigenetic gene silencing and human disease. Mutat Res 618(1–2):163–174
Schwartz YB, Kahn TG, Stenberg P, Ohno K, Bourgon R, Pirrotta V (2010) Alternative epigenetic chromatin states of polycomb target genes. PLoS Genet 6(1):e1000805
Vermeulen L, Vanden Berghe W, Beck IM, De Bosscher K, Haegeman G (2009) The versatile role of MSKs in transcriptional regulation. Trends Biochem Sci 34(6):311–318
Chi P, Allis CD, Wang GG (2010) Covalent histone modifications–miswritten, misinterpreted and mis-erased in human cancers. Nat Rev Cancer 10(7):457–469
Kouzarides T (2007) Chromatin modifications and their function. Cell 128(4):693–705
Sebova K, Fridrichova I (2010) Epigenetic tools in potential anticancer therapy. Anticancer Drug 21(6):565–577
Eisen JA, Sweder KS, Hanawalt PC (1995) Evolution of the SNF2 family of proteins: subfamilies with distinct sequences and functions. Nucleic Acids Res 23(14):2715–2723
Boyer LA, Logie C, Bonte E, Becker PB, Wade PA, Wolffe AP, Wu C, Imbalzano AN, Peterson CL (2000) Functional delineation of three groups of the ATP-dependent family of chromatin remodeling enzymes. J Biol Chem 275(25):18864–18870
Eberharter A, Becker PB (2004) ATP-dependent nucleosome remodelling: factors and functions. J Cell Sci 117(Pt 17):3707–3711
Miyata T, Oyama T, Mayanagi K, Ishino S, Ishino Y, Morikawa K (2004) The clamploading complex for processive DNA replication. Nat Struct Mol Biol 11:632–636
Iida T, Suetake I, Tajima S, Morioka H, Ohta S, Obuse C, Tsurimoto T (2002) PCNA clamp facilitates action of DNA cytosine methyltransferase 1 on hemimethylated DNA. Genes Cells 7:997–1007
Chuang LS, Ian HI, Koh TW, Ng HH, Xu G, Li BF (1997) Human DNA-(cytosine-5) methyltransferase-PCNA complex as a target for p21WAF1. Science 277:1996–2000
Fan G, Beard C, Chen RZ, Csankovszki G, Sun Y, Siniaia M, Biniszkiewicz D, Bates B, Lee PP, Ku¨hn R, Trumpp A, Poon Chi-Sang, Wilson CB, Jaenisch R (2001) DNA hypomethylation perturbs the function and survival of CNS neurons in postnatal animals. J Neurosci 21(3):788–797
Lei H, Oh S, Okano M, Juttermann R, Goss K, Jaenisch R, Li E (1996) De novo DNA cytosine methyltransferase activities in mouse embryonic stem cells. Development 122:3195–3205
Ito K, Caramori G, Lim S et al (2002) Expression and activity of histone deacetylases (HDACs) in human asthmatic airways. Am J Respir Crit Care Med 166:392–396
Cosio BG, Mann B, Ito K et al (2004) Histone acetylase and deacetylase activity in alveolar macrophages and blood monocytes in asthma. Am J Respir Crit Care Med 170:141–147
Barnes PJ, Adcock IM, Ito K (2005) Histone acetylation and deacetylation: importance in inflammatory lung diseases. Eur Respir J 25:552–563
Greenhalgh CJ, Hilton DJ (2001) Negative regulation of cytokine signaling. J Leukoc Biol 70:348–356
Larsen L, Ropke C (2002) Suppressors of cytokine signalling: SOCS. APMIS 110:833–844
Verma M, Chattopadhyay BD, Paul BN (2012) Epigenetic regulation of DNMT1 gene in mouse model of asthma disease. Mol Biol Rep 40(3):2357–2368
Li Y, Deuring J, Peppelenbosch MP, Kuipers EJ, de Haar C, van der Woude CJ (2012) IL-6 induced DNMT1 mediates SOCS3 promoter hypermethylation in ulcerative colitis related colorectal cancer. Carcinogenesis 10:1889–1896
Espada J, Ballestar E, Fraga MF et al (2004) Human DNA methyltransferase 1 is required for maintenance of the histone H3 modification pattern. J Biol Chem 279:37175–37184
Fuks F, Burgers WA, Brehm A, Hughes-Davies L, Kouzarides T (2000) DNA methyltransferase DNMT1 associates with histone deacetylase activity. Nat Genet 24:88–91
Ratthe C, Pelletier M, Chiasson S, Girard D (2007) Molecular mechanisms involved in interleukin-4-induced human neutrophils: expression and regulation of suppressor of cytokine signaling. J Leukoc Biol 81:1287–1296
Lee PP, Fitzpatrick DR, Beard C, Lehar S, Jessup, Makar KW, Perez-Melgosa M, Sweetser MT, Schlissel MS, Naguyen S, Cherry SR, Tsai JH, Tuckar SM, Weaver WM, Kelso A, Jaenisch R, Wilson CB (2001) A critical role for DNMT1 and DNA methylation in T cell development, function and survival. Immunity 15:763–774
Craig JM, Wong NC (editor) (2011). Epigenetics: a reference manual. Caister Academic Press. ISBN 978-1-904455-88-2
Robertson KD (2005) DNA methylation and human disease. Nat Rev Genet 6:597–610
Tokita T, Maesawa C, Kimura T, Kotani K, Takahashi K, Akasaka T, Masuda T (2007) Methylation status of the SOCS3 gene in human malignant melanomas. Int J Oncol 30:689–694
Logie C, Tse C, Hansen JC, Peterson CL (1999) The core histone N-terminal domains are required for multiple rounds of catalytic chromatin remodeling by the SWI/SNF and RSC complexes. Biochemistry 38:2514–2522
Morales V, Richard-Foy H (2000) Role of histone N-terminal tails and their acetylation in nucleosome dynamics. Mol Cell Biol 20:7230–7237
Davie JR, Spencer VA (1999) Control of histone modifications. J Cell Biochem Suppl 32–3:141–148
Lee DY, Hayes JJ, Pruss D, Wolffe AP (1993) A positive role for histone acetylation in transcription factor access to nucleosomal DNA. Cell 72:73–84
Eden S, Hashimshony T, Keshet I, Cedar H, Thorne AW (1998) DNA methylation models histone acetylation. Nature 394:842
Rountree MR, Bachman KE, Baylin SB (2000) DNMT1 binds HDAC2 and a new co-repressor, DMAP1, to form a complex at replication foci. Nat Genet 25:269–277
Murahidy A, Ito M, Adcock IM, Barnes PJ, Ito K (2005) Reduction of histone deacetylase expression and activity in smoking asthmatics: a mechanism of steroid resistance. Proc Am Thorac Soc 2:A889
Thomson NC, Chaudhuri R, Livingston E (2003) Active cigarette smoking and asthma. Clin Exp Allergy 33:1471–1475
Annuziato AT, Seale RL (1983) Histone deacetylation is required for the maturation of newly replicated chromatin. J Biol Chem 258:12675–12684
Kishikawa S, Murata T, Kimura H, Shiota K, Yokoyama KK (2002) Regulation of transcription of the DNMT1 gene by Sp1 and Sp3 zinc finger proteins. Eur J Biochem 269:2961–2970
Kishikawa S, Murata T, Ugai H, Yamazaki T, Yokoyama KK (2003) Control elements of DNMT1 gene are regulated in cell-cycle dependent manner. Nucleic Acids Res Suppl 3:307–308
Ehlting C, Aussinger DH, Bode JG (2005) Sp3 is involved in the regulation of SOCS3 gene expression. Biochem J 387:737–745
McCabe MT, Davis JN, Day ML (2005) Regulation of DNA methyltransferase 1 by the pRb/E2F1 pathway. Cancer Res 65:3624–3632
Tan HH, Porter AG (2009) p21WAF1 negatively regulates DNMT1 expression in mammalian cells. Biochem Biophys Res Commun 382:171–176
Xiao H, Chung J, Kao HY, Yang YC (2003) Tip60 is a co-repressor for STAT3. J Biol Chem 278(13):11197–11204
Xu W, Edmondson DG, Roth SY (1998) Mammalian GCN5 and P/CAF acetyltransferases have homologous amino-terminal domains important for recognition of nucleosomal substrates. Mol Cell Biol 18(10):5659–5669
Jarvis CD, Geiman T, Vila-Storm MP, Osipovich O, Akella U, Candeias S, Nathan I, Durum SK, Muegge K (1996) A novel putative helicase produced in early murine lymphocytes. Gene 169:203–207
Dennis K, Fan T, Geiman T, Yan Q, Muegge K (2001) Lsh, a member of the SNF2 family, is required for genome-wide methylation. Genes Dev 15:2940–2944
Acknowledgments
We thank Dr. K.C. Gupta, Director, Indian Institute of Toxicology Research for support and encouragement. We would like to express our gratitude to Dr. C. Kesavachandran, Dr. Dhirendra Singh, Dr Mahadeo Kumar, for encouragement and inspiration. I am obliged to technical staff of this institution who helped us to peruse this work. We are thankful to Dr S.L Nagale Immunobiology division who has helped us a lot. I am especially thankful to Dr. Amit Sharma for manuscript preparation. This work was financially supported by the Indian Council of Medical Research, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Verma, M., Chattopadhyay, B.D., Kumar, S. et al. DNA methyltransferase 1(DNMT1) induced the expression of suppressors of cytokine signaling3 (Socs3) in a mouse model of asthma. Mol Biol Rep 41, 4413–4424 (2014). https://doi.org/10.1007/s11033-014-3312-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-014-3312-5