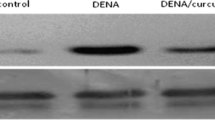
Abstract
In this study we investigated the effects of curcumin, derived from plant Curcuma longa, on oxidative toxicity, and the possible molecular mechanism of antitumour of curcumin in liver cancer rats. Results showed that blood levels of Gamma-glutamyltransferase, aspartate aminotransferase, alanine aminotransferase, glutathione S-transferase, and liver level of MD were significantly decreased after curcumin feeding. Levels of the liver malondialdehyde MDA, nitric oxide and antioxidant enzymes were significantly increased. Moreover, RT-PCR and Western blot analysis results showed that curcumin treatment significantly decreased liver vascular endothelial growth factor (VEGF), CyclinD1 and CDK4 mRNA expression levels and CyclinD1 and CDK4 proteins levels in liver cancer rats. These findings were confirmed by histopathology. It is concluded that curcumin can protect the liver from the damage caused by N-nitrosodiethylamine. Moreover, curcumin has the potential to be used in a therapy for liver cancer. The present data provide evidence to support the presence of free radicals and VEGF, CyclinD1 and CDK4 mRNA in rat tumour cells. Studies are in progress in order to further characterize the role of VEGF, CyclinD1 and CDK4 mRNA in liver cancer cells and in hepatic therapeutics.



Similar content being viewed by others
References
Loguercio C, De Girolamo V, De Sio I, Tuccillo C, Ascione A, Baldi F, Budillon G (2001) Non-alcoholic fatty liver disease in an area of southern Italy: main clinical, histological and pathophysiological aspects. J Hepatol 35:568–574
Levi F, Horvath C, Mechkouri M, Roulon A, Bailleul F, Lemaigre G, Reinber GA, Mathe G (1987) Circadian time dependence of murine tolerance for the alkylating agent peptichemio. Eur J Cancer Clin Oncol 23:487–497
Song JG, Nakano S, Ohdo S, Ogawa N (1993) Chronotoxicity and chronopharmacokinetics of methotrexate in mice: modification by feeding schedule. Jpn J Pharmacol 62:373–378
Bjarnason GA, Hrushesky WJM (1994) Cancer chronotherapy. In: Hrushesky WJM (ed) Circadian cancer therapy. CRC Press, Boca Raton, pp 241–263
Cole GM, Teter B, Frautschy SA (2007) Neuroprotective effects of curcumin. Adv Exp Med Biol 595:197–212
Yang F, Lim GP, Begum AN, Ubeda OJ, Simmons MR, Ambegaokar SS (2005) Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J Biol Chem 280:5892–5901
Lim GP, Chu T, Yang F, Beech W, Frautschy SA, Cole GM (2001) The curry spice curcumin reduces oxidative damage and amyloid pathology in an Alzheimer transgenic mouse. J Neurosci 21:8370–8377
Kapoor S, Priyadarsini KI (2001) Protection of radiation-induced protein damage by curcumin. Biophys Chem 92(1–2):119–126
Lim GP, Chu T, Yang F, Beech W, Frautschy SA, Cole GM (2001) The curry spice curcumin reduces oxidative damage and amyloid pathology in an Alzheimer transgenic mouse. J Neurosci 21(21):8370–8377
Shukla PK, Khanna VK, Khan MY, Srimal RC (2003) Protective effect of curcumin against lead neurotoxicity in rat. Hum Exp Toxicol 22:653–658
Daniel S, Limpson JL, Dairam A, Watkins GM, Daya S (2004) Through metal binding, curcumin protects against lead and cadmium induced lipid peroxidation in rat brain homogenates and against lead induced tissue damage in rat brain. J Inorg Chem 98:266–275
Duvoix A, Blasius R, Delhalle S, Schnekenburger M, Morceau F, Henry E (2005) Chemopreventive and therapeutic effects of curcumin. Cancer Lett 223:181–190
Rajeswari A, Sabesan M (2008) Inhibition of monoamine oxidase-B by the polyphenolic compound, curcumin and its metabolite tetrahydrocurcumin, in a model of Parkinson’s disease induced by MPTP neurodegeneration in mice. Inflammopharmacology 16:96–99
Aggarwal B, Kumar A, Bhati AC (2003) Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res 23:363–398
Shishodia S, Sethi G, Aggarwal BB (2005) Curcumin: getting back to the roots. Ann N.Y. Sci 1056:206–217
Ramassamy C (2006) Emerging role of polyphenolic compounds in the treatment of neurodegenerative diseases: a review of their intracellular targets. Eur J Pharmacol 545:51–64
Yousef IM, El-Demerdash MM, Radwan FME (2008) Sodium arsenite induced biochemical perturbations in rats: ameliorating effect of curcumin. Food Chem Toxicol 48:3506–3511
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol 186:421–431
Beutler E, Duron O, Kelly BM (1963) An improved method for the detection of blood glutathione. J Lab Clin Med 61:882–888
Sharma M, Gupta YK (2002) Chronic treatment with trans resveratrol prevents intracerebroventricular streptozotocin induced cognitive impairment and oxidative stress in rats. Life Sci 7:2489–2498
Sinha AK (1972) Colorimetric assay of catalase. Anal Biochem 47:389–394
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Carlberg I, Mannervik B (1975) Glutathione reductase levels in rat brain. J Biol Chem 250:5475–5480
Guha Mazumder DN (2005) Effect of chronic intake of arsenic contaminated water on liver. Toxicol Appl Pharmacol 206:169–175
Klaassen CD, Watkin JB (1984) Mechanism of formation, hepatic uptake and biliary excretion. Pharmacol Rev 36:1–67
El-Demerdash FM, Yousef MI, Radwan FME (2009) Ameliorating effect of curcumin on sodium arsenite-induced oxidative damage and lipid peroxidation in different rat organs. Food Chem Toxicol 47:249–254
Halliwell B (2002) Gutteridge JMC. Free radicals in biology and medicine, vol. 3. Oxford University, Oxford, pp 105–245
Mo J, Xia Y, Wade TJ, Schmitt M, Le XC, Dang R, Mumford JL (2006) Chronic arsenic exposure and oxidative stress: OGG1 expression and arsenic exposure nail selenium, and skin hyperkeratosis in Inner Mongolia. Environ Health Perspect 114:835–841
Gupta M, Mazumder UK, Kumar RS, Sivakumar T, Vamsi ML (2004) Antitumor activity and antioxidant status of Caesalpinia bonducella against enrlich ascites carcinoma in Swiss albino mice. J Pharmacol Sci 94:177–184
Niki E, Yoshida Y, Saito Y, Noguchi N (2005) Lipid peroxidation mechanisms, inhibition and biological effects. Biochem Biophys Res Commun 338:668–676
Ferrara N, Henzel WJ (1989) Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells. Biochem Biophys Res Commun 161:851–858
Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey VS, Dvorak HF (1983) Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science 219:983–985
Gross JL, Moscatelli D, Jaffe EA, Rifkin DB (1982) Plasminogen activator and collagenase production by cultured capillary endothelial cells. J Cell Biol 95:974–981
Nagy JA, Brown LF, Senger, Lanir N, Van de Water L, Dvorak AM, Dvorak HF (1989) Pathogenesis of tumor stroma generation: A critical role for leaky blood vessels and fibrin deposition. Biochim Biophys Acta 948:305–326
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, C.Z., Huang, W.Z., Zhang, G. et al. In vivo study on the effects of curcumin on the expression profiles of anti-tumour genes (VEGF, CyclinD1 and CDK4) in liver of rats injected with DEN. Mol Biol Rep 40, 5825–5831 (2013). https://doi.org/10.1007/s11033-013-2688-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-013-2688-y