Abstract
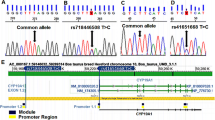
The major limitation to the development of embryo transfer technique in cattle is the highly variable between individuals in ovulatory response to FSH-induced superovulation. The objective of this study was to identify a predictor to forecast superovulation response on the basis of associations between superovulation performance and gene polymorphism, variation in the bovine luteinizing hormone/choriogonadotropin receptor (LHCGR) gene was investigated using PCR-single-strand conformational (PCR-SSCP) and DNA sequencing. Four single nucleotide polymorphisms (SNPs) of G51656T, A51703G, A51726G and G51737A were identified at the intron 9 of the LHCGR gene in 171 Chinese Holstein cows treated for superovulation, and evaluated its associations with superovulatory response. Association analysis showed that these four SNPs had significant effects on the total number of ova (TNO) (P < 0.05). Moreover, the A51703G and A51726G polymorphisms significantly associated with the number of transferable embryos (NTE) (P < 0.05). In addition, significant additive effect on TNO was detected in polymorphisms of G51656T (P < 0.05) and A51703G (P < 0.01), and the A51703G polymorphism also had significant additive effects on NTE (P < 0.01). These results indicate that LHCGR gene is a potential marker for superovulation response and can be used to predict the most appropriate dose of FSH for superovulation in Chinese Holstein cows.


Similar content being viewed by others
References
Rico C, Fabre S, Médigue C, di Clemente N, Clément F, Bontoux M, Touzé JL, Dupont M, Briant E, Rémy B, Beckers JF, Monniaux D (2009) Anti-mullerian hormone is an endocrine marker of ovarian gonadotropin-responsive follicles and can help to predict superovulatory responses in the cow. Biol Reprod 80(1):50–59. doi:10.1095/biolreprod.108.072157
Lerner SP, Thayne WV, Baker RD, Henschen T, Meridith S, Inskeep EK, Dailey RA, Lewis PE, Butcher RL (1986) Age, dose of FSH and other factors affecting superovulation in Holstein cows. J Anim Sci 63:176–183
Mapletoft RJ, Steward KB, Adams GP (2002) Recent advances in the superovulation in cattle. Reprod Nutr Dev 42(6):601–611
Takagi M, Kim IH, Izadyar F, Hyttel P, Bevers MM, Dielman SJ (2001) Impaired final follicles maturation in heifers after superovulation with recombinant human FSH. Reproduction 121:941–951
Bó GA, Baruselli PS, Moreno D, Cutaia L, Caccia M, Tríbulo R, Tríbulo H, Mapletoft RJ (2002) The control of follicular wave development for self-appointed embryo transfer programs in cattle. Theriogenology 57:53–72
Fortune JE, Rivera GM, Yang MY (2004) Follicular development: the role of the follicular microenvironment in selection of the dominant follicle. Anim Reprod Sci 82-83:109–126. doi:10.1016/j.anireprosci.2004.04.031
Gasser CL, Burke CR, Mussard ML, Behlke EJ, Grum DE, Kinder JE, Day ML (2006) Induction of precocious puberty in heifers II: advanced ovarian follicular development. J Anim Sci 84:2042–2049. doi:10.2527/jas.2005-637
Mossa F, Duffy P, Naitana S, Lonergan P, Evans AC (2007) Association between numbers of ovarian follicles in the first follicle wave and superovulatory response in ewes. Anim Reprod Sci 100:391–396. doi:10.1016/j.anireprosci.2006.10.016
Malhi PS, Adams GP, Mapletoft RJ, Singh J (2008) Superovulatory response in a bovine model of reproductive aging. Anim Reprod Sci 109:100–109. doi:10.1016/j.anireprosci.2007.12.002
Tabor HK, Risch NJ, Myers RM (2002) Candidate-gene approaches for studying complex genetic traits: practical considerations. Nat Rev Genet 3:391–397. doi:10.1038/nrg796
Ribadu AY, Nakada K, Moriyoshi M, Zhang WC, Tanaka Y, Nakao T (2000) The role of LH pulse frequency in ACTH-induced ovarian follicular cysts in heifers. Anim Reprod Sci 64(1–2):21–31. doi:10.1016/S0378-4320(00)00196-2
Piersma D, Verhoef-Post M, Look MP, Uitterlinden AG, Pols HA, Berns EM, Themmen AP (2007) Polymorphic variations in exon 10 of the luteinizing hormone receptor: functional consequences and associations with breast cancer. Mol Cell Endocrinol 276:63–70. doi:10.1016/j.mce.2007.06.007
Day ML (2004) Hormonal induction of estrous cycles in anestrous Bos taurus beef cows. Anim Reprod Sci 82–83:487–494. doi:10.1016/j.anireprosci.2004.05.002
Ramirez-Godinez JA, Kiracofe GH, Carnahan DL, Spire MF, Beeman KB, Stevenson JS, Schalles RR (1982) Evidence for ovulation and fertilization in beef cows with short estrous cycles. Theriogenology 17(4):409–414
Cassar CA, Dow MP, Pursley JR, Smith GW (2002) Effect of the preovulatory LH surge on bovine follicular progesterone receptor mRNA expression. Domest Anim Endocrinol 22(3):179–187. doi:10.1016/S0739-7240(02)00124-8
Mihm M, Bleach ECL (2003) Endocrine regulation of ovarian antral follicle development in cattle. Anim Reprod Sci 78:217–237. doi:10.1016/S0378-4320(03)00092-7
Minegishi T, Nakamura K, Yamashita S, Ikeda S, Koguer K (2008) Regulation of human luteinizing hormone receptor in the ovary. Reprod Med Biol 7:11–16
Barros CM, Ereno RL, Simões RA, Fernandes P, Buratini J, Nogueira MF (2010) Use of knowledge regarding LH receptors to improve superstimulatory treatments in cattle. Reprod Fertil Dev 22(1):132–137. doi:10.1071/RD09227
Fauser BC, Diedrich K, Devroey P (2008) Predictors of ovarian response: progress towards individualized treatment in ovulation induction and ovarian stimulation. Hum Reprod Update 14:1–14. doi:10.1093/humupd/dmm034
Tang KQ, Li SJ, Yang WC, Yu JN, Han L, Li X, Yang LG (2011) An MspI polymorphism in the inhibin alpha gene and its associations with superovulation traits in Chinese Holstein cows. Mol Biol Rep 38(1):17–21
Shi YY, He L (2005) SHEsis, a powerful software platform for analyses of linkage disequilibrium, haplotype construction, and genetic association at polymorphism loci. Cell Res 15:97
Liu BH (1998) Statistical genomics: linkage, mapping and QTL analysis. CRC Press, LLC, Boca Raton, pp 404–409
Yang WC, Li SJ, Tang KQ, Hua GH, Zhang CY, Yu JN, Han L, Yang LG (2010) Polymorphisms in the 5′ upstream region of the FSH receptor gene, and their association with superovulation traits in Chinese Holstein cows. Anim Reprod Sci 119:172–177. doi:10.1016/j.anireprosci.2010.02.004
Lindsell CE, Rajkumar K, Manning AW, Emery SK, Mapletoft RJ, Murphy BD (1986) Variability in FSH:LH ratios among batches of commercially available gonadotrophins. Theriogenology 25:167
Chupin D, Combarnous Y, Procureur R (1984) Antagonistic effect of LH on FSH induced superovulation in cattle. Theriogenology 21:229
Krawczak M, Thomas NS, Hundrieser B, Mort M, Wittig M, Hampe J, Cooper DN (2007) Single base-pair substitutions in exon–intron junctions of human genes: nature, distribution and consequences for mRNA splicing. Hum Mutat 28:150–158
Van Laere AS, Nguyen M, Braunschweig M, Nezer C, Collette C, Moreau L, Archibald AL, Haley CS, Buys N, Tally M, Andersson G, Georges M, Andersson L (2003) A regulatory mutation in IGF2 causes a major QTL effect on muscle growth in the pig. Nature 425:832–836. doi:10.1038/nature02064
Niu BY, Ye LZ, Li FE, Deng CY, Jiang SW, Lei MG, Xiong YZ (2008) Identification of polymorphism and association analysis with reproductive traits in the porcine RNF4 gene. Anim Reprod Sci 110(3–4):283–292. doi:10.1016/j.anireprosci.2008.01.020
Pang Y, Wang J, Zhang C, Lei C, Lan X, Yue W, Gu C, Chen D, Chen H (2011) The polymorphisms of bovine VEGF gene and their associations with growth traits in Chinese cattle. Mol Biol Rep 38(2):755–759. doi:10.1007/s11033-010-0163-6
Acknowledgments
This work was by supported by Huazhong Agricultural University Scientific & Technological Self-innovation Foundation (No.2009SC005) and the earmarked fund for Modern Agro-industry Technology Research System (CARS-37-04B).
Author information
Authors and Affiliations
Corresponding author
Additional information
Wu-Cai Yang, and Ke-Qiong Tang are the Co-first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, WC., Tang, KQ., Li, SJ. et al. Polymorphisms of the bovine luteinizing hormone/choriogonadotropin receptor (LHCGR) gene and its association with superovulation traits. Mol Biol Rep 39, 2481–2487 (2012). https://doi.org/10.1007/s11033-011-0999-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-011-0999-4