Abstract
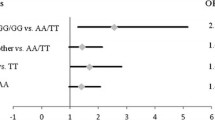
Prostate cancer is the most commonly diagnosed cancer in men worldwide and is the second leading cause of cancer related mortality. Genetic background may account for the difference in susceptibility of individuals to different diseases and the relationship between genetic polymorphism and some diseases has been extensively studied. There are several common polymorphisms in genes encoding DNA repair enzymes, some of these polymorphisms are reported to result in subtle structural alterations of the repair enzyme and modulation of the repair capacity. The aim of the present study was to analyze the effect of XPG Asp 1104His and XRCC1 Arg309Gln polymorphisms on risk of prostate cancer in north Indian population. Statistically significant increased risk of prostate cancer was observed on individuals that posses His/His genotype of XPG (OR 2.53, 95% CI 0.99–6.56, P = 0.031). In this study 150 prostate cancer diagnosed patients, 150 healthy controls and 150 BPH (benign prostate hyper plasia) were recruited from north Indian population. Moreover, individuals that carried the Gln/Gln genotype of XRCC1 also showed statistically increased risk of prostate cancer (OR 2.06, 95% CI 1.07–4.00, P = 0.033). The Asp/Asp of XPG and Gln/Gln of XRCC1 in combination showed statistically increased risk of prostate cancer in cases (OR 3.29, 95% CI 1.09–10.16, P = 0.032).




Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, Thun MJ (2008) Cancer statistics. CA Cancer J Clin 58:71–96
Grant WB (2004) A multi country ecologic study of risk and risk reduction factors for prostate cancer mortality. Eur Urol 45:271–279
Goode EL, Ulrich CM, Potter JD (2202) Polymorphisms in DNA repair genes and associations with cancer risk. Cancer Epidemiol Biomark Prev 11:1513–1530
Shields PG, Harris CC (2000) Cancer risk and low-penetrance susceptibility genes in gene-environment interactions. J Clin Oncol 18:2309–2315
Berwick M, Vineis P (2000) Markers of DNA repair and susceptibility to cancer in humans: an epidemiologic review. J Natl Cancer Inst 92:874–897
Kiyohara C, Yoshimasu K (2007) Genetic polymorphisms in the nucleotide excision repair pathway and lung cancer risk: a meta analysis. Int J Med Sci 4:59–71
Klungland A, Hoss M, Gunz D, Constantinou A, Clarkson G, Doetsch W, Bolton H, Wood D, Lindahl T (1999) Base excision repair of oxidative DNA damage activated by XPG protein. Mol Cell 3:33–42
Cleoment V, Dunand-Sauthier I, Clarkson SG (2006) Suppression of UV-induced apoptosis by the human DNA repair protein XPG. Cell Death Differ 13:478–488
Jeon HS, Kim KM, Park SH, Lee SY, Choi JE, Lee GY, Kam S, Park RW, Kim IS, Kim CH, Jung TH, Park JY (2003) Relationship between XPG codon 1104 polymorphism and risk of primary lung cancer. Carcinogenesis 24:168–1677
Cui Y, Morgenstern H, Greenland S, Tashkin DP, Mao J, Cao W, Cozen W, Mack TM, Zhang ZF (2006) Polymorphism of xeroderma pigmentosum group G and the risk of lung cancer and squamous cell carcinomas of the oropharynx, larynx and esophagus. Int J Cancer 118:714–720
He X, Ye F, Zhang J, Cheng Q, Shen J, Chen H (2008) Susceptibility of XRCC3, XPD, and XPG genetic variants to cervical carcinoma. Pathobiology 75:356–363
Thompson LH, West MG (2000) XRCC1 keeps DNA from getting stranded. Mutat Res 459:1–18
Dianova II, Sleeth KM, Allinson SL, Parsons JL, Breslin C, Caldecott KW, Dianov GL (2004) XRCC1-DNA polymerase beta interaction is required for efficient base excision repair. Nucleic Acids Res 32:2550–2555
Whitehouse CJ, Taylor RM, Thistlethwaite A, Zhang H, Karimi-Busheri F, Lasko DD, Weinfeld M, Caldecott KW (2001) XRCC1 stimulates human polynucleotide kinase activity at damaged DNA termini and accelerates DNA single-strand break repair. Cell 104(1):107–117
Goode EL, Ulrich CM, Potter JD (2002) Polymorphisms in DNA repair genes and associations with cancer risk. Cancer Epidemiol Biomark Prev 11:1513–1530
Roe BA, Crabtree JS, Khan AS (2008) Methods for DNA isolation. Part III. In: Protocols for recombinant DNA isolation, cloning, and sequencing [Internet edition]. University of Oklahoma, Norman. Available at http://www.genome.ou.edu/protocol_book/protocol-index.html. Accessed on 27 Sept 2008
Duell EJ, Wiencke JK, Cheng TJ, Varkonyi A, Zuo ZF, Ashok TD, Mark EJ, Wain JC, Christian DC, Kelsey KT (2000) Polymorphisms in the DNA repair genes XRCC1 and ERCC2 and biomarkers of DNA damage in human blood mononuclear cells. Carcinogenesis 21:965–971
Lichtenstein P, Holm NV, Verkasalo PK, Iliadou A, Kaprio J, Koskenvuo M (2000) Environmental and heritable factors in the causation of cancer—analyses of cohorts of twins from Sweden, Denmark, and Finland. N Engl J Med 343(2):78–85
Pathak SK, Sharma RA, Steward WP, Mellon JK, Griffiths TR, Gescher AJ (2005) Oxidative stress and cyclooxygenase activity in prostate carcinogenesis: targets for chemopreventive strategies. Eur J Cancer 41(1):61–70
Khandrika L, Kumar B, Koul S, Maroni P, Koul HK (2009) Oxidative stress in prostate cancer. Cancer Lett 282(2):125–136
Cooke MS, Evans MD, Dizdaroglu M, Lunec J (2003) Oxidative DNA damage: mechanisms, mutation, and disease. Faseb J 17(10):1195–1214
Sikka SC (2003) Role of oxidative stress response elements and antioxidants in prostate cancer pathobiology and chemoprevention—a mechanistic approach. Curr Med Chem 10(24):2679–2692
Rybicki BA, Nock NL, Savera AT, Tang D, Rundle A (2006) Polycyclic aromatic hydrocarbon-DNA adduct formation in prostate carcinogenesis. Cancer Lett 239(2):157–167
Tang D, Liu JJ, Rundle A, Neslund-Dudas C, Savera AT, Bock CH (2007) Grilled meat consumption and PhIP-DNA adducts in prostate carcinogenesis. Cancer Epidemiol Biomark Prev 16(4):803–808
Christmann M, Tomicic MT, Roos WP, Kaina B (2003) Mechanisms of human DNA repair: an update. Toxicology 193(1–2):3–34
Hyytinen ER, Frierson HF Jr, Sipe TW, Li CL, Degeorges A, Sikes RA (1999) Loss of heterozygosity and lack of mutations of the XPG/ERCC5 DNA repair gene at 13q33 in prostate cancer. Prostate 41:190–195
Hooker S, Bonilla C, Akereyeni F, Ahaghotu C, Kittles RA (2007) NAT2 and NER genetic variants and sporadic prostate cancer susceptibility in African–Americans. Prostate Cancer Prostatic Dis 11(4):349–356
Cheng L, Spitz MR, Hong WK, Wei Q (2000) Reduced expression levels of nucleotide excision repair genes in lung cancer: a case–control analysis. Carcinogenesis 21:1527–1530
Sanyal S, Festa F, Sakano S, Zhang Z, Steineck G, Norming U, Wijkstrom H, Larsson P, Kumar R, Hemmink K (2004) Polymorphisms in DNA repair and metabolic genes in bladder cancer. Carcinogenesis 25:729–734
Sasaki M, Sakano S, Okayama N, Akao J, Hara T, Kawai Y, Ohmi C, Hinoda Y, Naito K (2008) DNA repairing gene polymorphism may be associated with prognosis of upper urinary tract transitional cell carcinoma. Neoplasia 10:255–265
Sobti RC, Berhane N, Mahdi SA, Kler R, Hussien AS, Kutat V, Wanchu A (2010) Association and impact of XPG gene polymorphism in HIV1 disease progression to AIDS among north Indian seropositive individuals. Mol Biol Rep 37:317–324
Li C, Hu Z, Liu Z, Wang LE, Strom SS, Gershenwald JE, Lee JE, Ross MI, Mansfield PF, Cormier JN, Prieto VG, Duvic M, Grimm EA, Wei Q (2006) Polymorphisms in the DNA repair genes XPC, XPD, and XPG and risk of cutaneous melanoma: a case–control analysis. Cancer Epidemiol Biomark Prev 15(12):2526–2532
Barrett-Connor E, Khaw KT (1987) Cigarette smoking and increased endogenous estrogen levels in men. Am J Epidemiol 126:187–192
Michnovicz JJ, Hershcopf RJ, Haley NJ, Bradlow HL, Fishman J (1989) Cigarette smoking alters hepatic estrogen metabolism in men: implications for atherosclerosis. Metabolism 38:537–541
Daniell HW (1995) A worse prognosis for smokers with prostate cancer. J Urol 154:153–157
Shen H, Xu Y, Yu R, Qin Y, Zhou L, Wang X, Spitz MR, Wei Q (2000) Polymorphism in DAN repair gene XRCC1 and risk of gastric cancer in a Chinese population. Int J Cancer 88:601–606
Divine EK, Gilliland ED, Crowell RE, Stidley CA, Bocklage TJ, Strom SS, Spitz MR (2001) Polymorphisms of DNA repair gene XRCC1 glutamine allele is a risk factor for adenocarcinoma of the lung. Mutat Res 461:273–278
Park KS, Mok JW, Ko HE, Tokunaga K, Lee MH (2002) Polymorphisms of tumour necrosis factors A and B in breast cancer. Eur J Immunogenet 29:7–10
Wang Y, Spitz MR, Zhu Y, Dong Q, Shete S, Wu X (2003) From gene type to phenotype: correlating XRCC1 polymorphisms with mutagen sensitivity. DNA Repair 2:901–908
Rybicki BA, Conti DV, Moreira A, Cicek M, Casey G, Witte JS (2004) DNA repair gene XRCC1 and XPD polymorphisms and risk of prostate cancer. Cancer Epidemiol Biomark Prev 13:23–29
Lodish H, Berk A, Matsudaira P, Kaiser CA, Krieger M, Scott MP, Zipursky SL, Darnell J (2004) Molecular Biology of the Cell, 5th edn. WH Freeman, New York P963
Geng J, Zhang YW, Huang GC, Chen LB (2008) XRCC1 genetic polymorphism Arg399Gln and gastric cancer risk: a meta-analysis. World J Gastroenterol 14(43):6733–6737
Kiyohara C, Takayama K, Nakanishi Y (2006) Association of genetic polymorphisms in the base excision repair pathway with lung cancer risk: a meta-analysis. Lung Cancer 54:267–283
Saadat M, Ansari-Lari M (2008) Polymorphism of XRCC1 (at codon 399) and susceptibility to breast cancer, a meta-analysis of the literatures. Breast Cancer Res Treat 115:137–144
Sakano S, Hinoda Y, Okayama N, Kawai Y, Korenaga Y, Eguchi S, Nagao K, Ohmi C, Naito K (2007) The association of DNA repair gene polymorphisms with the development and progression of renal cell carcinoma. Annals of oncology doi:10.1093/annonc/mdm337
Acknowledgments
The authors of this article are thankful to the clinical staff of the department of urology, post graduate institute of medical education and research, Chandigarh, Government College of Medical Education and hospital (Patiala) and All Indian Institute of Medical Science, New Delhi for the kind assistance in collecting samples and providing valuable data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Berhane, N., Sobti, R.C. & Mahdi, S.A. DNA repair genes polymorphism (XPG and XRCC1) and association of prostate cancer in a north Indian population. Mol Biol Rep 39, 2471–2479 (2012). https://doi.org/10.1007/s11033-011-0998-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-011-0998-5