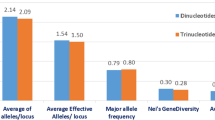
Abstract
Asparagus bean (V. unguiculata ssp. sesquipedialis), a specific form of cowpea (V. unguiculata L. Walp.), is cultivated as a vegetable crop throughout eastern and southern Asia for its tender long pods. Little is known about the genetic relationship between asparagus bean and the broader species, particularly the dominant ssp. unguiculata. We report here the development and transferability of simple sequence repeat (SSR) markers, over 40% of which are EST-derived, from ssp. unguiculata to asparagus bean and the use of a subset of the polymorphic markers to assess the genetic diversity of asparagus bean cultivars from diverse geographic origins across China. A total of 410 EST derived SSR (eSSR) markers and 600 SSR markers derived from cowpea genespace sequences (GSS) were developed, with a cross-subspecies transferability of 100% and 98.5%, respectively. In a recombinant inbred line population of asparagus bean, a 1:1 segregation was observed for most loci. Principal coordinate analysis (PCA) and phylogenetic clustering based on 62 alleles detected by 14 polymorphic SSR markers distinguished ssp. unguiculata and sesquipedialis into separate groups. Improved asparagus bean cultivars in China generally have a narrow genetic basis compared with landraces varieties. This suggests that asparagus bean breeding programs need to consider utilizing landrace germplasm to enhance genetic variability and ensure long-term gains from selection and reduce genetic vulnerability to pathogen/pest epidemics. Because of their transferability across subspecies, the SSR markers described in this study could be effectively employed in cross-subspecies trait introgression breeding from ssp. unguiculata to sesquipedialis.




Similar content being viewed by others
Abbreviations
- AFLP:
-
Amplified fragment length polymorphism
- EST:
-
Expressed sequence tag
- GSS:
-
Gene space sequence
- PCA:
-
Principal coordinate analysis
- PIC:
-
Polymorphism information content
- RAPD:
-
Rapid amplification polymorphism of DNA
- RFLP:
-
Restriction fragment length polymorphism
- RIL:
-
Recombinant inbred line
- SFP:
-
Single feature polymorphisms
- SSR:
-
Simple sequence repeat
References
Anderson JA, Churchill GA, Autrique JE, Tanksley SD, Sorrells ME (1993) Optimizing parental selection for genetic linkage maps. Genome 36:181–186
Arumuganathan K, Earle ED (1991) Nuclear DNA content of some important plant species. Plant Mol Biol Rep 9:208–218
Barrett B, Griffiths A, Schreiber M, Ellison N, Mercer C, Bouton J, Ong B, Forster J, Sawbridge T, Spangenberg G, Bryan G, Woodfield D (2004) A microsatellite map of white clover. Theor Appl Genet 109:596–608
Bassam BJ, Gaetano-Anollé G, Gresshoff PM (1991) Fast and sensitive silver staining of DNA in polyacrylamide gels. Anal Biochem 196:80–83
Chen XF, Laudeman TW, Rushton PJ, Thomas TA, Timko MP (2007) CGKB: an annotation knowledge base for cowpea (Vigna unguiculata L.) methylation filtered genomic genespace sequences. BMC Bioinformatics 8:129
Choudhary S, Sethy NK, Shokeen B, Bhatia S (2009) Development of chickpea EST-SSR markers and analysis of allelic variation across related species. Theor Appl Genet 118:591–608
Coulibaly S, Pasquet RS, Papa R, Gepts P (2002) AFLP analysis of the phenetic organization and genetic diversity of cowpea [Vigna unguiculata (L.) Walp.] reveals extensive gene flow between wild and domesticated types. Theor Appl Genet 104:258–266
Das S, Bhat PR, Sudhakar C, Ehlers JD, Wanamaker S, Roberts PA, Cui XP, Close TJ (2008) Detection and validation of single feature polymorphisms in cowpea (Vigna unguiculata L. Walp) using a soybean genome array. BMC Genomics 9:107
Dudley JW (1982) Theory for transfer of alleles. Crop Sci 22:631–637
Ehlers JD, Hall AE (1997) Cowpea (Vigna unguiculata L. Walp). Field Crops Res 53:187–204
Ehlers JD, Diop NN, Fenton R, Muchero W, Bhat P, Pottorff M, Wanamaker S, et al. (2009) Development of high-throughput SNP genotyping platform for cowpea to enable modern breeding, diversity analysis and genetic fingerprinting. In: Proceedings of the American Society Horticultural Science Annu. Meeting Southern Region. Am Soc Hortic Sci, Alexandria, VA
Fall L, Diouf D, Fall-Ndiaye MA, Badiane FA, Gueye M (2003) Genetic diversity in cowpea [Vigna unguiculata (L.) Walp.] varieties determined by ARA and RAPD techniques. Afr J Biotechnol 2:48–50
Fang JG, Chao CCT, Roberts PA, Ehlers JD (2007) Genetic diversity of cowpea [Vigna unguiculata (L.) Walp.] in four West African and USA breeding programs as determined by AFLP analysis. Genet Resour Crop Evol 54:1197–1209
Farisa JD, Laddomada B, Gilla BS (1998) Molecular mapping of segregation distortion loci in Aegilops tauschii. Genetics 149:319–327
Fatokun CA, Danesh D, Young ND, Stewart EL (1993) Molecular taxonomic relationships in the genus Vigna based on RFLP analysis. Theor Appl Genet 86:97–104
Hanai LR, de Campos T, Camargo LE, Benchimol LL, de Souza AP, Melotto M et al (2007) Development, characterization, and comparative analysis of polymorphism at common bean SSR loci isolated from genic and genomic sources. Genome 50:266–277
Hisano H, Sato S, Isobe S, Sasamoto S, Wada T, Matsuno A, Fujishiro T, Yamada M, Nakayama S, Nakamura Y, Watanabe S, Harada K, Tabata S (2007) Characterization of the soybean genome using EST-derived microsatellite markers. DNA Res 14:271–281
Jenczewski E, Gherardi M, Bonnin I, Prosperi JM, Olivieri I, Huguet T (1997) Insight on segregation distortion in two intraspecific crosses between annual species of Medicago (Leguminosae). Theor Appl Genet 94:682–691
Kantety RV, Rota ML, Matthews DE, Sorrells ME (2002) Data mining for simple sequence repeats in expressed sequence tags from barley, maize, rice, sorghum and wheat. Plant Mol Biol 48:501–510
Li CD, Fatokun CA, Ubi B, Singh BB, Scoles GJ (2001) Determining genetic similarities and relationships among cowpea breeding lines and cultivars by microsatellite markers. Crop Sci 41:189–197
Lu H, Romero-Severson J, Bernardo R (2002) Chromosomal regions associated with segregation distortion in maize. Theor Appl Genet 105:622–628
Ochanda N, Yu J, Bramel PJ, Menkir A, Tuinstra MR, Witt MD (2009) Selection before backcross during exotic germplasm introgression. Field Crops Res 112:37–42
Panella L, Gepts P (1992) Genetic relationship within Vigna unguiculata (L.) Walp. based on isozyme analyses. Genet Resour Crop Evol 39:71–88
Pasquet RS (1999) Genetic relationships among subspecies of Vigna unguiculata (L.) Walp. Based on allozyme variation. Theor Appl Genet 98:1104–1119
Pasquet RS (2000) Allozyme diversity of cultivated cowpea Vigna unguiculata (L.) Walp. Theor Appl Genet 101:211–219
Rohlf FJ (2000) NTSYSpc, numerical taxonomy and multivariate analysis system, version 2.1 user guide. Exeter Software, Setauket
Singh BB (2002) Recent genetic studies in cowpea. In: Fatokun CA, Tarawali SA, Singh BB, Kormawa PM, Tamo M (eds) Challenges and opportunities for enhancing sustainable cowpea production. International Institute of Tropical Agriculture, Ibadan, pp 3–13
Timko MP, Ehlers JD, Roberts PA (2007) Cowpea. In: Kole C (ed) Pulses, sugar and tuber crops. Theoretical and applied genetics, genome mapping and molecular breeding in plants, vol 3. Springer, Berlin, pp 49–67
Timko MP, Rushton PJ, Laudeman TW, Bokowiec MT, Chipumuro E, Foo Cheung F, Town CD, Chen XF (2008) Sequencing and analysis of the gene-rich space of cowpea. BMC Genomics 9:103
Acknowledgments
This study was partially supported by the National Science Foundation of China (No. 30871711), the Science Foundation of Zhejiang Province (No. Y306224), and the fund from Science and Technology Department of Zhejiang Province, China (No. 2007C24018). The authors would thank Dr. Dave Edwards of University of Queensland, Australia, for his help in bioinformatics.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Xu, P., Wu, X., Wang, B. et al. Development and polymorphism of Vigna unguiculata ssp. unguiculata microsatellite markers used for phylogenetic analysis in asparagus bean (Vigna unguiculata ssp. sesquipedialis (L.) Verdc.). Mol Breeding 25, 675–684 (2010). https://doi.org/10.1007/s11032-009-9364-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-009-9364-x