Abstract
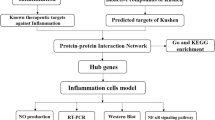
The dried rhizome of Musa basjoo Sieb. et Zucc. is Rhizoma Musae. It has been used to treat diabetes in Miao medicine in China. Lupenone was isolated from Rhizoma Musae and has good anti-diabetic activity. Its mechanism of action is unclear. Diabetes is a chronic low-level systemic inflammatory disease, and lupenone has anti-inflammatory activity, but the underlying mechanism is not fully elucidated. In this study, we aimed to construct the drug-target biologic network and predict the anti-inflammatory mechanism of lupenone. The network-based pharmacologic analysis platform was used to identify the target proteins related to inflammation. Furthermore, the effects of lupenone on acute, subacute and diabetic pancreatic inflammation were evaluated. The “component-target-disease” network was constructed using Cytoscape. Lupenone could regulate transcription factor p65, NF-kappa-B inhibitor alpha, transcription factor AP-1, NF-kappa-B essential modulator, nuclear factor NF-kappa-B p105 subunit, epidermal growth factor receptor, hypoxia-inducible factor 1-alpha and other proteins related to the PI3K-Akt, Toll-like receptor and NF-kappa B signaling pathways. In addition, lupenone significantly decreased acute and subacute inflammation in mice as well as the IL-1β and IFN-γ levels in the pancreas of diabetic rats. The above results provide strong support for studying the molecular mechanism of lupenone in the treatment of diabetes from the perspective of anti-inflammation.
Graphical abstract





Similar content being viewed by others
References
Ogurtsova K, da Rocha Fernandes JD, Huang Y, Linnenkamp U, Guariguata L, Cho NH, Cavan D, Shaw JE, Makaroff LE (2017) IDF Diabetes Atlas: global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res Clin Pract 128:40–50. https://doi.org/10.1016/j.diabres.2017.03.024
Peiró C, Lorenzo Ó, Carraro R, Sánchez-Ferrer CF (2017) IL-1β inhibition in cardiovascular complications associated to diabetes mellitus. Front Pharmacol 8:363. https://doi.org/10.3389/fphar.2017.00363
Chatterjee S, Khunti K, Davies MJ (2017) Type 2 diabetes. Lancet 10085:2239–2251. https://doi.org/10.1016/S0140-6736(17)30058-2
Na M, Kim BY, Osada H, Ahn JS (2009) Inhibition of protein tyrosine phosphatase 1B by lupeol and lupenone isolated from Sorbus commixta. J Enzym Inhib Med Chem 4:1056–1059. https://doi.org/10.1080/14756360802693312
Xu F, Wu H, Wang X, Ye Y, Wang YM, Qian HB, Zhang YY (2014) RP-HPLC characterization of lupenone and β-sitosterol in Rhizoma Musae and evaluation of the anti-diabetic activity of lupenone in diabetic Sprague–Dawley rats. Molecules 9:14114–14127. https://doi.org/10.3390/molecules190914114
Wu HM, Xu F, Hao JJ, Yang Y, Wang XP (2015) Antihyperglycemic activity of banana (Musa nana Lour.) peel and its active ingredients in alloxan-induced diabetic mice. In: 3rd international conference on material, mechanical and manufacturing engineering, pp 231–238. https://doi.org/10.2991/ic3me-15.2015.44
Wu HM, Xu F, Wang YM, Qian HB, Wang XP (2017) Influence of general situation, glucose tolerance and insulin tolerance for lupenone in insulin resistance of type 2 diabetes rats. Lishizhen Med Mater Med Res 5:1035–1037
Kahn SE (2003) The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of type 2 diabetes. Diabetologia 46:3–19. https://doi.org/10.1007/s00125-002-1009-0
Khodabandehloo H, Gorgani-Firuzjaee S, Panahi G, Meshkani R (2016) Molecular and cellular mechanisms linking inflammation to insulin resistance and β-cell dysfunction. Transl Res 1:228–256. https://doi.org/10.1016/j.trsl.2015.08.011
Eguchi K, Nagai R (2017) Islet inflammation in type 2 diabetes and physiology. J Clin Invest 1:14–23. https://doi.org/10.1172/JCI88877
Donath MY, Shoelson SE (2011) Type 2 diabetes as an inflammatory disease. Nat Rev Immunol 2:98–107. https://doi.org/10.1038/nri2925
Marzban L (2015) New insights into the mechanisms of islet inflammation in type 2 diabetes. Diabetes 64:1094–1096. https://doi.org/10.2337/db14-1903
Eizirik DL, Colli ML, Ortis F (2009) The role of inflammation in insulitis and β-cell loss in type 1 diabetes. Nat Rev Endocrinol 4:219. https://doi.org/10.1038/nrendo.2009.21
Xue JT, Shi YL, Li CY, Song HJ (2018) Network pharmacology-based prediction of the active ingredients, potential targets, and signaling pathways in compound Lian-Ge granules for treatment of diabetes. J Cell Biochem. https://doi.org/10.1002/jcb.27933
Ge Q, Chen L, Tang M, Zhang S, Liu L, Gao L, Ma S, Liu L, Kong M, Yao Q, Chen K, Feng F (2018) Analysis of mulberry leaf components in the treatment of diabetes using network pharmacology. Eur J Pharmacol 833:50–62. https://doi.org/10.1016/j.ejphar.2018.05.021
Pereira AS, Bester MJ, Apostolides Z (2017) Exploring the anti-proliferative activity of Pelargonium sidoides DC with in silico target identification and network pharmacology. Mol Divers 4:809–820. https://doi.org/10.1007/s11030-017-9769-0
Wang XP, Hao JJ, Xu SN (2012) The chemical constituents in ethyl acetate extraction from the Rhizoma Musae. Lishizhen Med Mater Med Res 23:515–516
Ahn EK, Oh JS (2013) Lupenone isolated from Adenophora triphylla var. japonica extract inhibits adipogenic differentiation through the downregulation of PPARγ in 3T3-L1 Cells. Phytother Res 27:761–766. https://doi.org/10.1002/ptr.4779
Ru J, Li P, Wang J, Zhou W, Li B, Huang C, Li P, Guo Z, Tao W, Yang Y, Xu X, Li Y, Wang Y, Yang L (2014) TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J Cheminform 1:13. https://doi.org/10.1186/1758-2946-6-13
Wang X, Shen Y, Wang S, Li S, Zhang W, Liu X, Lai L, Pei J, Li H (2017) PharmMapper 2017 update: a web server for potential drug target identification with a comprehensive target pharmacophore database. Nucleic Acids Res 45(W1):356–360. https://doi.org/10.1093/nar/gkx374
Chen Y, Wei J, Zhang Y, Sun W, Li Z, Wang Q, Xu X, Li C, Li P (2018) Anti-endometriosis mechanism of Jiawei Foshou San based on network pharmacology. Front Pharmacol 9:811. https://doi.org/10.3389/fphar.2018.00811
da Huang W, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4:44–57. https://doi.org/10.1038/nprot.2008.211
Hsin KY, Matsuoka Y, Asai Y, Kamiyoshi K, Watanabe T, Kawaoka Y, Kitano H (2016) systemsDock: a web server for network pharmacology-based prediction and analysis. Nucleic Acids Res 44(W1):507–513. https://doi.org/10.1093/nar/gkw335
Saeed MK, Deng Y, Dai R, Li W, Yu Y, Iqbal Z (2010) Appraisal of antinociceptive and anti-inflammatory potential of extract and fractions from the leaves of Torreya grandis Fort Ex. Lindl. J Ethnopharmacol 2:414–418. https://doi.org/10.1016/j.jep.2009.10.024
Wilches I, Tobar V, Peñaherrera E, Cuzco N, Jerves L, Vander Heyden Y, León-Tamariz F, Vila E (2015) Evaluation of anti-inflammatory activity of the methanolic extract from Jungia rugosa leaves in rodents. J Ethnopharmacol 173:166–171. https://doi.org/10.1016/j.jep.2015.07.004
Xu F, Huang X, Wu H, Wang X (2018) Beneficial health effects of lupenone triterpene: a review. Biomed Pharmacother 103:198–203. https://doi.org/10.1016/j.biopha.2018.04.019
Hopkins AL (2008) Network pharmacology: the next paradigm in drug discovery. Nat Chem Biol 11:682. https://doi.org/10.1038/nchembio.118
Stumvoll M, Goldstein BJ, van Haeften TW (2005) Type 2 diabetes: principles of pathogenesis and therapy. Lancet 365:1333. https://doi.org/10.1016/S0140-6736(05)61032-X
Hong YS, Chang Y, Ryu S, Cainzos-Achirica M, Kwon MJ, Zhang Y, Chol Y, Ahn J, Rampal S, Zhao D, Pastor-Barriuso R, Lazo M, Shin H, Cho J, Guallar E (2017) Hepatitis B and C virus infection and diabetes mellitus: a cohort study. Sci Rep UK 1:4606. https://doi.org/10.1038/s41598-017-04206-6
Hum J, Jou JH, Green PK, Berry K, Lundblad J, Hettinger BD, Chang M, Loannou GN (2017) Improvement in glycemic control of type 2 diabetes after successful treatment of hepatitis C virus. Diabetes Care 1:85. https://doi.org/10.2337/dc17-0485
Stone VM, Hankaniemi MM, Svedin E, Sioofy-Khojine A, Oikarinen S, Hyöty H, Laitinen OH, Hytönen VP, Flodström-Tullberg M (2018) A Coxsackievirus B vaccine protects against virus-induced diabetes in an experimental mouse model of type 1 diabetes. Diabetologia 2:476–481. https://doi.org/10.1007/s00125-017-4492-z
Yasukawa K, Yu SY, Yamanouchi S, Takido M, Akihisa T, Tamura T (1995) Some lupane-type triterpenes inhibit tumor promotion by 12-O-tetradecanoylphorbol13-acetate in two-stage carcinogenesis in mouse skin. Phytomedicine 4:309–313. https://doi.org/10.1016/S0944-7113(11)80008-5
Boni-Schnetzler M, Thorne J, Parnaud G, Marselli L, Ehses JA, Kerr-Conte J, Pattou F, Halban PA, Weir GC, Donath MY (2008) Increased interleukin (IL)-1beta messenger ribonucleic acid expression in beta-cells of individuals with type 2 diabetes and regulation of IL-1beta in human islets by glucose and autostimulation. J Clin Endocr Metab 10:4065–4074. https://doi.org/10.1210/jc.2008-0396
Sloan-Lancaster J, Abu-Raddad E, Polzer J, Miller JW, Scherer JC, De Gaetano A, Berg JK, Landschulz WH (2013) Double-blind, randomized study evaluating the glycemic and anti-inflammatory effects of subcutaneous LY2189102, a neutralizing IL-1β antibody, in patients with type 2 diabetes. Diabetes Care 8:2239–2246. https://doi.org/10.2337/dc12-1835
Cavelti-Weder C, Timper K, Seelig E, Keller C, Osranek M, Lässing U, Spohn G, Maurer P, Müller P, Jennings GT, Willers J, Saudan P, Donath MY, Bachmann MF (2016) Development of an interleukin-1β vaccine in patients with type 2 diabetes. Mol Ther 5:1003–1012. https://doi.org/10.1038/mt.2015.227
Kiernan K, MacIver NJ (2018) Viral infection “interferes” with glucose tolerance. Immunity 1:6–8. https://doi.org/10.1016/j.immuni.2018.06.013
Acknowledgements
This work was supported by Guiyang University of Chinese Medicine Doctor Startup Fund (2017), NSFC (81860737), The Guizhou domestic first-class construction project [(Chinese Materia Medica) (GNYL [2017] 008)] and Guizhou Province Science and Technology Project (QiankeheLHzi [2016] 7116).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Human and animal rights
This research involves animals, and the experiments were approved by the Animal Ethics Committee for animal experimentation.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, F., Yang, L., Huang, X. et al. Lupenone is a good anti-inflammatory compound based on the network pharmacology. Mol Divers 24, 21–30 (2020). https://doi.org/10.1007/s11030-019-09928-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-019-09928-5