Abstract
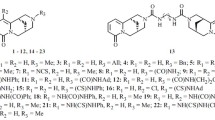
Twenty benzimidazole derivatives bearing in position 1 a (\(\omega \)-tert-amino)alkyl chain (mainly quinolizidin-1-ylmethyl) and in position 2 an aromatic moiety (phenyl, benzyl or benzotriazol-1/2-ylmethyl) were evaluated at the National Cancer Institute (NCI) for anti-proliferative activity against a panel of 60 human cancer cell lines. Four compounds (6, 7, 9 and 10) displayed a large spectrum of activity with \(\text{ GI }_{50}<\) 10 \(\upmu \text{ M }\) on 24–57 cell lines, while thirteen compounds exhibited sub-micromolar or even nanomolar activity against single cell lines, such as leukemia CCRF-CEM, HL-60 and MOLT-4, CNS cancer SF-268 and, particularly, renal cancer UO-31, sometimes with outstanding selectivity (compounds 5-7, 11, 13 and 18).



Similar content being viewed by others
References
Pagani F, Sparatore F (1965) Benzotriazolyl-alkyl-benzimidazoles and their dialkyl-aminoalkyl derivatives. Boll Chim Farm 104:427–431
Sparatore F, Boido V, Fanelli F (1968) Dialkylaminoalkylbenzimidazoles of pharmacological interest. Farmaco Ed Sci 23:344–359
Paglietti G, Sparatore F (1972) Dialkylaminoalkylbenzimidazoles of pharmacological interest. III. Farmaco Ed Sci 27:333–342
Paglietti G, Boido V, Sparatore F (1975) Dialkylaminoalkylbenzimidazoles of pharmacological interest. IV. Farmaco Ed Sci 30:505–511
Boido A, Vazzana I, Sparatore F, Cenicola ML, Donnoli D, Marmo E (1991) Preparation and pharmacological activity of some 1-lupinylbenzimidazoles and 1-lupinylbenzotriazoles. Farmaco 46:775–788
Sparatore F (1968) 2-Benzyl-1(1-quinolizidinylmethyl)-5-(trifluoromethyl)-benzimidazoles. US Pat 3394141 (July 23, 1968); Chem Abstr 69:86995
Sun Q, Gatto B, Yu C, Liu A, Liu LF, LaVoie EJ (1994) Structure activity of topoisomerase I poisons related to Hoechst 33342. Bioorg Med Chem Lett 4:2871–2876. doi:10.1016/S0960-894X(01)80831-3
Sun Q, Gatto B, Yu C, Liu A, Liu LF, LaVoie EJ (1995) Synthesis and evaluation of terbenzimidazoles as topoisomerase I inhibitors. J Med Chem 38:3638–3644. doi:10.1021/jm00018a024
Kim JS, Gatto B, Yu C, Liu A, Liu LF, LaVoie EJ (1996) Substituted 2,5’-bi-1H-benzimidazoles: topoisomerase I inhibition and cytotoxicity. J Med Chem 39:992–998. doi:10.1021/jm950412w
Kim JS, Yu C, Liu A, Liu LF, LaVoie EJ (1997) Terbenzimidazoles: influence of 2”-, 4-, and 5-substituents on cytotoxicity and relative potency as topoisomerase I poisons. J Med Chem 40:2818–2824. doi:10.1021/jm960658g
Palmer BD, Smaill JB, Boyd M, Boschelli DH, Doherty AM, Hamby JM, Khatana SS, Kramer JB, Kraker AJ, Panek RL, Lu GH, Dahring TK, Winters RT, Showalter HDH, Denny WA (1998) Structure-activity relationships for 1-phenylbenzimidazoles as selective ATP site inhibitors of the platelet-derived growth factor receptor. J Med Chem 41:5457–5465. doi:10.1021/jm9804681
Palmer BD, Kraker AJ, Hartl BG, Panopoulos AD, Panek RL, Batley BL, Lu GH, Trumpp-Kallmeyer S, Showalter HDH, Denny WA (1999) Structure-activity relationships for 5-substituted 1-phenylbenzimidazoles as selective inhibitors of the platelet-derived growth factor receptor. J Med Chem 42:2373–2382. doi:10.1021/jm980658b
White AW, Almassy R, Calvert AH, Curtin NJ, Griffin RJ, Hostomsky Z, Maegley K, Newell DR, Srinivasan S, Golding BT (2000) Resistance-modifying agents. 9. Synthesis and biological properties of benzimidazole inhibitors of the DNA repair enzyme poly(ADP-ribose)polymerase. J Med Chem 43:4084–4097. doi:10.1021/jm000950v
Xu YJ, Miao HQ, Pan W, Navarro EC, Tonra JR, Mitelman S, Camara MM, Deevi DS, Kiselyov AS, Kussie P, Wong WC, Liu H (2006) N-(4-[4-(1H-Benzoimidazol-2-yl)-arylamino]-methyl-phenyl)-benzamide derivatives as small molecule heparanase inhibitors. Bioorg Med Chem Lett 16:404–408. doi:10.1016/j.bmcl.2005.09.070
Pan W, Miao HQ, Xu YJ, Navarro EC, Tonra JR, Corcoran E, Lahiji A, Kussie P, Kiselyov AS, Wong WC, Liu H (2006) 1-[4-(1H-Benzoimidazol-2-yl)-phenyl]-3-[4-(1H-benzoimidazol-2-yl)-phenyl]-urea derivatives as small molecule heparanase inhibitors. Bioorg Med Chem Lett 16:409–412. doi:10.1016/j.bmcl.2005.09.069
Kim JS, Sun Q, Gatto B, Yu C, Liu A, Liu LF, LaVoie EJ (1996) Structure-activity relationships of benzimidazoles and related heterocycles as topoisomerase I poisons. Bioorg Med Chem 4:621–630. doi:10.1016/0968-0896(96)00047-8
Tonelli M, Paglietti G, Boido V, Sparatore F, Marongiu F, Marongiu E, La Colla P, Loddo R (2008) Antiviral activity of benzimidazole derivatives. I. Antiviral activity of 1-substituted-2-[(benzotriazol-1/2-yl)methyl]benzimidazoles. Chem Biodivers 5:2386–2401. doi:10.1002/cbdv.200890203
Tonelli M, Simone M, Tasso B, Novelli F, Boido V, Sparatore F, Paglietti G, Pricl S, Giliberti G, Blois S, Ibba C, Sanna G, Loddo R, La Colla P (2010) Antiviral activity of benzimidazole derivatives. II. Antiviral activity of 2-phenylbenzimidazole derivatives. Bioorg Med Chem 18:2937–2953. doi:10.1016/j.bmc.2010.02.037
Shoemaker RH (2006) The NCI 60 human tumour cell line anticancer drug screen. Nat Rev Cancer 6:813–823. doi:10.1038/nrc1951
Boyd MR, Paull KD (1995) Some practical considerations and applications of the National Cancer Institute in vitro anticancer drug discovery screen. Drug Dev Res 34:91–109. doi:10.1002/ddr.430340203
Stinson SF, Alley MC, Kopp WC, Fiebig HH, Mullendore LA, Pittman AF, Kenney S, Keller J, Boyd MR (1992) Morphological and immunocytochemical characteristics of human tumor cell lines for use in a disease-oriented anticancer drug screen. Anticancer Res 12:1035–1053
Akee RK, Ransom T, Ratnayake R, McMahon JB, Beutler JA (2012) Chlorinated englerins with selective inhibition of renal cancer cell growth. J Nat Prod 75:459–463. doi:10.1021/np200905u
Chan KP, Chen DYK (2011) Chemical synthesis and biological evaluation of the englerin analogues. ChemMedChem 6:420–423. doi:10.1002/cmdc.201000544
Ratnayake R, Covell D, Ransom TT, Gustafson KR, Beutler JA (2008) Englerin A, a selective inhibitor of renal cancer cell growth, from Phyllanthus engleri. Org Letters 11:57–60. doi:10.1021/ol802339w
Sulzmaier FJ, Li Z, Nakashige ML, Fash DM, Chain WJ, Ramos JW (2012) Englerin a selectively induces necrosis in human renal cancer cells. PLoS One 7:e48032. doi:10.1371/journal.pone.0048032
Barbieri F, Alama A, Tasso B, Boido V, Bruzzo C, Sparatore F (2003) Quinolizidinyl derivatives of iminodibenzyl and phenothiazine as multidrug resistance modulators in ovarian cancer cells. Invest New Drugs 21:413–420. doi:10.1023/A:1026295017158
Alama A, Tasso B, Novelli F, Sparatore F (2009) Organometallic compounds in oncology: implications of novel organotins as antitumor agents. Drug Discov Today 14:500–508. doi:10.1016/j.drudis.2009.02.002
Sparatore A, Veronese M, Sparatore F (1987) Quinolizidine derivatives with antimicrobial activity. Farmaco Ed Sci 42:159–174
Sparatore A, Basilico N, Parapini S, Romeo S, Novelli F, Sparatore F, Taramelli D (2005) 4-Aminoquinoline quinolizidinyl- and quinolizidinylalkyl-derivatives with antimalarial activity. Bioorg Med Chem 13:5338–5345. doi:10.1016/j.bmc.2005.06.047
Sparatore A, Basilico N, Casagrande M, Parapini S, Taramelli D, Brun R, Wittlin S, Sparatore F (2008) Antimalarial activity of novel pyrrolizidinyl derivatives of 4-aminoquinoline. Bioorg Med Chem Lett 18:3737–3740. doi:10.1016/j.bmcl.2008.05.042
Tonelli M, Vettoretti G, Tasso B, Novelli F, Boido V, Sparatore F, Busonera B, Ouhtit A, Farci P, Blois S, Giliberti G, La Colla P (2011) Acridine derivatives as anti-BVDV agents. Antiviral Res 91:133–141. doi:10.1016/j.antiviral.2011.05.005
Villa V, Tonelli M, Thellung S, Corsaro A, Tasso B, Novelli F, Canu C, Pino A, Chiovitti K, Paludi D, Russo C, Sparatore A, Aceto A, Boido V, Sparatore F, Florio T (2011) Efficacy of novel acridine derivatives in the inhibition of hPrP90-231 prion protein fragment toxicity. Neurotox Res 19:556–574. doi:10.1007/s12640-010-9189-8
Kashfi K (2009) Anti-inflammatory agents as cancer therapeutics. Adv Pharmacol 57:31–89. doi:10.1016/S1054-3589(08)57002-5
Mantovani A, Allavena P, Sica A, Balkwill F (2008) Cancer-related inflammation. Nature 454:436–444. doi:10.1038/nature07205
Wang D, Dubois RN (2006) Prostaglandins and cancer. Gut 55:115–122. doi:10.1136/gut.2004.047100
Shriner RL, Land AH (1941) The structure of the bisulfite compound of acetaldehyde. J Org Chem 6:888–894. doi:10.1021/jo01206a014
Mulliez M, Naudi C (1993) Synthèse d’\(\alpha \)-hydroxysulfinates. Tetrahedron. 2469–2473. doi:10.1016/S0040-4020(01)86325-0
Geiger C, Zelenka C, Lehmkuhl K, Schepmann D, Englberger W, Wünsch B (2010) Conformationally constrained kappa receptor agonists: stereoselective synthesis and pharmacological evaluation of 6,8-diazabicyclo[3.2.2]nonane derivatives. J Med Chem 53:4212–4222. doi:10.1021/jm100182p
She J, Jiang Z, Wang Y (2009) One-pot synthesis of functionalized benzimidazoles and 1H-pyrimidines via cascade reactions of o-aminoanilines or naphthalene-1,8-diamine with alkynes and p-tolylsulfonyl azide. Synlett:2023–2027. doi: 10.1055/s-0029-1217515
Sparatore F, Boido V, Preziosi P, Miele E, De Natale G (1969) Synthesis and pharmacodynamic properties of some lupinane derivatives. Farmaco Ed Sci 24:587–621
Acknowledgments
The Authors gratefully acknowledge the members of the National Cancer Institute Development Therapeutics Program (Bethesda, Maryland, USA) for performing the anti-cancer screening.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tonelli, M., Tasso, B., Mina, L. et al. Primary anti-proliferative activity evaluation of 1-(quinolizidin-1’-yl)methyl- and 1-(\(\omega \)-tert-amino)alkyl-substituted 2-phenyl-, 2-benzyl- and 2-[(benzotriazol-1/2-yl)methyl]benzimidazoles on human cancer cell lines. Mol Divers 17, 409–419 (2013). https://doi.org/10.1007/s11030-013-9440-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-013-9440-3