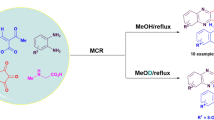
Abstract
In this study, we report the synthesis of several novel dihydroisoxazole-, tetrahydroisoxazole- and dihydropyrazole-fused pyrido[2,3-b]quinolines, α-carbolines, and pyrido[2,3-c]coumarins, respectively, from simple precursors and by exploring intramolecular 1,3-dipolar cycloaddition reactions involving nitrile oxides, nitrones, and nitrile imines as 1,3-dipoles.
Similar content being viewed by others
References
Carruthers W (1990) In: Cycloaddition reactions in organic synthesis, tetrahedron organic chemistry series, vol 8. Pergamon press, Oxford
Tietze LF (1996) Domino reactions in organic synthesis. Chem Rev 96: 115–136
Grootaert WM, Clercq PJD (1982) Intramolecular Diels Alder reaction with furan-diene. An expeditious entry into a functionalised gibbane. Tetrahedron Lett 23: 3291–3294. doi:10.1016/S0040-4039(00)87595-4
Benwell M, Flyan B, Hockless D (1997) Convergent total synthesis of lamellarin K. Chem Commun 23: 2259–2260. doi:10.1039/a705874h
Jung ME, Lam PY, Mansuri MM, Speltz LM (1985) Stereoselective synthesis of an analog of podophyllotoxin by an intramolecular Diels-Alder reaction. J Org Chem 50: 1087–1105. doi:10.1021/jo00207a034
Elder field RC (1960) In: Heterocyclic Compounds, chap 111, vol 4. Wiley, London
Kournetsov VV, Mendez LYV, Gomez CMM (2005) Recent progress in the synthesis of quinolines. Curr Org Chem 9: 141–143
Baruah B, Bhuyan PJ (2009) Synthesis of some complex pyrano[2,3-b]- and pyrido[2,3-b]quinolines from simple acetanilides via intramolecular domino hetero Diels–Alder reactions of 1-oxa-1,3-butadienes in aqueous medium. Tetrahedron 65: 7099–7104. doi:10.1016/j.tet.2009.06.036
Peters, W, Richards, WHG (eds) (1984) Antimalarial drugs II. Springer, Berlin
Ramirez MA, Drimal J, Aviado M (1972) Cardiopulmonary effects of antimalarial drugs: Coronary vascular effects of pyridoquinolines. Toxicol Appl Pharm 21: 482–494. doi:10.1016/0041-008X(72)90005-1
Venkat RG, Qi L, Pierce M, Robbins PB, Sahasrabudhe SR, Selliah R (2007) Int Patent WO2007076085
Arakawa H, Monden Y, Nakatsuru Y, Kodera T (2003) Int Patent WO2003080077
Li X, Vince R (2006) Conformationally restrained carbazolone-containing α,γ -diketo acids as inhibitors of HIV integrase. Bioorg Med Chem 14: 2942–2955. doi:10.1016/j.bmc.2005.12.013
Gelbard HA, Maggirwar SB, Dewhurst S, Schifitto GP (2007) Int Patent WO2007076372
Paolini L Sci Rep 1st Super Sanita. 1:86
Okamoto T, Akase T, Izumi S, Inaba S, Yamamoto H (1972) Japanese patent 7220196. CA, 77, 152142
Winter J, Mola ND (1975) West German Patent 2442513. CA 82, 156255
Stubbs MC, Armstrong SA (2007) FLT3 as a therapeutic target in childhood acute leukemia. Curr Drug Targets 8: 703–714
Shenoy S, Vasania VS, Gopal M, Mehta A (2007) 8-Methyl-4-(3-diethylamino propylamino) pyrimido[4′,5′,4,5]thieno(2,3-b)quinoline (MDPTQ), a quinoline derivative that causes ROS-mediated apoptosis in leukemia cell lines. Toxicol Appl Pharmacol 222: 80–88. doi:10.1016/j.taap.2007.04.005
Massip AM, Pallis M, Carter GI, Grundy M, Shang S, Russel NH (2006) DNA-repair contributes to the drug-resistant phenotype of primary acute myeloid leukemia cells with FLT3 internal tandem duplications and is reversed by the FLT3 inhibitor PKC412. Leukemia 20: 2130–2136. doi:10.1038/sj.leu.2404439
Joseph J, Meijer L, Liger F (2006) Fr Patent FR2876377
Das S, Brown JW, Dong Q, Gong X, Kaldor SW, Liu Y, Paraselli BR, Scorah N, Stafford JA, Wallace MB (2007) Int Patent WO2007044779
Fong TM, Erondu NE, Macneil DJ, Mcintyre JH, Pleog LHTV (2004) Int Patent WO2004110368
Molina P, Alajarín M, Vidal A, Sánchez-Aranda P (1992) C = C-conjugated carbodiimides as 2-aza dienes in intramolecular [4+2] cycloadditions. One pot preparation of quinoline, α-carboline and quinindoline derivatives. J Org Chem 57: 929–934. doi:10.1021/jo00029a026
Vera-Luque P, Alajarin R, Alvarez-Builla J, Vaquero JJ (2006) An improved synthesis of α-carbolines under microwave irradiation. J Org Lett 8: 415–418. doi:10.1021/ol052552y
Majumder S, Bhuyan PJ (2011) The tertiary amino effect: an efficient method for the synthesis of α-carbolines. Synlett 173–176 doi:10.1002/chin 201121147
Majumder S, Bhuyan PJ (2011) An efficient one-pot, three component reaction: synthesis of complex–annelated α-carbolines via an intramolecular [3+2] dipolar cycloaddition reaction. Synlett 1547–1550 doi:10.1055/S-0030-1260787
Singh I, Kaur H, Kumar S, Kumar A, Lata S, Kumar A (2010) Synthesis of new coumarin derivatives as antibacterial agents. Int J Chem Tech Res 2: 1745–1752
Arndt F, Loewe L, On R, Ayca E (1951) Coumarindiol and coumarin-chromone tautomerie. Chem Ber 84: 319–329. doi:10.1002/cber.19510840312
Jung JC, Lee JH, Oh S, Lee JG, Park OS (2004) Synthesis & antitumor activity of 4-hydroxy coumarin derivatives. Bioorg Med Chem Lett 14: 5527–5531. doi:10.1016/j.bmcl.2004.09.009
Unangst PC, Capiris T, Connor DT, Heffner TG, MacKenzie RG, Miller SR, Pugsley TA, Wise LD (1997) Chromeno[3,4-c]pyridin-5-ones: selective human dopamine D4 receptor antagonists as potential antipsychotic agents. J Med Chem 40: 2688–2693. doi:10.1021/jm970170v
O’Kennedy R, Thornes RD (1997) Coumarins: biology, applications and mode of action. Wiley, Chichester
Connor DT, Unangst PC, Schwender CF, Sorenson RJ, Carethers ME, Puchalski C, Brown RE, Finkel MP (1989) 3-(aminoalkyl)-1,2,3,4-tetrahydro-5H[1]benzopyrano [3,4- c]pyridin-5-ones as potential anticholinergic bronchodi-lators. J Med Chem 32: 683–688. doi:10.1021/jm00123a032
Feuer G (1974) In: Ellis GP, West GB (eds) Progress in medicinal chemistry, vol 10. North-Holland, New York, pp 85–158
Upadhyay KK, Mishra KR, Kumar A (2008) A convenient synthesis of some coumarin derivatives using SnCl2·2H2O as catalyst. Catal Lett 121: 118–120. doi:10.1007/s10562-007-9307-2
Henry CE, Kwon O (2007) Phosphine-catalyzed synthesis of highly functionalised coumarins. Org Lett 9: 3069–3072. doi:10.1021/ol071181d
Khoshkholgh MJ, Lotfi M, Balalaie S, Rominger F (2009) Efficient synthesis of pyrano[2,3- c]coumarins via intramolecular domino Knoevenagel hetero-Diels–Alder reactions. Tetrahedron 65: 4228–4234. doi:10.1016/j.tet.2009.03.032
Kuroki Y, Akao R, Inazumi T, Noguchi M (1994) Thermal ene reaction of 4-(2-alkenylamino)-3-formyl-2(2H)-chromenones. Tetrahedron 50: 1063–1072. doi:10.1016/S0040-4020(01)80817-6
Kalita PK, Baruah B, Bhuyan PJ (2006) Synthesis of novel pyrano[2,3-b] quinolines from simple acetanilides via intramolecular 1,3-dipolar cycloaddition. Tetrahedron Lett 47: 7779–7782. doi:10.1016/j.tetlet.2006.08.086
Read MW, Ray PS (1995) Synthesis of a 1,8-naphthyridin-5-one derivative via an intramolecular 1,3-dipolar cycloaddition reaction. J Het Chem 32: 1595–1598. doi:10.1002/jhet.5570320531
Soledade M, Pedras C, Suchy M, Ahiahonu PWK (2006) Unprecedented chemical structure and biomimetic synthesis of erucalexin, a phytoalexin from the wild crucifer erucastrum gallicum. Org Biomol Chem 4: 691–701. doi:10.1039/b515331j
Sabatie A, Vegh D, Loupy A, Floch L (2001) Synthesis of aromatic and heteroaromatic annelated [1,4]diazapines. Arkivoc vi: 122–128. ISSN: 1424-6376
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
The Below is the Electronic Supplementary Material.
Rights and permissions
About this article
Cite this article
Majumder, S., Borah, P. & Bhuyan, P.J. Intramolecular 1,3-dipolar cycloaddition reactions in the synthesis of complex annelated quinolines, α-carbolines and coumarins. Mol Divers 16, 279–289 (2012). https://doi.org/10.1007/s11030-012-9358-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-012-9358-1