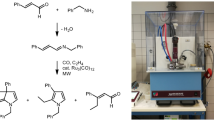
Abstract
The new tertiary furfurylamine with triple bond as a dienophylic part i. e. N-(5-methyl-2-furfuryl)-N-prop-2-ynyl-p-toluidine (1) was prepared and the intramolecular Diels-Alder reaction of the amine (1) was performed under microwave irradiation conditions and by heating a benzene solution of the amine under nitrogen. Comparing the results of the usual thermal and the MAOS reaction, we confirmed our expectations that MAOS could promote the outcome of IMDA reaction of the suitably N-substituted tertiary 2-furfuryl-amines. In the present example, N-p-tolyl-5-methyl-5,7a-dihydro-5,7a-epoxyisoindoline was obtained in much better yield and of higher purity.
Similar content being viewed by others
References
Carruthers, W. Cycloaddition Reactions in Organic Synthesis, Pergamon Press, Oxford, 1990., and references cited therein.
Hayes, B.L., Microwave Synthesis: Chemistry at the Speed of Light, CEM Publishing, Matthews, 2002., and references cited therein; Loupy, A. (Ed), Wiley-VCH, 2002.
de la Hoz, A., Diaz-Ortis, A., Moreno A., and Langa, Fr., Cycloadditions under Microwave Irradiation Conditions: Methods and Applications, Eur. J. Org. Chem. (2000) 1308–1320.
Bilović, D., Stojanac, Ž. and Hahn, V., A Novel Type of Intramolecular Diels-Alder Reaction in the Furan Series, Tetrahedron Lett. (1964) 2071–2074. Bilović, D. and Hahn,V., On the Preparation of Some Tertiary Amines Containing the 2-Furfuryl Group. Isomerization of Allyl-aryl-(2-furfuryl)-amines to N-Aryl-4H-5,7a-Epoxyisoindolines, Croat. Chem. Acta. 39 (1967) 189–197.
Klepo, Ž. and Jakopčić, K., The influence of Substituents on the Rate of the Intramolecular Diels-Alder Reaction of Allyl aryl(2-furfuryl)amines, Croat. Chem. Acta. 47 (1975) 45–50. Klepo, Ž. and Jakopčić, K., Studies in the Furan Series. 23. Preparation of some New 5-Substituted furfurylallylarylamines. Influence of Substituents on the Intramolecular Diels-Alder (IMDA) Reaction, J. Heterocyclic Chem., 24 (1987) 1787–1791.
Klepo, Ž. and Jakopćić, K., Studies in the Furan Series. 23. Preparation of some New 5-Substituted furfurylallylarylamines. Influence of Substituents on the Intramolecular Diels-Alder (IMDA) Reaction, J. Heterocyclic Chem., 24 (1987) 1787–1791. Mance, A.D. and Jakopćić, K., The Intramolecular Diels-Alder Reaction of N-Aryl-cyclohexenyl-(2-furfuryl)-amines. The New Rout to Benzo[c, d]isoindole Derivative, Vestn. Slov. Kem. Društva. 33 (1986) 287–294. Mance, A.D., Jakopćić, K. and Šindler-Kulyk, M., New Substituted Alkenyl-Furfuryl-Aryl Amines: Synthesis and their Characterization, Synthetic Commun. 26 (1996) 923–933. Mance, A.D., Šindler-Kulyk, M., Jakopćić, K., Hergold-Brundi, A. and Nagl, A., New Epoxyisoindolines by Intramolecular Diels-Alder Reaction of some Methyl-substituted Allylaryl-2-furfurylamines, J. Heterocyclic Chem., 34 (1997) 1315–1322. Mance, A.D., Borovićka, B., Karaman, B. and Jakopćić, K., New Synthesis of Substituted Isoindolines from Furans via Epoxyisoindolines, J. Heterocyclic Chem., 36 (1999) 1337–1341. Mance, A.D., Borovićka, B. and Jakopćić, K., New Compounds in Ring-opening Reaction of 5-Substituted Epoxyisoindolines, J. Heterocyclic Chem. 39 (2002) 277–285.
Kappe, C.O., Murphee, S.S. and Padwa, A., Synthetic Aplication of Furan Diels-Alder Chemistry, Tetrahedron, 53 (1997) 14179–14233, and references cited therein.
Sauer, J. and Sustmann, R., Mechanistic Aspects of Diels-Alder Reaction: A Critical Survey, Angew. Chem., Int. Ed. Engl., 19 (1980) 779–807.
Spreitzer, H. and Mustafa, S., Isoindole in der pharmazeutischen Forschung, Pharmazie in Unserer Zeit (1991) 83–87.
Padwa, A., Brodney, M.A., Liu, B.,Satake, K., and Wu, T., A Cycloaddition Approach toward the Synthesys of Substituted Indolines and Tetrahydroisoquinolines, J. Org. Chem. (1999) 3595–3607 and references cited therein.
Lee, M., Moritomo, H. and Kanematsu, K., A New route to Isoindole Nucleus via Furan-Pyrrol Ring-Exchange, J. Chem. Soc. Chem. Commun. (1994) 1535.
Katritzky, A.R., Paluchowska, M.H. and Gallos, J.K., Intramolecular Diels-Alder Cyclization of N-Furfurylpropargylamines: A Novel General Synthesis of Isoindoles, J. Heterocyclic Chem., 26 (1989) 421–426.
Hunt, I.R, Rogers, C., Woo, S., Rauk, A. and Keay, B.A., Why Do Catalytic Quantities of Lewis Acid Generally Yield More Product Then 1.1 equi in the Intramolecular Diels-Alder Reaction with a Furan Diene? Competitive Complexation NMR Studies Provide an Ansver, J. Am. Chem. Soc., 117 (1995) 1049–1056.
Tagmazyan, K. Ts., Mkrtchyan, R.S. and Babayan, A.T., Studies of the Amino Compounds, Zh. Org. Khim., 10 (1974) 1642–1648.
Mance, A.D., Jakopćić, K. and Šindler-Kulyk, M., New Substituted Alkenyl-furfuryl-Aryl Amines: Synthesis and their Characterization, Synthetic Commun., 26 (1996) 923–933.
Hergold-Brundić, A., Pavlović, G., Mance, A.D. and Jakopćić, K., N-(4-Methylphenyl)-N-(5-nitro-2-furfuryl)-N-prop-2-ynylamine, Acta Cryst. (2000). C56, e520–e521.
Babayan, A.T., Tagmazyan, K. Ts., Ioffe, A.I., Mkrtchyan, R.S. and Torosyan, G.O., Doklady Akad. Nauk Arm, VIII (1974) 275–279.
Author information
Authors and Affiliations
Corresponding author
Additional information
The acronym for: Intramolecular Diels-Alder Reaction of Furan.
The acronym for: Microwave-Assisted Organic Synthesis.
Rights and permissions
About this article
Cite this article
Mance, A.D., Jakopčić, K. Microwave assisted IMDAF# reaction: Microwave irradiation applied with success to cycloaddition reaction of N-propargyl-N-p-tolyl-N-2-furfurylamines. Mol Divers 9, 229–231 (2005). https://doi.org/10.1007/s11030-005-3434-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11030-005-3434-8