Abstract
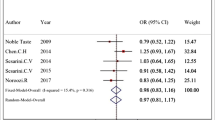
Autism spectrum disorder (ASD) is a neurodevelopmental disorder with various epidemiologic, genetic, epigenetic, and environmental factors being associated with it. The observed sex bias in ASD towards male has prompted investigators to propose sex-dependent mechanisms for ASD. Retinoic acid-related orphan receptor-alpha (RORA) is a new autism candidate gene that has been shown to be differentially regulated by male and female hormones. Previous studies have shown deregulation of its expression in the prefrontal cortex and the cerebellum of ASD patients. In the present study we aimed at identification of the possible associations between two functional polymorphisms in the RORA gene (rs11639084 and rs4774388) and the risk of ASD in 518 Iranian ASD patients and 472 age, gender, and ethnic-matched healthy controls by means of tetra primer-amplification refractory mutation system-PCR. The allele and genotype frequencies of rs11639084 were not significantly different between patients and controls. However, the allele frequencies of rs4774388 showed significant overrepresentation of T allele in patients compared with controls (P = 0.04, OR (95% CI) =1.21 (1.01–1.46)). The rs4774388-TT genotype was significantly higher in patients compared with controls and was associated with ASD risk in dominant inheritance model (P = 0.04, OR (95% CI) =0.77 (0.59–0.99)). Haplotype analysis showed significant association of two estimated blocks of rs11639084/ rs4774388 with ASD risk. Consequently, the present data provide further evidence for RORA participation in the pathogenesis of ASD.


Similar content being viewed by others
References
APA - American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders: DSM-5. American Psychiatric Association Publishing, Arlington
Auyeung B, Baron-Cohen S, Chapman E, Knickmeyer R, Taylor K, Hackett G (2006) Foetal testosterone and the child systemizing quotient. Eur J Endocrinol 155:S123–S130
Baron-Cohen S, Lombardo MV, Auyeung B, Ashwin E, Chakrabarti B, Knickmeyer R (2011) Why are autism spectrum conditions more prevalent in males? PLoS Biol 9:e1001081
Behnia F et al. (2015) Fetal DNA methylation of autism spectrum disorders candidate genes: association with spontaneous preterm birth. American journal of obstetrics and gynecology 212:533. e531-533. e539
Ben-David E, Shifman S (2012) Networks of neuronal genes affected by common and rare variants in autism spectrum disorders. PLoS Genet 8:e1002556. doi:10.1371/journal.pgen.1002556
Chakrabarti B et al (2009) Genes related to sex steroids, neural growth, and social–emotional behavior are associated with autistic traits, empathy, and Asperger syndrome. Autism Res 2:157–177
Clark AG (2004) The role of haplotypes in candidate gene studies. Genet Epidemiol 27:321–333
Correia CT et al (2010) Increased BDNF levels and NTRK2 gene association suggest a disruption of BDNF/TrkB signaling in autism. Genes Brain Behav 9:841–848. doi:10.1111/j.1601-183X.2010.00627.x
Eftekharian MM et al (2016) RAR-related orphan receptor a (RORA): a new susceptibility gene for multiple sclerosis. J Neurol Sci 369:259–262
Fombonne C (2005) The epidemiology of pervasive developmental disorders recent developments in autism research. MF Casanova, ed (New York, Nova science publishers Inc): 1-25
Glickman G (2010) Circadian rhythms and sleep in children with autism. Neurosci Biobehav Rev 34:755–768. doi:10.1016/j.neubiorev.2009.11.017
Hu VW (2012) Is retinoic acid-related orphan receptor-alpha (RORA) a target for gene–environment interactions contributing to autism? Neurotoxicology 33:1434–1435
Hu VW et al (2009) Gene expression profiling differentiates autism case–controls and phenotypic variants of autism spectrum disorders: evidence for circadian rhythm dysfunction in severe autism. Autism Res 2:78–97
Knickmeyer RC, Baron-Cohen S (2006) Topical review: fetal testosterone and sex differences in typical social development and in autism. J Child Neurol 21:825–845
Lehman NL (2009) The ubiquitin proteasome system in neuropathology. Acta Neuropathol 118:329–347. doi:10.1007/s00401-009-0560-x
Lettice L, Hecksher-Sorensen J, Hill R (2001) The role of Bapx1 (Nkx3.2) in the development and evolution of the axial skeleton. J Anat 199:181–187. doi:10.1046/j.1469-7580.2001.19910181.x
McMahon AP (2000) Neural patterning: the role of Nkx genes in the ventral spinal cord. Genes Dev 14:2261–2264. doi:10.1101/Gad.840800
Miles JH (2011) Autism spectrum disorders—a genetics review. Genetics in Medicine 13:278–294
Mitra I et al (2016) Pleiotropic mechanisms indicated for sex differences in autism. PLoS Genet 12:e1006425. doi:10.1371/journal.pgen.1006425
Nguyen A, Rauch TA, Pfeifer GP, Hu VW (2010) Global methylation profiling of lymphoblastoid cell lines reveals epigenetic contributions to autism spectrum disorders and a novel autism candidate gene, RORA, whose protein product is reduced in autistic brain. FASEB J 24:3036–3051
Noroozi R et al (2016) Glutamate receptor, metabotropic 7 (GRM7) gene variations and susceptibility to autism: a case–control study. Autism Res 9:1161
Pattaro C, Ruczinski I, Fallin DM, Parmigiani G (2008) Haplotype block partitioning as a tool for dimensionality reduction in SNP association studies. BMC Genomics 9:405
Sarachana T, Hu VW (2013) Genome-wide identification of transcriptional targets of RORA reveals direct regulation of multiple genes associated with autism spectrum disorder. Molecular autism 4:1
Sarachana T, Xu M, Wu R-C, Hu VW (2011) Sex hormones in autism: androgens and estrogens differentially and reciprocally regulate RORA, a novel candidate gene for autism. PLoS One 6:e17116
Sato TK et al (2004) A functional genomics strategy reveals Rora as a component of the mammalian circadian clock. Neuron 43:527–537. doi:10.1016/j.neuron.2004.07.018
Schaefer GB (2016) Clinical genetic aspects of ASD Spectrum disorders. Int J Mol Sci 17:180. doi:10.3390/ijms17020180
Scott CE et al (2010) SOX9 induces and maintains neural stem cells. Nat Neurosci 13:1181–1189. doi:10.1038/nn.2646
Solé X, Guinó E, Valls J, Iniesta R, Moreno V (2006) SNPStats: a web tool for the analysis of association studies. Bioinformatics 22:1928–1929
Steffen J, Seeger M, Koch A, Kruger E (2010) Proteasomal degradation is transcriptionally controlled by TCF11 via an ERAD-dependent feedback loop. Mol Cell 40:147–158. doi:10.1016/j.molcel.2010.09.012
Stolt CC, Lommes P, Sock E, Chaboissier MC, Schedl A, Wegner M (2003) The Sox9 transcription factor determines glial fate choice in the developing spinal cord. Genes Dev 17:1677–1689. doi:10.1101/gad.259003
Theodorou E, Dalembert G, Heffelfinger C, White E, Weissman S, Corcoran L, Snyder M (2009) A high throughput embryonic stem cell screen identifies Oct-2 as a bifunctional regulator of neuronal differentiation. Genes Dev 23:575–588. doi:10.1101/gad.1772509
Tucker AS, Watson RP, Lettice LA, Yamada G, Hill RE (2004) Bapx1 regulates patterning in the middle ear: altered regulatory role in the transition from the proximal jaw during vertebrate evolution. Development 131:1235–1245. doi:10.1242/dev.01017
Ward LD, Kellis M (2016) HaploReg v4: systematic mining of putative causal variants, cell types, regulators and target genes for human complex traits and disease. Nucleic Acids Res 44:D877–D881. doi:10.1093/nar/gkv1340
Ylisaukko-oja T et al (2005) Analysis of four neuroligin genes as candidates for autism. European journal of human genetics : EJHG 13:1285–1292. doi:10.1038/sj.ejhg.5201474
Zeng L, Kempf H, Murtaugh LC, Sato ME, Lassar AB (2002) Shh establishes an Nkx3.2/Sox9 autoregulatory loop that is maintained by BMP signals to induce somitic chondrogenesis. Genes Dev 16:1990–2005. doi:10.1101/gad.1008002
Zhang J, Gonit M, Salazar M, Shatnawi A, Shemshedini L, Trumbly R, Ratnam M (2010) C/EBPα redirects androgen receptor signaling through a unique bimodal interaction. Oncogene 29:723–738
Zhao K et al (2007) An example of association mapping in structured samples. PLoS Genet 3:E4
Acknowledgments
This study was supported financially by Shahid Beheshti University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare have no conflict of interest.
Research involving human participants and/or animals
The study has been approved by the appropriate institutional and/or national research ethics committee and has been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Sayad, A., Noroozi, R., Omrani, M.D. et al. Retinoic acid-related orphan receptor alpha (RORA) variants are associated with autism spectrum disorder. Metab Brain Dis 32, 1595–1601 (2017). https://doi.org/10.1007/s11011-017-0049-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-017-0049-6