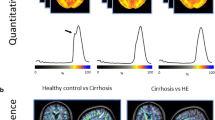
Abstract
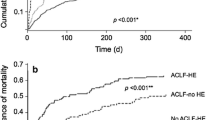
Although hepatic encephalopathy (HE) on the background of acute on chronic liver failure (ACLF) is associated with high mortality rates, it is unknown whether this is due to increased blood-brain barrier permeability. Specific gravity of cerebrospinal fluid measured by CT is able to estimate blood-cerebrospinal fluid-barrier permeability. This study aimed to assess cerebrospinal fluid specific gravity in acutely decompensated cirrhosis and to compare it in patients with or without ACLF and with or without hepatic encephalopathy. We identified all the patients admitted for acute decompensation of cirrhosis who underwent a brain CT-scan. Those patients could present acute decompensation with or without ACLF. The presence of hepatic encephalopathy was noted. They were compared to a group of stable cirrhotic patients and healthy controls. Quantitative brain CT analysis used the Brainview software that gives the weight, the volume and the specific gravity of each determined brain regions. Results are given as median and interquartile ranges and as relative variation compared to the control/baseline group. 36 patients presented an acute decompensation of cirrhosis. Among them, 25 presented with ACLF and 11 without ACLF; 20 presented with hepatic encephalopathy grade ≥ 2. They were compared to 31 stable cirrhosis patients and 61 healthy controls. Cirrhotic patients had increased cerebrospinal fluid specific gravity (CSF-SG) compared to healthy controls (+0.4 %, p < 0.0001). Cirrhotic patients with ACLF have decreased CSF-SG as compared to cirrhotic patients without ACLF (−0.2 %, p = 0.0030) that remained higher than in healthy controls. The presence of hepatic encephalopathy did not modify CSF-SG (−0.09 %, p = 0.1757). Specific gravity did not differ between different brain regions according to the presence or absence of either ACLF or HE. In patients with acute decompensation of cirrhosis, and those with ACLF, CSF specific gravity is modified compared to both stable cirrhotic patients and healthy controls. This pattern is observed even in the absence of hepatic encephalopathy suggesting that blood-CSF barrier impairment is manifest even in absence of overt hepatic encephalopathy.

Similar content being viewed by others
References
Aoki I, Naruse S, Tanaka C (2004) Manganese-enhanced magnetic resonance imaging (MEMRI) of brain activity and applications to early detection of brain ischemia. NMR Biomed 17:569–580
Bajaj JS, Schubert CM, Heuman DM et al (2010) Persistence of cognitive impairment after resolution of overt hepatic encephalopathy. Gastroenterology 138:2332–2340
Cascino A, Cangiano C, Fiaccadori F et al (1982) Plasma and cerebrospinal fluid amino acid patterns in hepatic encephalopathy. Dig Dis Sci 27:828–832
Chavarria L, Alonso J, García-Martínez R et al (2013) Brain magnetic resonance spectroscopy in episodic hepatic encephalopathy. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 33:272–277
Cordoba J, Ventura-Cots M, Simón-Talero M et al (2014) Characteristics, risk factors, and mortality of cirrhotic patients hospitalized for hepatic encephalopathy with and without acute-on-chronic liver failure (ACLF. J Hepatol 60:275–281
Degos V, Lescot T, Zouaoui A et al (2006) Computed tomography-estimated specific gravity of noncontused brain areas as a marker of severity in human traumatic brain injury. Anesth Analg 103:1229–1236
Degos V, Lescot T, Icke C et al (2012) Computed tomography-estimated specific gravity at hospital admission predicts 6-month outcome in mild-to-moderate traumatic brain injury patients admitted to the intensive care unit. Anesth Analg 114:1026–1033
Ghersi-Egea J-F, Strazielle N (2002) Choroid plexus transporters for drugs and other xenobiotics. J Drug Target 10:353–357
Ghersi-Egea JF, Leininger-Muller B, Cecchelli R, Fenstermacher JD (1995) Blood-brain interfaces: relevance to cerebral drug metabolism. Toxicol Lett 82–83:645–653
Gove CD, Hughes RD, Ede RJ, Williams R (1997) Regional cerebral edema and chloride space in galactosamine-induced liver failure in rats. Hepatol Baltim Md 25:295–301
Jalan R, Bernuau J (2007) Induction of cerebral hyperemia by ammonia plus endotoxin: does hyperammonemia unlock the blood-brain barrier? J Hepatol 47:168–171
Jalan R, Saliba F, Pavesi M et al (2014) Development and validation of a prognostic score to predict mortality in patients with acute-on-chronic liver failure. J Hepatol 61:1038–1047
James JH, Escourrou J, Fischer JE (1978) Blood-brain neutral amino acid transport activity is increased after portacaval anastomosis. Science 200:1395–1397
Jellinger K, Riederer P, Rausch WD, Kothbauer P (1978) Brain monoamines in hepatic encephalopathy and other types of metabolic coma. J Neural Transm Suppl Epub 1978(14):103–120
Joshi D, O’Grady J, Patel A, et al (2014) Cerebral oedema is rare in acute-on-chronic liver failure patients presenting with high-grade hepatic encephalopathy. Liver Int 34(3):362–366
Lescot T, Bonnet M-P, Zouaoui A et al (2005) A quantitative computed tomography assessment of brain weight, volume, and specific gravity in severe head trauma. Intensive Care Med 31:1042–1050
Lescot T, Degos V, Zouaoui A, Préteux F, Coriat P, Puybasset L (2006) Opposed effects of hypertonic saline on contusions and noncontused brain tissue in patients with severe traumatic brain injury. Crit Care Med 34:3029–3033
Lescot T, Degos V, Puybasset L (2008) Does the brain become heavier or lighter after trauma? Eur J Anaesthesiol Suppl 42:110–114
Mattson LR, Lindor NM, Goldman DH, Goodwin JT, Groover RV, Vockley J (1995) Central pontine myelinolysis as a complication of partial ornithine carbamoyl transferase deficiency. Am J Med Genet 60:210–213
Moreau R, Jalan R, Gines P et al (2013) Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology 144:1426–1437 1437.e1–e9
Nitsch C, Fujiwara K, Kuroiwa T, Klatzo I (1984) Specific gravity increases and brain water content decreases during short epileptiform seizures in discrete rabbit brain areas. J Neurol Sci 64:119–129
Ong JP, Aggarwal A, Krieger D et al (2003) Correlation between ammonia levels and the severity of hepatic encephalopathy. Am J Med 114:188–193
Ono J, Hutson DG, Dombro RS, Levi JU, Livingstone A, Zeppa R (1978) Tryptophan and hepatic coma. Gastroenterology 74:196–200
Orr JG, Homer T, Ternent L et al (2014) Health related quality of life in people with advanced chronic liver disease. J Hepatol 61:1158–1165
Puybasset L, Cluzel P, Chao N, Slutsky AS, Coriat P, Rouby JJ (1998) A computed tomography scan assessment of regional lung volume in acute lung injury. The CT scan ARDS study group. Am J Respir Crit Care Med 158:1644–1655
Romero-Gómez M (2005) Role of phosphate-activated glutaminase in the pathogenesis of hepatic encephalopathy. Metab Brain Dis 20:319–325
Ropper A, Samuels M, Klein J (2014) Adams and Victor’s principles of neurology 10th edition, 10 edn. McGraw-Hill Professional, New York
Rose C, Butterworth RF, Zayed J et al (1999) Manganese deposition in basal ganglia structures results from both portal-systemic shunting and liver dysfunction. Gastroenterology 117:640–644
Rovira A, Grivé E, Pedraza S, Rovira A, Alonso J (2001) Magnetization transfer ratio values and proton MR spectroscopy of normal-appearing cerebral white matter in patients with liver cirrhosis. AJNR Am J Neuroradiol 22:1137–1142
Shawcross DL, Davies NA, Williams R, Jalan R (2004) Systemic inflammatory response exacerbates the neuropsychological effects of induced hyperammonemia in cirrhosis. J Hepatol 40:247–254
Stepanova M, Mishra A, Venkatesan C, Younossi ZM (2012) In-hospital mortality and economic burden associated with hepatic encephalopathy in the United States from 2005 to 2009. Clin Gastroenterol Hepatol Off Clin Pract J Am Gastroenterol Assoc 10:1034–1041.e1
Tominaga S, Watanabe A, Tsuji T (1991) Synergistic effect of bile acid, endotoxin, and ammonia on brain edema. Metab Brain Dis 6:93–105
Vieira SR, Puybasset L, Richecoeur J et al (1998) A lung computed tomographic assessment of positive end-expiratory pressure-induced lung overdistension. Am J Respir Crit Care Med 158:1571–1577
Weiss N, Miller F, Cazaubon S, Couraud P-O (2009) The blood-brain barrier in brain homeostasis and neurological diseases. Biochim Biophys Acta 1788:842–857
Weiss N, Junot C, Rudler M, Thabut D (2016a) Hepatic versus drug-induced encephalopathy in cirrhotic patients? Liver Int 36(8):1233–1234
Weiss N, de Saint Hilaire, PB, Colsch B, et al (2016b) Cerebrospinal fluid metabolomics highlights dysregulation of energy metabolism in overt hepatic encephalopathy. J Hepatol. doi:10.1016/j.jhep.2016.07.046
Wright G, Davies NA, Shawcross DL et al (2007) Endotoxemia produces coma and brain swelling in bile duct ligated rats. Hepatol Baltim Md 45:1517–1526
Young SN, Lal S, Sourkes TL, Feldmuller F, Aronoff A, Martin JB (1975) Relationships between tryptophan in serum and CSF, and 5-hydroxyindoleacetic acid in CSF of man: effect of cirrhosis of liver and probenecid administration. J Neurol Neurosurg Psychiatry 38:322–330
Acknowledgment
The authors gratefully acknowledge the help of Dr. Dominic Yu, Consultant Radiologist at Royal Free Hospital.
Financial support
none.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Nicolas Weiss declares the following potential conflicts of interest.
Research grant
Eumedica, Gilead.
Speaker fees
Alpha-Wasserman, Norgine, Gore.
Dominique Thabut declares the following potential conflicts of interest. Research grant: Eumedica, Gilead. Speaker fees: Alpha-Wasserman, Norgine, Gore, Gilead, BMS, Janssen.
Rajiv Jalan declares the following potential conflicts of interest. Inventor: Ornithinephenylacetate, a treatment for hepatic encephalopathy; UCL Liver dialysis device. Research grant: Ocera therapeutics, Gambro, Sequana and Grifols. Consultant: Ocera therapeutics, Conatus. Founder: Yaqrit ltd. (UCL spin out). Speaker fees: Grifols, Norgine.
The other authors have nothing to declare.
Additional information
Nicolas Weiss and Matteo Rosselli contributed equally to the work
Electronic supplementary material

Supplemental figure
Specific gravity of different brain regions according to the presence or not of cirrhosis and the presence or not of acute decompensation of cirrhosis. (JPEG 43 kb)
Rights and permissions
About this article
Cite this article
Weiss, N., Rosselli, M., Mouri, S. et al. Modification in CSF specific gravity in acutely decompensated cirrhosis and acute on chronic liver failure independent of encephalopathy, evidences for an early blood-CSF barrier dysfunction in cirrhosis. Metab Brain Dis 32, 369–376 (2017). https://doi.org/10.1007/s11011-016-9916-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9916-9