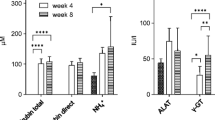
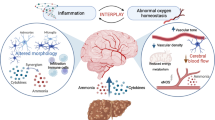
Abstract
Hepatic encephalopathy (HE) is a neuropsychiatric syndrome which frequently accompanies acute or chronic liver disease. It is characterized by a variety of symptoms of different severity such as cognitive deficits and impaired motor functions. Currently, HE is seen as a consequence of a low grade cerebral oedema associated with the formation of cerebral oxidative stress and deranged cerebral oscillatory networks. However, the pathogenesis of HE is still incompletely understood as liver dysfunction triggers exceptionally complex metabolic derangements in the body which need to be investigated by appropriate technologies. This review summarizes technological approaches presented at the ISHEN conference 2014 in London which may help to gain new insights into the pathogenesis of HE. Dynamic in vivo 13C nuclear magnetic resonance spectroscopy was performed to analyse effects of chronic liver failure in rats on brain energy metabolism. By using a genomics approach, microRNA expression changes were identified in plasma of animals with acute liver failure which may be involved in interorgan interactions and which may serve as organ-specific biomarkers for tissue damage during acute liver failure. Genomics were also applied to analyse glutaminase gene polymorphisms in patients with liver cirrhosis indicating that haplotype-dependent glutaminase activity is an important pathogenic factor in HE. Metabonomics represents a promising approach to better understand HE, by capturing the systems level metabolic changes associated with disease in individuals, and enabling monitoring of metabolic phenotypes in real time, over a time course and in response to treatment, to better inform clinical decision making. Targeted fluxomics allow the determination of metabolic reaction rates thereby discriminating metabolite level changes in HE in terms of production, consumption and clearance.



Similar content being viewed by others
Abbreviations
- ALF:
-
Acute liver failure
- ALI:
-
Acute liver injury
- APAP:
-
Acetaminophen
- BDL:
-
Bile duct-ligation
- CFF:
-
Critical flicker frequency
- CPP:
-
Cerebral perfusion pressure
- DAMP:
-
Damage-associated molecular pattern molecules
- GLS:
-
Glutaminase
- HCC:
-
Hepatocellular carcinoma
- HE:
-
Hepatic encephalopathy
- MHE:
-
Minimal hepatic encephalopathy
- miRNA:
-
Micro RNA
- MRS:
-
Magnetic resonance spectroscopy
- NMR:
-
Nuclear magnetic resonance
- PHES:
-
Psychometric hepatic encephalopathy score
- TCA:
-
Tricarboxylic acid cycle
References
Abbassi-Ghadi N et al. (2014) Discrimination of lymph node metastases using desorption electrospray ionisation-mass spectrometry imaging. Chem Commun 50:3661–3664. doi:10.1039/c3cc48927b
Baker LA et al. (2015) Circulating microRNAs reveal time course of organ injury in a porcine model of acetaminophen-induced acute liver failure. PLoS One 10:e0128076. doi:10.1371/journal.pone.0128076
Balog J et al. (2013) Intraoperative tissue identification using rapid evaporative ionization mass spectrometry. Sci Transl Med 5:194ra193. doi:10.1126/scitranslmed.3005623
Bartel DP (2009) MicroRNAs: target recognition and regulatory functions. Cell 136:215–233. doi:10.1016/j.cell.2009.01.002
Bosoi CR, Zwingmann C, Marin H, Parent-Robitaille C, Huynh J, Tremblay M, Rose CF (2014) Increased brain lactate is central to the development of brain edema in rats with chronic liver disease. J Hepatol 60:554–560. doi:10.1016/j.jhep.2013.10.011
Bosoi CR, Oliveira MM, Clément M-A, Tremblay M, Ten Have GA, Deutz NE et al. (2015) Poster session 3: experimental studies in cirrhosis and portal HTN; health economics and cost effectiveness of viral hepatitis. Hepatology 62:950A–961A. doi:10.1002/hep.28232
Butterworth RF (2014) Pathophysiology of brain dysfunction in hyperammonemic syndromes: the many faces of glutamine. Mol Genet Metab 113:113–117. doi:10.1016/j.ymgme.2014.06.003
Dabos KJ, Parkinson JA, Sadler IH, Plevris JN, Hayes PC (2015) (1)H nuclear magnetic resonance spectroscopy-based metabonomic study in patients with cirrhosis and hepatic encephalopathy. World J Hepatol 7:1701–1707. doi:10.4254/wjh.v7.i12.1701
de Graaf RA, Mason GF, Patel AB, Behar KL, Rothman DL (2003) In vivo 1H-[13C]-NMR spectroscopy of cerebral metabolism. NMR Biomed 16:339–357
Engelen MP, Com G, Anderson PJ, Deutz NE (2014) New stable isotope method to measure protein digestibility and response to pancreatic enzyme intake in cystic fibrosis. Clin Nutr 33:1024–1032. doi:10.1016/j.clnu.2013.11.004
Fonville JM et al. (2013) Hyperspectral visualization of mass spectrometry imaging data. Anal Chem 85:1415–1423. doi:10.1021/ac302330a
Gruetter R (2002) In vivo 13C NMR studies of compartmentalized cerebral carbohydrate metabolism. Neurochem Int 41:143–154
Gruetter R, Tkac I (2000) Field mapping without reference scan using asymmetric echo-planar techniques. Magn Reson Med 43:319–323
Gruetter R, Seaquist ER, Ugurbil K (2001) A mathematical model of compartmentalized neurotransmitter metabolism in the human brain. Am J Physiol 281:E100–E112
Henry PG, Tkac I, Gruetter R (2003) 1H-localized broadband 13C NMR spectroscopy of the rat brain in vivo at 9.4 T. Magn Reson Med 50:684–692
Henry PG et al. (2006) In vivo 13C NMR spectroscopy and metabolic modeling in the brain: a practical perspective. Magn Reson Imaging 24:527–539
Jalan R, Olde Damink SW, Deutz NE, Hayes PC, Lee A (2004) Moderate hypothermia in patients with acute liver failure and uncontrolled intracranial hypertension. Gastroenterology 127:1338–1346
Jonker R, Deutz NE, Erbland ML, Anderson PJ, Engelen MP (2014) Hydrolyzed casein and whey protein meals comparably stimulate net whole-body protein synthesis in COPD patients with nutritional depletion without an additional effect of leucine co-ingestion. Clin Nutr 33:211–220. doi:10.1016/j.clnu.2013.06.014
Kinross JM, Holmes E, Darzi AW, Nicholson JK (2011) Metabolic phenotyping for monitoring surgical patients. Lancet 377:1817–1819. doi:10.1016/S0140-6736(11)60171-2
Kircheis G, Wettstein M, Timmermann L, Schnitzler A, Haussinger D (2002) Critical flicker frequency for quantification of low-grade hepatic encephalopathy. Hepatology 35:357–366. doi:10.1053/jhep.2002.30957
Kircheis G, Hilger N, Haussinger D (2014) Value of critical flicker frequency and psychometric hepatic encephalopathy score in diagnosis of low-grade hepatic encephalopathy. Gastroenterology 146:961–969. doi:10.1053/j.gastro.2013.12.026
Ladep NG et al. (2014) Discovery and validation of urinary metabotypes for the diagnosis of hepatocellular carcinoma in west Africans. Hepatology 60:1291–1301. doi:10.1002/hep.27264
Lanz B, Duarte JM, Kunz N, Mlynarik V, Gruetter R, Cudalbu C (2013a) Which prior knowledge? Quantification of in vivo brain 13C MR spectra following 13C glucose infusion using AMARES. Magn Reson Med 69:1512–1522. doi:10.1002/mrm.24406
Lanz B, Gruetter R, Duarte JM (2013b) Metabolic flux and compartmentation analysis in the brain. Front Endocrinol 4:156
Lee KC et al. (2013) A reproducible, clinically relevant, intensively managed, pig model of acute liver failure for testing of therapies aimed to prolong survival. Liver Int Off J Int Assoc Study Liver 33:544–551. doi:10.1111/liv.12042
Luiking YC, Poeze M, Ramsay G, Deutz NE (2009) Reduced citrulline production in sepsis is related to diminished de novo arginine and nitric oxide production. Am J Clin Nutr 89:142–152. doi:10.3945/ajcn.2007.25765
Luiking YC, Poeze M, Deutz NE (2015) Arginine infusion in patients with septic shock increases nitric oxide production without haemodynamic instability. Clin Sci 128:57–67. doi:10.1042/CS20140343
Mason GF, Rothman DL (2004) Basic principles of metabolic modeling of NMR 13C isotopic turnover to determine rates of brain metabolism in vivo. Metab Eng 6:75–84
Mirnezami R et al. (2014) Chemical mapping of the colorectal cancer microenvironment via MALDI imaging mass spectrometry (MALDI-MSI) reveals novel cancer-associated field effects. Molecular Oncology 8:39–49. doi:10.1016/j.molonc.2013.08.010
Mitchell PS et al. (2008) Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci U S A 105:10513–10518. doi:10.1073/pnas.0804549105
Nicholson JK, Lindon JC, Holmes E (1999) ‘Metabonomics’: understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica; The Fate of Foreign Compounds in Biological Systems 29:1181–1189. doi:10.1080/004982599238047
O’Grady JG, Schalm SW, Williams R (1993) Acute liver failure: redefining the syndromes. Lancet 342:273–275
Olde Damink SW, Dejong CH, Deutz NE, Soeters PB (1997) Decreased plasma and tissue isoleucine levels after simulated gastrointestinal bleeding by blood gavages in chronic portacaval shunted rats. Gut 40:418–424
Olde Damink SW, Jalan R, Redhead DN, Hayes PC, Deutz NE, Soeters PB (2002) Interorgan ammonia and amino acid metabolism in metabolically stable patients with cirrhosis and a TIPSS. Hepatology 36:1163–1171. doi:10.1053/jhep.2002.36497
Olde Damink SW et al. (2003) The kidney plays a major role in the hyperammonemia seen after simulated or actual GI bleeding in patients with cirrhosis. Hepatology 37:1277–1285. doi:10.1053/jhep.2003.50221
Romero-Gomez M, Ampuero J (2014) Deciphering the spectrum of low-grade hepatic encephalopathy in clinical practice. Gastroenterology 146:887–890. doi:10.1053/j.gastro.2014.02.019
Romero-Gomez M, Ramos-Guerrero R, Grande L, de Teran LC, Corpas R, Camacho I, Bautista JD (2004) Intestinal glutaminase activity is increased in liver cirrhosis and correlates with minimal hepatic encephalopathy. J Hepatol 41:49–54. doi:10.1016/j.jhep.2004.03.021
Romero-Gomez M et al. (2007) Value of the critical flicker frequency in patients with minimal hepatic encephalopathy. Hepatology 45:879–885. doi:10.1002/hep.21586
Romero-Gomez M et al. (2010) Variations in the promoter region of the glutaminase gene and the development of hepatic encephalopathy in patients with cirrhosis: a cohort study. Ann Intern Med 153:281–288. doi:10.7326/0003-4819-153-5-201009070-00002
Sands CJ et al. (2015) Metabolic phenotyping for enhanced mechanistic stratification of chronic hepatitis C-induced liver fibrosis. Am J Gastroenterol 110:159–169. doi:10.1038/ajg.2014.370
Sharma V et al. (2012) Nitric oxide and L-arginine metabolism in a devascularized porcine model of acute liver failure. Am J Physiol Gastrointest Liver Physiol 303:G435–G441. doi:10.1152/ajpgi.00268.2011
Swann J et al. (2009) Gut microbiome modulates the toxicity of hydrazine: a metabonomic study. Mol BioSyst 5:351–355. doi:10.1039/b811468d
Szabo G, Bala S (2013) MicroRNAs in liver disease. Nat Rev Gastroenterol Hepatol 10:542–552. doi:10.1038/nrgastro.2013.87
Tsien C et al. (2015) Metabolic and molecular responses to leucine-enriched branched chain amino acid supplementation in the skeletal muscle of alcoholic cirrhosis. Hepatology 61:2018–2029. doi:10.1002/hep.27717
Tuvdendorj D, Chinkes DL, Bahadorani J, Zhang XJ, Sheffield-Moore M, Killewich LA, Wolfe RR (2014) Comparison of bolus injection and constant infusion methods for measuring muscle protein fractional synthesis rate in humans. Metab Clin Exp 63:1562–1567. doi:10.1016/j.metabol.2014.09.009
Wilkinson DJ, Atherton PJ, Phillips BE, Greenhaff PL, Smith K (2015) Application of deuterium oxide (D2O) to metabolic research: just D2O it? Depends just how you D2O it! Am J Physiol 308:E847. doi:10.1152/ajpendo.00581.2014
Acknowledgments
The authors warmly thank Dr. Boris Görg for his role as writing coordinator for this review article. Bernard Lanz gratefully acknowledges Dr. Cristina Cudalbu, Dr. Valérie McLin, Dr. Olivier Braissant and Dr. Rolf Gruetter for their contributions in the 13C MRS project. The 13C MRS study was supported by Centre d’Imagerie BioMédicale (CIBM) of the UNIL, UNIGE, HUG, CHUV, EPFL, the Leenaards and Jeantet Foundations and SNF grant 131087.
Author information
Authors and Affiliations
Corresponding author
Additional information
Luisa Baker and Bernard Lanz contributed equally to the present work and thus share first authorship.
Rights and permissions
About this article
Cite this article
Baker, L., Lanz, B., Andreola, F. et al. New technologies – new insights into the pathogenesis of hepatic encephalopathy. Metab Brain Dis 31, 1259–1267 (2016). https://doi.org/10.1007/s11011-016-9906-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9906-y