Abstract
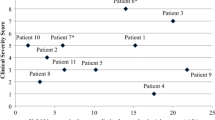
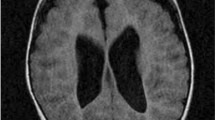
Pyridoxine-dependent epilepsy (PDE) is a pharmacoresistant epileptogenic encephalopathy controlled by pyridoxine supplementation at pharmacological doses. Despite supplementation, the long-term outcome is often poor possibly because of recurrent seizures and developmental structural brain abnormalities. We report on five patients with PDE from three unrelated families. The diagnosis was confirmed by ALDH7A1 sequencing, which allowed for the characterization of two homozygous variations [NM_001182.3:c.1279G > C - p.(Glu427Gln) and c.834G > A - p.(Val278Val)]. Brain autopsy was conducted for one untreated patient with molecularly confirmed antiquitin deficiency. Macroscopic and histological examination revealed a combination of lesions resulting from recurrent seizures and consisting of extensive areas of cortical necrosis, gliosis, and hippocampic sclerosis. The examination also revealed developmental abnormalities including corpus callosum dysgenesis and corticospinal pathfinding anomalies. This case is the second to be reported in the literature, and our findings show evidence that antiquitin is required for normal brain development and functioning. Despite prophylactic prenatal pyridoxine supplementation during the last trimester of pregnancy in one of the three families and sustained pyridoxine treatment in three living patients, the clinical outcome remained poor with delayed acquisition of neurocognitive skills. Combined therapy (pyridoxine/arginine supplementation and lysine-restricted diet) should be considered early in the course of the disease for a better long-term outcome. Enhanced knowledge of PDE features is required to improve treatment strategies.

Similar content being viewed by others
References
Babcock HE et al. (2014) aldh7a1 regulates eye and limb development in zebrafish. PLoS One 9:e101782. doi:10.1371/journal.pone.0101782
Baxter P (2001) Pyridoxine dependent and pyridoxine responsive seizures. In: Baxter P (ed) Vitamin responsive conditions in paediatric neurology. edn. MacKeith Press., London, pp 109–165
Baxter P, Griffiths P, Kelly T, Gardner-Medwin D (1996) Pyridoxine-dependent seizures: demographic, clinical, MRI and psychometric features, and effect of dose on intelligence quotient. Dev Med Child Neurol 38:998–1006
Bennett CL, Chen Y, Hahn S, Glass IA, Gospe SM Jr (2009) Prevalence of ALDH7A1 mutations in 18 North American pyridoxine-dependent seizure (PDS) patients. Epilepsia 50:1167–1175. doi:10.1111/j.1528-1167.2008.01816.x
Bok LA et al. (2010) The EEG response to pyridoxine-IV neither identifies nor excludes pyridoxine-dependent epilepsy. Epilepsia 51:2406–2411. doi:10.1111/j.1528-1167.2010.02747.x
Bok LA et al. (2012) Long-term outcome in pyridoxine-dependent epilepsy. Dev Med Child Neurol 54:849–854. doi:10.1111/j.1469-8749.2012.04347.x
Coughlin CR 2nd et al. (2015) Triple therapy with pyridoxine, arginine supplementation and dietary lysine restriction in pyridoxine-dependent epilepsy: neurodevelopmental outcome. Mol Genet Metab 116:35–43. doi:10.1016/j.ymgme.2015.05.011
Friedman SD et al. (2014) Callosal alterations in pyridoxine-dependent epilepsy. Dev Med Child Neurol 56:1106–1110. doi:10.1111/dmcn.12511
Guihard-Costa AM, Menez F, Delezoide, AL (2002) Organ weights in human fetuses after formalin fixation: standards by gestational age and body weight Pediatric and developmental pathology : the official journal of the Society for Pediatric Pathology and the Paediatric Pathology Society 5:559–578 doi:10.1007/s10024-002-0036-7
Hellstrom-Westas L, Blennow G, Rosen, I (2002) Amplitude-integrated encephalography in pyridoxine-dependent seizures and pyridoxine-responsive seizures. Acta Paediatr. (Oslo, Norway : 1992) 91:977–980
Jansen LA, Hevner RF, Roden WH, Hahn SH, Jung S, Gospe SM Jr (2014) Glial localization of antiquitin: implications for pyridoxine-dependent epilepsy. Ann Neurol 75:22–32. doi:10.1002/ana.24027
Lott IT, Coulombe T, Di Paolo RV, Richardson EP Jr, Levy HL (1978) Vitamin B6-dependent seizures: pathology and chemical findings in brain. Neurology 28:47–54
Ludwig, J (2002) Handbook of autopsy practice. third edition edn
Mills PB et al. (2006) Mutations in antiquitin in individuals with pyridoxine-dependent seizures. Nat Med 12:307–309. doi:10.1038/nm1366
Mills PB et al. (2010) Genotypic and phenotypic spectrum of pyridoxine-dependent epilepsy (ALDH7A1 deficiency). Brain J Neurol 133:2148–2159. doi:10.1093/brain/awq143
Miyasaki K, Matsumoto J, Murao S, Nakamura K, Yokoyama S, Hayano M, Nakamura H (1978) Infantile convulsion suspected of pyridoxine responsive seizures. Acta Pathol Jpn 28:741–749
Oliveira R et al. (2013) Pyridoxine-dependent epilepsy due to antiquitin deficiency: achieving a favourable outcome Epileptic disorders: international epilepsy journal with videotape 15:400–406 doi:10.1684/epd.2013.0610
Percudani R, Peracchi A (2009) The B6 database: a tool for the description and classification of vitamin B6-dependent enzymatic activities and of the corresponding protein families. BMC Biochem 10:273. doi:10.1186/1471-2105-10-273
Perez B et al. (2013) Clinical, biochemical, and molecular studies in pyridoxine-dependent epilepsy. Antisense therapy as possible new therapeutic option. Epilepsia 54:239–248. doi:10.1111/epi.12083
Proudfoot M, Jardine P, Straukiene A, Noad R, Parrish A, Ellard S, Weatherby, S (2013) Long-term follow-up of a successfully treated case of congenital pyridoxine-dependent epilepsy JIMD Rep 10:103–106 doi:10.1007/8904_2012_210
Rankin PM, Harrison S, Chong WK, Boyd S, Aylett SE (2007) Pyridoxine-dependent seizures: a family phenotype that leads to severe cognitive deficits, regardless of treatment regime. Dev Med Child Neurol 49:300–305. doi:10.1111/j.1469-8749.2007.00300.x
Riikonen R, Mankinen K, Gaily, E (2015) Long-term outcome in pyridoxine-responsive infantile epilepsy European journal of paediatric neurology : EJPN: official journal of the European Paediatric Neurology Society 19:647–651 doi:10.1016/j.ejpn.2015.08.001
Salomons GS et al. (2007) An intriguing "silent" mutation and a founder effect in antiquitin (ALDH7A1). Ann Neurol 62:414–418. doi:10.1002/ana.21206
Skvorak AB et al. (1997) An ancient conserved gene expressed in the human inner ear: identification, expression analysis, and chromosomal mapping of human and mouse antiquitin (ATQ1). Genomics 46:191–199. doi:10.1006/geno.1997.5026
Stockler S et al. (2011) Pyridoxine dependent epilepsy and antiquitin deficiency: clinical and molecular characteristics and recommendations for diagnosis, treatment and follow-up. Mol Genet Metab 104:48–60. doi:10.1016/j.ymgme.2011.05.014
Striano P et al. (2009) Two novel ALDH7A1 (antiquitin) splicing mutations associated with pyridoxine-dependent seizures. Epilepsia 50:933–936. doi:10.1111/j.1528-1167.2008.01741.x
van Karnebeek CD, Jaggumantri S (2015) Current treatment and management of pyridoxine-dependent epilepsy. Curr Treat Options Neurol 17:335. doi:10.1007/s11940-014-0335-0
van Karnebeek CD et al. (2014) Lysine-restricted diet as adjunct therapy for pyridoxine-dependent epilepsy: the pde consortium consensus recommendations. JIMD Rep 15:1–11. doi:10.1007/8904_2014_296
Acknowledgments
The authors are grateful to Hélène DRANGUET and Carine TARDIVEL-PILON for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have stated that they had no interests that might be perceived as posing a conflict or bias.
Electronic supplementary material
Supplementary Figure 1
Main imaging lesions observed in family 1. (A) T1 weighted images of coronal section passing through the hippocampi, displaying absent commissuration of callosal fibers with Probst bundles (thin white arrow) and aberrantly dispersed callosal fibers in the cincular gyrus (thick white arrow). (B) T2 weighted images of coronal section passing through the parieto-occipital lobes, showing diffuse white matter edema, with narrowing of the sulci (white arrow). (GIF 120 kb)
Supplementary Figure 2
Family trees and disease-causing variations. (A) Family trees: (red star) patients with documented epilepsy. (B) c.1279G > C - p.(Glu427Gln), control and patient P2 sequence. The mutated base is indicated by an arrow. This variation has been identified in families 1 and 2 (patients P1, P2 and P3). (C) c.834G > A - p.(Val278Val) control and patient P4 sequence. The mutated base is indicated by an arrow. This variation was identified in family 3 (patients P4 and P5). (GIF 255 kb)
Rights and permissions
About this article
Cite this article
Marguet, F., Barakizou, H., Tebani, A. et al. Pyridoxine-dependent epilepsy: report on three families with neuropathology. Metab Brain Dis 31, 1435–1443 (2016). https://doi.org/10.1007/s11011-016-9869-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9869-z