Abstract
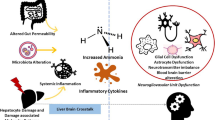
A clinical science workshop was held at the ISHEN meeting in London on Friday 11th September 2014 with the aim of thrashing out how we might translate what we know about the central role of the gut-liver-brain axis into targets which we can use in the treatment of hepatic encephalopathy (HE). This review summarises the integral role that inter-organ ammonia metabolism plays in the pathogenesis of HE with specific discussion of the roles that the small and large intestine, liver, brain, kidney and muscle assume in ammonia and glutamine metabolism. Most recently, the salivary and gut microbiome have been shown to underpin the pathophysiological changes which culminate in HE and patients with advanced cirrhosis present with enteric dysbiosis with small bowel bacterial overgrowth and translocation of bacteria and their products across a leaky gut epithelial barrier. Resident macrophages within the liver are able to sense bacterial degradation products initiating a pro-inflammatory response within the hepatic parenchyma and release of cytokines such as tumour necrosis factor alpha (TNF-α) and interleukin-8 into the systemic circulation. The endotoxemia and systemic inflammatory response that are generated predispose both to the development of infection as well as the manifestation of covert and overt HE. Co-morbidities such as diabetes and insulin resistance, which commonly accompany cirrhosis, may promote slow gut transit, promote bacterial overgrowth and increase glutaminase activity and may need to be acknowledged in HE risk stratification assessments and therapeutic regimens. Therapies are discussed which target ammonia production, utilisation or excretion at an individual organ level, or which reduce systemic inflammation and endotoxemia which are known to exacerbate the cerebral effects of ammonia in HE. The ideal therapeutic strategy would be to use an agent that can reduce hyperammonemia and reduce systemic inflammation or perhaps to adopt a combination of therapies that can address both.


Similar content being viewed by others
References
Acharya SK, Bhatia V, Sreenivas V, Khanal S, Panda SK (2009) Efficacy of L-ornithine L-aspartate in acute liver failure: a double-blind, randomized, placebo-controlled study. Gastroenterology 136:2159–2168. doi:10.1053/j.gastro.2009.02.050
Agrawal A, Sharma BC, Sharma P, Sarin SK (2012) Secondary prophylaxis of hepatic encephalopathy in cirrhosis: an open-label, randomized controlled trial of lactulose, probiotics, and no therapy. Am J Gastroenterol 107:1043–1050. doi:10.1038/ajg.2012.113
Ahboucha S, Pomier-Layrargues G, Mamer O, Butterworth RF (2006) Increased levels of pregnenolone and its neuroactive metabolite allopregnanolone in autopsied brain tissue from cirrhotic patients who died in hepatic coma. Neurochem Int 49:372–378. doi:10.1016/j.neuint.2006.02.002
Als-Nielsen B, Gluud LL, Gluud C (2004) Non-absorbable disaccharides for hepatic encephalopathy: systematic review of randomised trials. BMJ 328:1046. doi:10.1136/bmj.38048.506134.EE
Ampuero J et al (2012) Metformin inhibits glutaminase activity and protects against hepatic encephalopathy. PLoS One 7:e49279. doi:10.1371/journal.pone.0049279
Bai G, Rama Rao KV, Murthy CR, Panickar KS, Jayakumar AR, Norenberg MD (2001) Ammonia induces the mitochondrial permeability transition in primary cultures of rat astrocytes. J Neurosci Res 66:981–991
Bajaj JS (2014) The role of microbiota in hepatic encephalopathy. Gut Microbes 5:397–403. doi:10.4161/gmic.28684
Bajaj JS et al (2011) Rifaximin improves driving simulator performance in a randomized trial of patients with minimal hepatic encephalopathy. Gastroenterology 140:478–487. doi:10.1053/j.gastro.2010.08.061, e471
Bajaj JS et al (2012a) Colonic mucosal microbiome differs from stool microbiome in cirrhosis and hepatic encephalopathy and is linked to cognition and inflammation. Am J Physiol Gastrointest Liver Physiol 303:G675–G685. doi:10.1152/ajpgi.00152.2012
Bajaj JS et al (2012b) Linkage of gut microbiome with cognition in hepatic encephalopathy. Am J Physiol Gastrointest Liver Physiol 302:G168–G175. doi:10.1152/ajpgi.00190.2011
Bajaj JS et al (2013) Modulation of the metabiome by rifaximin in patients with cirrhosis and minimal hepatic encephalopathy. PLoS One 8:e60042. doi:10.1371/journal.pone.0060042
Bajaj JS et al (2014a) Randomised clinical trial: lactobacillus GG modulates gut microbiome, metabolome and endotoxemia in patients with cirrhosis. Aliment Pharmacol Ther 39:1113–1125. doi:10.1111/apt.12695
Bajaj JS et al (2014b) Altered profile of human gut microbiome is associated with cirrhosis and its complications. J Hepatol 60:940–947. doi:10.1016/j.jhep.2013.12.019
Bajaj JS et al (2015) Salivary microbiota reflects changes in gut microbiota in cirrhosis with hepatic encephalopathy. Hepatology. doi:10.1002/hep.27819
Banares R et al (2013) Extracorporeal albumin dialysis with the molecular adsorbent recirculating system in acute-on-chronic liver failure: the RELIEF trial. Hepatology 57:1153–1162. doi:10.1002/hep.26185
Banks WA, Ortiz L, Plotkin SR, Kastin AJ (1991) Human interleukin (IL) 1 alpha, murine IL-1 alpha and murine IL-1 beta are transported from blood to brain in the mouse by a shared saturable mechanism. J Pharmacol Exp Ther 259:988–996
Banks WA, Kastin AJ, Gutierrez EG (1994) Penetration of interleukin-6 across the murine blood–brain barrier. Neurosci Lett 179:53–56
Baquero F, Nombela C (2012) The microbiome as a human organ. Clin Microbiol Infect 18(Suppl 4):2–4. doi:10.1111/j.1469-0691.2012.03916.x
Bass NM et al (2010) Rifaximin treatment in hepatic encephalopathy. New Engl J Med 362:1071–1081
Basu S, Zethelius B, Helmersson J, Berne C, Larsson A, Arnlov J (2011) Cytokine-mediated inflammation is independently associated with insulin sensitivity measured by the euglycemic insulin clamp in a community-based cohort of elderly men. Int J Clin Exp Med 4:164–168
Bernal W, Hall C, Karvellas CJ, Auzinger G, Sizer E, Wendon J (2007) Arterial ammonia and clinical risk factors for encephalopathy and intracranial hypertension in acute liver failure. Hepatology 46:1844–1852. doi:10.1002/hep.21838
Berry PA et al (2011) Severity of the compensatory anti-inflammatory response determined by monocyte HLA-DR expression may assist outcome prediction in cirrhosis. Intensive Care Med 37:453–460. doi:10.1007/s00134-010-2099-7
Blei AT, Cordoba J (2001) Practice parameters committee of the American College of G. Hepatic encephalopathy. Am J Gastroenterol 96:1968–1976. doi:10.1111/j.1572-0241.2001.03964.x
Blei AT, Olafsson S, Therrien G, Butterworth RF (1994) Ammonia-induced brain edema and intracranial hypertension in rats after portacaval anastomosis. Hepatology 19:1437–1444
Bosoi CR, Rose CF (2009) Identifying the direct effects of ammonia on the brain. Metab Brain Dis 24:95–102. doi:10.1007/s11011-008-9112-7
Bosoi CR, Yang X, Huynh J, Parent-Robitaille C, Jiang W, Tremblay M, Rose CF (2012) Systemic oxidative stress is implicated in the pathogenesis of brain edema in rats with chronic liver failure. Free Radic Biol Med 52:1228–1235. doi:10.1016/j.freeradbiomed.2012.01.006
Caly WR, Strauss E (1993) A prospective study of bacterial infections in patients with cirrhosis. J Hepatol 18:353–358
Cauli O, Rodrigo R, Piedrafita B, Boix J, Felipo V (2007) Inflammation and hepatic encephalopathy: ibuprofen restores learning ability in rats with portacaval shunts. Hepatology 46:514–519. doi:10.1002/hep.21734
Cauli O, Mansouri MT, Agusti A, Felipo V (2009) Hyperammonemia increases GABAergic tone in the cerebellum but decreases it in the rat cortex. Gastroenterology 136(1359–1367):e1351–e1352. doi:10.1053/j.gastro.2008.12.057
Cenit MC, Matzaraki V, Tigchelaar EF, Zhernakova A (2014) Rapidly expanding knowledge on the role of the gut microbiome in health and disease. Biochim Biophys Acta 1842:1981–1992. doi:10.1016/j.bbadis.2014.05.023
Chastre A, Jiang W, Desjardins P, Butterworth RF (2010) Ammonia and proinflammatory cytokines modify expression of genes coding for astrocytic proteins implicated in brain edema in acute liver failure. Metab Brain Dis 25:17–21. doi:10.1007/s11011-010-9185-y
Chen MF, Mo LR, Lin RC, Kuo JY, Chang KK, Liao C, Lu FJ (1997) Increase of resting levels of superoxide anion in the whole blood of patients with decompensated liver cirrhosis. Free Radic Biol Med 23:672–679
Chen Y et al (2011) Characterization of fecal microbial communities in patients with liver cirrhosis. Hepatology 54:562–572. doi:10.1002/hep.24423
Cirera I et al (2001) Bacterial translocation of enteric organisms in patients with cirrhosis. J Hepatol 34:32–37
Clemmesen JO, Larsen FS, Kondrup J, Hansen BA, Ott P (1999) Cerebral herniation in patients with acute liver failure is correlated with arterial ammonia concentration. Hepatology 29:648–653. doi:10.1002/hep.510290309
Cordoba J et al (2001) The development of low-grade cerebral edema in cirrhosis is supported by the evolution of (1)H-magnetic resonance abnormalities after liver transplantation. J Hepatol 35:598–604
Cordoba J et al (2004) Normal protein diet for episodic hepatic encephalopathy: results of a randomized study. J Hepatol 41:38–43. doi:10.1016/j.jhep.2004.03.023
Courson A, Jones GM, Twilla JD (2015) Treatment of acute hepatic encephalopathy: comparing the effects of adding rifaximin to lactulose on patient outcomes. J Pharm Pract. doi:10.1177/0897190014566312
de Vos WM (2013) Fame and future of faecal transplantations--developing next-generation therapies with synthetic microbiomes. Microb Biotechnol 6:316–325. doi:10.1111/1751-7915.12047
Erceg S, Monfort P, Cauli O, Montoliu C, Llansola M, Piedrafita B, Felipo V (2006) Role of extracellular cGMP and of hyperammonemia in the impairment of learning in rats with chronic hepatic failure. Therapeutic implications. Neurochem Int 48:441–446. doi:10.1016/j.neuint.2005.10.016
Felig P, Owen OE, Wahren J, Cahill GF Jr (1969) Amino acid metabolism during prolonged starvation. J Clin Invest 48:584–594. doi:10.1172/JCI106017
Gluud LL et al (2013) Oral branched-chain amino acids have a beneficial effect on manifestations of hepatic encephalopathy in a systematic review with meta-analyses of randomized controlled trials. J Nutr 143:1263–1268. doi:10.3945/jn.113.174375
Gluud LL et al (2015) Branched-chain amino acids for people with hepatic encephalopathy. Cochrane Database Syst Rev 2:CD001939. doi:10.1002/14651858.CD001939.pub2
Guarner F, Malagelada JR (2003) Gut flora in health and disease. Lancet 361:512–519. doi:10.1016/S0140-6736(03)12489-0
Hassanein TI et al (2007) Randomized controlled study of extracorporeal albumin dialysis for hepatic encephalopathy in advanced cirrhosis. Hepatology 46:1853–1862. doi:10.1002/hep.21930
Haussinger D, Lamers WH, Moorman AF (1992) Hepatocyte heterogeneity in the metabolism of amino acids and ammonia. Enzyme 46:72–93
Haussinger D, Kircheis G, Fischer R, Schliess F, vom Dahl S (2000) Hepatic encephalopathy in chronic liver disease: a clinical manifestation of astrocyte swelling and low-grade cerebral edema? J Hepatol 32:1035–1038
Hermenegildo C, Montoliu C, Llansola M, Munoz MD, Gaztelu JM, Minana MD, Felipo V (1998) Chronic hyperammonemia impairs the glutamate-nitric oxide-cyclic GMP pathway in cerebellar neurons in culture and in the rat in vivo. Eur J Neurosci 10:3201–3209
Hermenegildo C, Monfort P, Felipo V (2000) Activation of N-methyl-D-aspartate receptors in rat brain in vivo following acute ammonia intoxication: characterization by in vivo brain microdialysis. Hepatology 31:709–715. doi:10.1002/hep.510310322
Human Microbiome Project C (2012) Structure, function and diversity of the healthy human microbiome. Nature 486:207–214. doi:10.1038/nature11234
Jalan R, Kapoor D (2003) Enhanced renal ammonia excretion following volume expansion in patients with well compensated cirrhosis of the liver. Gut 52:1041–1045
Jalan R, Olde Damink SW, Hayes PC, Deutz NE, Lee A (2004) Pathogenesis of intracranial hypertension in acute liver failure: inflammation, ammonia and cerebral blood flow. J Hepatol 41:613–620. doi:10.1016/j.jhep.2004.06.011
Jalan R, Wright G, Davies NA, Hodges SJ (2007) L-Ornithine phenylacetate (OP): a novel treatment for hyperammonemia and hepatic encephalopathy. Med Hypotheses 69:1064–1069. doi:10.1016/j.mehy.2006.12.061
Jiang W, Desjardins P, Butterworth RF (2009) Minocycline attenuates oxidative/nitrosative stress and cerebral complications of acute liver failure in rats. Neurochem Int 55:601–605. doi:10.1016/j.neuint.2009.06.001
Jiang W et al (2015) Dysbiosis gut microbiota associated with inflammation and impaired mucosal immune function in intestine of humans with non-alcoholic fatty liver disease. Sci Rep 5:8096. doi:10.1038/srep08096
Jones EA (2003) Potential mechanisms of enhanced GABA-mediated inhibitory neurotransmission in liver failure. Neurochem Int 43:509–516
Kawaguchi T, Izumi N, Charlton MR, Sata M (2011) Branched-chain amino acids as pharmacological nutrients in chronic liver disease. Hepatology 54:1063–1070. doi:10.1002/hep.24412
Kircheis G, Wettstein M, Dahl S, Haussinger D (2002) Clinical efficacy of L-ornithine-L-aspartate in the management of hepatic encephalopathy. Metab Brain Dis 17:453–462
Lozupone CA, Stombaugh JI, Gordon JI, Jansson JK, Knight R (2012) Diversity, stability and resilience of the human gut microbiota. Nature 489:220–230. doi:10.1038/nature11550
Maffeo E, Montuschi A, Stura G, Giordana MT (2014) Chronic acquired hepatocerebral degeneration, pallidal T1 MRI hyperintensity and manganese in a series of cirrhotic patients. Neurol Sci 35:523–530. doi:10.1007/s10072-013-1458-x
Marchesini G, Fabbri A, Bianchi G, Brizi M, Zoli M (1996) Zinc supplementation and amino acid-nitrogen metabolism in patients with advanced cirrhosis. Hepatology 23:1084–1092. doi:10.1053/jhep.1996.v23.pm0008621138
Marchesini G et al (2003) Nutritional supplementation with branched-chain amino acids in advanced cirrhosis: a double-blind, randomized trial. Gastroenterology 124:1792–1801
Martinez-Hernandez A, Bell KP, Norenberg MD (1977) Glutamine synthetase: glial localization in brain. Science 195:1356–1358
McGee RG, Bakens A, Wiley K, Riordan SM, Webster AC (2011) Probiotics for patients with hepatic encephalopathy. Cochrane Database Syst Rev:CD008716 doi:10.1002/14651858.CD008716.pub2
Montoliu C et al (2011) 3-nitro-tyrosine as a peripheral biomarker of minimal hepatic encephalopathy in patients with liver cirrhosis. Am J Gastroenterol 106:1629–1637. doi:10.1038/ajg.2011.123
Morgan XC, Segata N, Huttenhower C (2013) Biodiversity and functional genomics in the human microbiome. Trends Genet 29:51–58. doi:10.1016/j.tig.2012.09.005
Muto Y et al (2005) Effects of oral branched-chain amino acid granules on event-free survival in patients with liver cirrhosis. Clin Gastroenterol Hepatol Off Clin Pract J Am Gastroenterol Assoc 3:705–713
Nagaraja TN, Brookes N (1998) Intracellular acidification induced by passive and active transport of ammonium ions in astrocytes. Am J Physiol 274:C883–C891
Nava GM, Stappenbeck TS (2011) Diversity of the autochthonous colonic microbiota. Gut Microbes 2:99–104. doi:10.4161/gmic.2.2.15416
Nolan JP (2010) The role of intestinal endotoxin in liver injury: a long and evolving history. Hepatology 52:1829–1835. doi:10.1002/hep.23917
Odeh M, Sabo E, Srugo I, Oliven A (2005) Relationship between tumor necrosis factor-alpha and ammonia in patients with hepatic encephalopathy due to chronic liver failure. Ann Med 37:603–612. doi:10.1080/07853890500317414
Olde Damink SW, Deutz NE, Dejong CH, Soeters PB, Jalan R (2002) Interorgan ammonia metabolism in liver failure. Neurochem Int 41:177–188
Olde Damink SW et al (2003) The kidney plays a major role in the hyperammonemia seen after simulated or actual GI bleeding in patients with cirrhosis. Hepatology 37:1277–1285. doi:10.1053/jhep.2003.50221
Ong JP et al (2003) Correlation between ammonia levels and the severity of hepatic encephalopathy. Am J Med 114:188–193
Owyang C, Wu GD (2014) The gut microbiome in health and disease. Gastroenterology 146:1433–1436. doi:10.1053/j.gastro.2014.03.032
Patel V et al (2014) Rifaximin is efficacious in the treatment of chronic overt hepatic encephalopathy: a UK liver multi-centre experience. Gut 63(Suppl 1):A14–A15. doi:10.1136/gutjnl-2014-307263.29
Plauth M et al (1993) Long-term treatment of latent portosystemic encephalopathy with branched-chain amino acids. A double-blind placebo-controlled crossover study. J Hepatol 17:308–314
Pomier-Layrargues G, Spahr L, Butterworth RF (1995) Increased manganese concentrations in pallidum of cirrhotic patients. Lancet 345:735
Poo JL et al (2006) Efficacy of oral L-ornithine-L-aspartate in cirrhotic patients with hyperammonemic hepatic encephalopathy. Results of a randomized, lactulose-controlled study. Ann Hepatol 5:281–288
Prasad S, Dhiman RK, Duseja A, Chawla YK, Sharma A, Agarwal R (2007) Lactulose improves cognitive functions and health-related quality of life in patients with cirrhosis who have minimal hepatic encephalopathy. Hepatology 45:549–559. doi:10.1002/hep.21533
Qin J et al (2010) A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464:59–65. doi:10.1038/nature08821
Qin N et al (2014) Alterations of the human gut microbiome in liver cirrhosis. Nature 513:59–64. doi:10.1038/nature13568
Quinlan GJ, Martin GS, Evans TW (2005) Albumin: biochemical properties and therapeutic potential. Hepatology 41:1211–1219. doi:10.1002/hep.20720
Qvartskhava N et al (2015) Hyperammonemia in gene-targeted mice lacking functional hepatic glutamine synthetase. Proc Natl Acad Sci U S A 112:5521–5526. doi:10.1073/pnas.1423968112
Rahimi RS, Singal AG, Cuthbert JA, Rockey DC (2014) Lactulose vs polyethylene glycol 3350--electrolyte solution for treatment of overt hepatic encephalopathy: the HELP randomized clinical trial. JAMA Intern Med 174:1727–1733. doi:10.1001/jamainternmed.2014.4746
Rockey DC et al (2014) Randomized, double-blind, controlled study of glycerol phenylbutyrate in hepatic encephalopathy. Hepatology 59:1073–1083. doi:10.1002/hep.26611
Rolando N, Wade J, Davalos M, Wendon J, Philpott-Howard J, Williams R (2000) The systemic inflammatory response syndrome in acute liver failure. Hepatology 32:734–739. doi:10.1053/jhep.2000.17687
Romero LI, Tatro JB, Field JA, Reichlin S (1996) Roles of IL-1 and TNF-alpha in endotoxin-induced activation of nitric oxide synthase in cultured rat brain cells. Am J Physiol 270:R326–R332
Romero-Gomez M, Ramos-Guerrero R, Grande L, de Teran LC, Corpas R, Camacho I, Bautista JD (2004) Intestinal glutaminase activity is increased in liver cirrhosis and correlates with minimal hepatic encephalopathy. J Hepatol 41:49–54. doi:10.1016/j.jhep.2004.03.021
Rose C, Michalak A, Pannunzio P, Therrien G, Quack G, Kircheis G, Butterworth RF (1998) L-ornithine-L-aspartate in experimental portal-systemic encephalopathy: therapeutic efficacy and mechanism of action. Metab Brain Dis 13:147–157
Sharma BC, Sharma P, Agrawal A, Sarin SK (2009) Secondary prophylaxis of hepatic encephalopathy: an open-label randomized controlled trial of lactulose versus placebo. Gastroenterology 137:885–891. doi:10.1053/j.gastro.2009.05.056, 891 e881
Shawcross DL, Jalan R (2004) Treatment of hepatic encephalopathy: it’s not lactulose. BMJ 329:112. doi:10.1136/bmj.329.7457.112, author reply 112
Shawcross DL, Davies NA, Williams R, Jalan R (2004) Systemic inflammatory response exacerbates the neuropsychological effects of induced hyperammonemia in cirrhosis. J Hepatol 40:247–254
Shawcross DL et al (2008) Ammonia impairs neutrophil phagocytic function in liver disease. Hepatology 48:1202–1212. doi:10.1002/hep.22474
Shawcross DL et al (2011) Infection and systemic inflammation, not ammonia, are associated with Grade 3/4 hepatic encephalopathy, but not mortality in cirrhosis. J Hepatol 54:640–649. doi:10.1016/j.jhep.2010.07.045
Shreiner AB, Kao JY, Young VB (2015) The gut microbiome in health and in disease. Curr Opin Gastroenterol 31:69–75. doi:10.1097/MOG.0000000000000139
Sidhu SS, Goyal O, Mishra BP, Sood A, Chhina RS, Soni RK (2011) Rifaximin improves psychometric performance and health-related quality of life in patients with minimal hepatic encephalopathy (the RIME Trial). Am J Gastroenterol 106:307–316. doi:10.1038/ajg.2010.455
Simon-Talero M et al (2013) Effects of intravenous albumin in patients with cirrhosis and episodic hepatic encephalopathy: a randomized double-blind study. J Hepatol 59:1184–1192. doi:10.1016/j.jhep.2013.07.020
Singh R et al (2011) Status of bacterial colonization, Toll-like receptor expression and nuclear factor-kappa B activation in normal and diseased human livers. Clin Immunol 138:41–49. doi:10.1016/j.clim.2010.09.006
Smith PM et al (2013) The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 341:569–573. doi:10.1126/science.1241165
Sorensen M, Keiding S (2007) New findings on cerebral ammonia uptake in HE using functional (13)N-ammonia PET. Metab Brain Dis 22:277–284. doi:10.1007/s11011-007-9066-1
Stenbog P, Busk T, Larsen FS (2013) Efficacy of liver assisting in patients with hepatic encephalopathy with special focus on plasma exchange. Metab Brain Dis 28:333–335. doi:10.1007/s11011-013-9403-5
Streetz K et al (2000) Tumor necrosis factor alpha in the pathogenesis of human and murine fulminant hepatic failure. Gastroenterology 119:446–460
Takada Y et al (2001) Increased intracranial pressure in a porcine model of fulminant hepatic failure using amatoxin and endotoxin. J Hepatol 34:825–831
Takuma Y, Nouso K, Makino Y, Hayashi M, Takahashi H (2010) Clinical trial: oral zinc in hepatic encephalopathy. Aliment Pharmacol Ther 32:1080–1090. doi:10.1111/j.1365-2036.2010.04448.x
Taylor NJ et al (2013) Circulating neutrophil dysfunction in acute liver failure. Hepatology 57:1142–1152. doi:10.1002/hep.26102
Taylor NJ et al (2014) The severity of circulating neutrophil dysfunction in patients with cirrhosis is associated with 90-day and 1-year mortality. Aliment Pharmacol Ther 40:705–715. doi:10.1111/apt.12886
Turnbaugh PJ, Ley RE, Hamady M, Fraser-Liggett CM, Knight R, Gordon JI (2007) The human microbiome project. Nature 449:804–810. doi:10.1038/nature06244
Vaquero J et al (2003) Infection and the progression of hepatic encephalopathy in acute liver failure. Gastroenterology 125:755–764. doi:10.1016/s0016-5085(03)01051-5
Ventura-Cots M et al (2013) Safety of ornithine phenylacetate in cirrhotic decompensated patients: an open-label, dose-escalating, single-cohort study. J Clin Gastroenterol 47:881–887. doi:10.1097/MCG.0b013e318299c789
Vilstrup H et al (2014) Hepatic encephalopathy in chronic liver disease: 2014 practice guideline by the American association for the study of liver diseases and the european association for the study of the liver. Hepatology 60:715–735. doi:10.1002/hep.27210
Wiest R, Garcia-Tsao G (2005) Bacterial translocation (BT) in cirrhosis. Hepatology 41:422–433. doi:10.1002/hep.20632
Wright G et al (2007a) Endotoxemia produces coma and brain swelling in bile duct ligated rats. Hepatology 45:1517–1526. doi:10.1002/hep.21599
Wright G, Shawcross D, Olde Damink SW, Jalan R (2007b) Brain cytokine flux in acute liver failure and its relationship with intracranial hypertension. Metab Brain Dis 22:375–388. doi:10.1007/s11011-007-9071-4
Xu J, Ma R, Chen LF, Zhao LJ, Chen K, Zhang RB (2014) Effects of probiotic therapy on hepatic encephalopathy in patients with liver cirrhosis: an updated meta-analysis of six randomized controlled trials. Hepatobiliary Pancreat Dis Int: HBPD INT 13:354–360
Yoshida Y, Higashi T, Nouso K, Nakatsukasa H, Nakamura SI, Watanabe A, Tsuji T (2001) Effects of zinc deficiency/zinc supplementation on ammonia metabolism in patients with decompensated liver cirrhosis. Acta Med Okayama 55:349–355
Ytrebo LM et al (2009) L-ornithine phenylacetate attenuates increased arterial and extracellular brain ammonia and prevents intracranial hypertension in pigs with acute liver failure. Hepatology 50:165–174. doi:10.1002/hep.22917
Zemtsova I, Gorg B, Keitel V, Bidmon HJ, Schror K, Haussinger D (2011) Microglia activation in hepatic encephalopathy in rats and humans. Hepatology 54:204–215. doi:10.1002/hep.24326
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patel, V.C., White, H., Støy, S. et al. Clinical science workshop: targeting the gut-liver-brain axis. Metab Brain Dis 31, 1327–1337 (2016). https://doi.org/10.1007/s11011-015-9743-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-015-9743-4