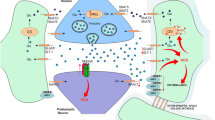
Abstract
Epilepsy is a family of brain disorders with a largely unknown etiology and high percentage of pharmacoresistance. The clinical manifestations of epilepsy are seizures, which originate from aberrant neuronal synchronization and hyperexcitability. Reactive astrocytosis, a hallmark of the epileptic tissue, develops into loss-of-function of glutamine synthetase, impairment of glutamate-glutamine cycle and increase in extracellular and astrocytic glutamate concentration. Here, we argue that chronically elevated intracellular glutamate level in astrocytes is instrumental to alterations in the metabolism of glycogen and leads to the synthesis of polyglucosans. Unaccessibility of glycogen-degrading enzymes to these insoluble molecules compromises the glycogenolysis-dependent reuptake of extracellular K+ by astrocytes, thereby leading to increased extracellular K+ and associated membrane depolarization. Based on current knowledge, we propose that the deterioration in structural homogeneity of glycogen particles is relevant to disruption of brain K+ homeostasis and increased susceptibility to seizures in epilepsy.
Similar content being viewed by others
Abbreviations
- FBPase:
-
Fructose-1,6-bisphosphatase
- GS:
-
Glutamine synthetase
- GSK3:
-
Glycogen synthase kinase 3
- LD:
-
Lafora disease
- MSO:
-
L-methionine-SR-sulfoximine
- NKA:
-
Na+-K+-activated adenosintrisphosphatase
- NREM:
-
Non rapid eye movement
- PC:
-
Pyruvate carboxylase
- PEPCK:
-
Phosphoenolpyruvate carboxykinase
- PGB:
-
Polyglucosan body
- TCA:
-
Tricarboxylic acid
- VIP:
-
Vasoactive intestinal peptide
References
Abel TJ, Hebb AO, Keene CD, Born DE, Silbergeld DL (2010) Parahippocampal corpora amylacea: case report. Neurosurgery 66:E1206–E1207
Agari T, Kobayashi K, Watanabe K, Date I, Ohtsuka Y (2012) Cryptogenic west syndrome and subsequent mesial temporal lobe epilepsy. Epileptic Disord 14:334–339
Amzica F, Massimini M, Manfridi A (2002) Spatial buffering during slow and paroxysmal sleep oscillations in cortical networks of glial cells in vivo. J Neurosci 22:1042–1053
Badawy RA, Curatolo JM, Newton M, Berkovic SF, Macdonell RA (2006) Sleep deprivation increases cortical excitability in epilepsy: syndrome-specific effects. Neurology 67:1018–1022
Ballanyi K, Grafe P, ten Bruggencate G (1987) Ion activities and potassium uptake mechanisms of glial cells in guinea-pig olfactory cortex slices. J Physiol 382:159–174
Benington JH, Heller HC (1995) Restoration of brain energy metabolism as the function of sleep. Prog Neurobiol 45:347–360
Bernard-Helary K, Lapouble E, Ardourel M, Hevor T, Cloix JF (2000) Correlation between brain glycogen and convulsive state in mice submitted to methionine sulfoximine. Life Sci 67:1773–1781
Boison D (2011) Methylxanthines, seizures, and excitotoxicity. Handb Exp Pharmacol 200:251–266
Boison D, Stewart KA (2009) Therapeutic epilepsy research: from pharmacological rationale to focal adenosine augmentation. Biochem Pharmacol 78:1428–1437
Boissonnet A, Hevor T, Landemarre L, Cloix JF (2013) Monoamines and glycogen levels in cerebral cortices of fast and slow methionine sulfoximine-inbred mice. Epilepsy Res 104:217–225
Bolanos JP, Almeida A (2010) The pentose-phosphate pathway in neuronal survival against nitrosative stress. IUBMB Life 62:14–18
Cavanagh JB (1999) Corpora-amylacea and the family of polyglucosan diseases. Brain Res Brain Res Rev 29:265–295
Cavanagh JB, Jones HB (2000) Glycogenosomes in the aging rat brain: their occurrence in the visual pathways. Acta Neuropathol 99:496–502
Cavus I, Kasoff WS, Cassaday MP, Jacob R, Gueorguieva R, Sherwin RS, Krystal JH, Spencer DD, Abi-Saab WM (2005) Extracellular metabolites in the cortex and hippocampus of epileptic patients. Ann Neurol 57:226–235
Cherian PJ, Radhakrishnan VV, Radhakrishnan K (2003) The significance of corpora amylacea in mesial temporal lobe epilepsy. Neurol India 51:277–279
Chever O, Djukic B, McCarthy KD, Amzica F (2010) Implication of Kir4.1 channel in excess potassium clearance: an in vivo study on anesthetized glial-conditional Kir4.1 knock-out mice. J Neurosci 30:15769–15777
Choi HB, Gordon GR, Zhou N, Tai C, Rungta RL, Martinez J, Milner TA, Ryu JK, McLarnon JG, Tresguerres M, Levin LR, Buck J, MacVicar BA (2012) Metabolic communication between astrocytes and neurons via bicarbonate-responsive soluble adenylyl cyclase. Neuron 75:1094–1104
Chow SY, Yen-Chow YC, White HS, Hertz L, Woodbury DM (1991) Effects of potassium on the anion and cation contents of primary cultures of mouse astrocytes and neurons. Neurochem Res 16:1275–1283
Chung MH, Horoupian DS (1996) Corpora amylacea: a marker for mesial temporal sclerosis. J Neuropathol Exp Neurol 55:403–408
Claudio OI, Berrios N, Garcia M, Casasnovas R, Ortiz JG (2002) Veratridine, but not elevated K+, inhibits excitatory amino acid transporter activity in rat hippocampal slices. Epilepsia 43(Suppl 5):184–187
Cloix JF, Hevor T (2009) Epilepsy, regulation of brain energy metabolism and neurotransmission. Curr Med Chem 16:841–853
Cloix JF, Hevor T (2011) Glycogen as a putative target for diagnosis and therapy in brain pathologies. ISRN Pathology. doi:10.5402/2011/930729
Criado O, Aguado C, Gayarre J, Duran-Trio L, Garcia-Cabrero AM, Vernia S, San Millan B, Heredia M, Roma-Mateo C, Mouron S, Juana-Lopez L, Dominguez M, Navarro C, Serratosa JM, Sanchez M, Sanz P, Bovolenta P, Knecht E, Rodriguez de Cordoba S (2012) Lafora bodies and neurological defects in malin-deficient mice correlate with impaired autophagy. Hum Mol Genet 21:1521–1533
Cruz NF, Dienel GA (2002) High glycogen levels in brains of rats with minimal environmental stimuli: implications for metabolic contributions of working astrocytes. J Cereb Blood Flow Metab 22:1476–1489
D’Ambrosio R (2004) The role of glial membrane ion channels in seizures and epileptogenesis. Pharmacol Ther 103:95–108
Dalsgaard MK, Madsen FF, Secher NH, Laursen H, Quistorff B (2007) High glycogen levels in the hippocampus of patients with epilepsy. J Cereb Blood Flow Metab 27:1137–1141
Das A, Balan S, Mathew A, Radhakrishnan V, Banerjee M, Radhakrishnan K (2011) Corpora amylacea deposition in the hippocampus of patients with mesial temporal lobe epilepsy: a new role for an old gene? Indian J Hum Genet 17(Suppl 1):S41–S47
de Lanerolle NC, Lee TS, Spencer DD (2010) Astrocytes and epilepsy. Neurotherapeutics 7:424–438
Delorme P, Hevor TK (1985) Glycogen particles in methionine sulfoximine epileptogenic rodent brain and liver after the administration of methionine and actinomycin D. Neuropathol Appl Neurobiol 11:117–128
Derry CP, Duncan S (2013) Sleep and epilepsy. Epilepsy Behav 26:394–404
Devinsky O, Vezzani A, Najjar S, De Lanerolle NC, Rogawski MA (2013) Glia and epilepsy: excitability and inflammation. Trends Neurosci 36:174–184
Dienel GA (2013) Astrocytic energetics during excitatory neurotransmission: what are contributions of glutamate oxidation and glycolysis? Neurochem Int 63:244–258
DiNuzzo M (2013) Kinetic analysis of glycogen turnover: relevance to human brain C-NMR spectroscopy. J Cereb Blood Flow Metab 33(10):1540–1548
DiNuzzo M, Mangia S, Maraviglia B, Giove F (2010) Glycogenolysis in astrocytes supports blood-borne glucose channeling not glycogen-derived lactate shuttling to neurons: evidence from mathematical modeling. J Cereb Blood Flow Metab 30:1895–1904
DiNuzzo M, Maraviglia B, Giove F (2011) Why does the brain (not) have glycogen? Bioessays 33:319–326
DiNuzzo M, Mangia S, Maraviglia B, Giove F (2012) The role of astrocytic glycogen in supporting the energetics of neuronal activity. Neurochem Res 37:2432–2438
DiNuzzo M, Mangia S, Maraviglia B, Giove F (2013) Regulatory mechanisms for glycogenolysis and K + uptake in brain astrocytes. Neurochem Int 63:458–464
Dringen R, Schmoll D, Cesar M, Hamprecht B (1993) Incorporation of radioactivity from [14C]lactate into the glycogen of cultured mouse astroglial cells. Evidence for gluconeogenesis in brain cells. Biol Chem Hoppe Seyler 374:343–347
Dudek FE, Yasumura T, Rash JE (1998) ‘Non-synaptic’ mechanisms in seizures and epileptogenesis. Cell Biol Int 22:793–805
Dufour S, Dufour P, Chever O, Vallee R, Amzica F (2011) In vivo simultaneous intra- and extracellular potassium recordings using a micro-optrode. J Neurosci Methods 194:206–217
Duran J, Tevy MF, Garcia-Rocha M, Calbo J, Milan M, Guinovart JJ (2012) Deleterious effects of neuronal accumulation of glycogen in flies and mice. EMBO Mol Med 4:719–729
Duran J, Gruart A, Garcia-Rocha M, Delgado-Garcia JM, Guinovart JJ (2014) Glycogen accumulation underlies neurodegeneration and autophagy impairment in Lafora disease. Hum Mol Genet. doi:10.1093/hmg/ddu024
During MJ, Spencer DD (1992) Adenosine: a potential mediator of seizure arrest and postictal refractoriness. Ann Neurol 32:618–624
Eid T, Thomas MJ, Spencer DD, Runden-Pran E, Lai JC, Malthankar GV, Kim JH, Danbolt NC, Ottersen OP, de Lanerolle NC (2004) Loss of glutamine synthetase in the human epileptogenic hippocampus: possible mechanism for raised extracellular glutamate in mesial temporal lobe epilepsy. Lancet 363:28–37
Engel J Jr, Brown WJ, Kuhl DE, Phelps ME, Mazziotta JC, Crandall PH (1982) Pathological findings underlying focal temporal lobe hypometabolism in partial epilepsy. Ann Neurol 12:518–528
Engel J Jr, Kuhl DE, Phelps ME, Rausch R, Nuwer M (1983) Local cerebral metabolism during partial seizures. Neurology 33:400–413
Erdamar S, Zhu ZQ, Hamilton WJ, Armstrong DL, Grossman RG (2000) Corpora amylacea and heat shock protein 27 in Ammon’s horn sclerosis. J Neuropathol Exp Neurol 59:698–706
Fisher RS, Pedley TA, Moody WJ Jr, Prince DA (1976) The role of extracellular potassium in hippocampal epilepsy. Arch Neurol 33:76–83
Folbergrova J (1973) Glycogen and glycogen phosphorylase in the cerebral cortex of mice under the influence of methionine sulphoximine. J Neurochem 20:547–557
Folbergrova J, Passonneau JV, Lowry OH, Schulz DW (1969) Glycogen, ammonia and related metabolities in the brain during seizures evoked by methionine sulphoximine. J Neurochem 16:191–203
Folbergrova J, Katsura KI, Siesjo BK (1996) Glycogen accumulated in the brain following insults is not degraded during a subsequent period of ischemia. J Neurol Sci 137:7–13
Gizak A, Mazurek J, Wozniak M, Maciaszczyk-Dziubinska E, Rakus D (2013) Destabilization of fructose 1,6-bisphosphatase-Z-line interactions is a mechanism of glyconeogenesis down-regulation in vivo. Biochim Biophys Acta 1833:622–628
Goldman SS (1988) Gluconeogenesis in the amphibian retina. Lactate is preferred to glutamate as the gluconeogenic precursor. Biochem J 254:359–365
Goldman SS (1990) Evidence that the gluconeogenic pathway is confined to an enriched muller cell fraction derived from the amphibian retina. Exp Eye Res 50:213–218
Hablitz JJ, Heinemann U (1989) Alterations in the microenvironment during spreading depression associated with epileptiform activity in the immature neocortex. Brain Res Dev Brain Res 46:243–252
Heinemann U, Lux HD (1977) Ceiling of stimulus induced rises in extracellular potassium concentration in the cerebral cortex of cat. Brain Res 120:231–249
Heinemann U, Konnerth A, Pumain R, Wadman WJ (1986) Extracellular calcium and potassium concentration changes in chronic epileptic brain tissue. Adv Neurol 44:641–661
Hejazi M, Fettke J, Haebel S, Edner C, Paris O, Frohberg C, Steup M, Ritte G (2008) Glucan, water dikinase phosphorylates crystalline maltodextrins and thereby initiates solubilization. Plant J 55:323–334
Helary-Bernard K, Ardourel MY, Cloix JF, Hevor T (2000) The xenobiotic methionine sulfoximine modulates carbohydrate anabolism and related genes expression in rodent brain. Toxicology 153:179–187
Hertz L (1978) An intense potassium uptake into astrocytes, its further enhancement by high concentrations of potassium, and its possible involvement in potassium homeostasis at the cellular level. Brain Res 145:202–208
Hertz L, Xu J, Song D, Du T, Yan E, Peng L (2013a) Brain glycogenolysis, adrenoceptors, pyruvate carboxylase, Na(+),K(+)-ATPase and Marie E. Gibbs' pioneering learning studies. Front Integr Neurosci 7:20
Hertz L, Xu J, Song D, Yan E, Gu L, Peng L (2013b) Astrocytic and neuronal accumulation of elevated extracellular K(+) with a 2/3 K(+)/Na(+) flux ratio-consequences for energy metabolism, osmolarity and higher brain function. Front Comput Neurosci 7:114
Hevor TK, Gayet J (1978) Fructose-1,6-biphosphatase and phosphofructokinase activities in the brain of mice submitted to methionine sulfoximine. Brain Res 150:210–215
Hevor TK, Delorme P, Beauvillain JC (1986) Glycogen synthesis and immunocytochemical study of fructose-1,6-biphosphatase in methionine sulfoximine epileptogenic rodent brain. J Cereb Blood Flow Metab 6:292–297
Ibrahim MZ (1975) Glycogen and its related enzymes of metabolism in the central nervous system. Adv Anat Embryol Cell Biol 52:3–89
Inoue N, Matsukado Y, Goto S, Miyamoto E (1988) Localization of glycogen synthase in brain. J Neurochem 50:400–405
Jensen MS, Azouz R, Yaari Y (1994) Variant firing patterns in rat hippocampal pyramidal cells modulated by extracellular potassium. J Neurophysiol 71:831–839
Jiruska P, de Curtis M, Jefferys JG, Schevon CA, Schiff SJ, Schindler K (2013) Synchronization and desynchronization in epilepsy: controversies and hypotheses. J Physiol 591:787–797
Kakita A, Kameyama S, Hayashi S, Masuda H, Takahashi H (2005) Pathologic features of dysplasia and accompanying alterations observed in surgical specimens from patients with intractable epilepsy. J Child Neurol 20:341–350
Karnovsky ML, Reich P, Anchors JM, Burrows BL (1983) Changes in brain glycogen during slow-wave sleep in the rat. J Neurochem 41:1498–1501
Kaufman EE, Driscoll BF (1992) Carbon dioxide fixation in neuronal and astroglial cells in culture. J Neurochem 58:258–262
Kawamura T, Morioka T, Nishio S, Fukui K, Fukui M (2002) Temporal lobe epilepsy and corpora amylacea in the hippocampus: clinicopathologic correlation. Neurol Res 24:563–569
Knecht E, Aguado C, Sarkar S, Korolchuk VI, Criado-Garcia O, Vernia S, Boya P, Sanz P, Rodriguez de Cordoba S, Rubinsztein DC (2010) Impaired autophagy in Lafora disease. Autophagy 6:991–993
Kong J, Shepel PN, Holden CP, Mackiewicz M, Pack AI, Geiger JD (2002) Brain glycogen decreases with increased periods of wakefulness: implications for homeostatic drive to sleep. J Neurosci 22:5581–5587
Konnerth A, Heinemann U, Yaari Y (1986) Nonsynaptic epileptogenesis in the mammalian hippocampus in vitro. I Development of seizurelike activity in low extracellular calcium J Neurophysiol 56:409–423
Kotagal P (2001) The relationship between sleep and epilepsy. Semin Pediatr Neurol 8:241–250
Kothare SV, Kaleyias J (2010) Sleep and epilepsy in children and adolescents. Sleep Med 11:674–685
Lai V, Mak HK, Yung AW, Ho WY, Hung KN (2010) Neuroimaging techniques in epilepsy. Hong Kong Med J 16:292–298
Lebovitz RM (1996) Quantitative examination of dynamic interneuronal coupling via single-spike extracellular potassium ion transients. J Theor Biol 180:11–25
Leel-Ossy L (1998) Corpora amylacea in hippocampal sclerosis. J Neurol Neurosurg Psychiatry 65:614
Liu Y, Zeng L, Ma K, Baba O, Zheng P, Liu Y, Wang Y (2013) Laforin-Malin Complex Degrades Polyglucosan Bodies in Concert with Glycogen Debranching Enzyme and Brain Isoform Glycogen Phosphorylase. Mol Neurobiol 49:645–657
Loffler T, Al-Robaiy S, Bigl M, Eschrich K, Schliebs R (2001) Expression of fructose-1,6-bisphosphatase mRNA isoforms in normal and basal forebrain cholinergic lesioned rat brain. Int J Dev Neurosci 19:279–285
Lomako J, Lomako WM, Kirkman BR, Whelan WJ (1994) The role of phosphate in muscle glycogen. Biofactors 4:167–171
Magistretti PJ (1988) Regulation of glycogenolysis by neurotransmitters in the central nervous system. Diabete Metab 14:237–246
Magistretti PJ (1990) VIP neurons in the cerebral cortex. Trends Pharmacol Sci 11:250–254
Malow BA (2004) Sleep deprivation and epilepsy. Epilepsy Curr 4:193–195
Mangia S, Giove F, DiNuzzo M (2012) Metabolic pathways and activity-dependent modulation of glutamate concentration in the human brain. Neurochem Res 37:2554–2561
Matos G, Andersen ML, do Valle AC, Tufik S (2010) The relationship between sleep and epilepsy: evidence from clinical trials and animal models. J Neurol Sci 295:1–7
McKenna MC (2013) Glutamate Pays Its Own Way in Astrocytes. Front Endocrinol (Lausanne) 4:191
McKenna MC, Sonnewald U, Huang X, Stevenson J, Zielke HR (1996) Exogenous glutamate concentration regulates the metabolic fate of glutamate in astrocytes. J Neurochem 66:386–393
McMahon J, Huang X, Yang J, Komatsu M, Yue Z, Qian J, Zhu X, Huang Y (2012) Impaired autophagy in neurons after disinhibition of mammalian target of rapamycin and its contribution to epileptogenesis. J Neurosci 32:15704–15714
McNamara JO (1994) Cellular and molecular basis of epilepsy. J Neurosci 14:3413–3425
Melendez R, Melendez-Hevia E, Cascante M (1997) How did glycogen structure evolve to satisfy the requirement for rapid mobilization of glucose? A problem of physical constraints in structure building. J Mol Evol 45:446–455
Nelken I, Yaari Y (1987) The role of interstitial potassium in the generation of low-calcium hippocampal seizures. Isr J Med Sci 23:124–131
Newby AC, Worku Y, Holmquist CA (1985) Adenosine formation. Evidence for a direct biochemical link with energy metabolism. Adv Myocardiol 6:273–284
Nishimoto M, Miyakawa H, Wada K, Furuta A (2011) Activation of the VIP/VPAC2 system induces reactive astrocytosis associated with increased expression of glutamate transporters. Brain Res 1383:43–53
Nishio S, Morioka T, Kawamura T, Fukui K, Nonaka H, Matsushima M (2001) Corpora amylacea replace the hippocampal pyramidal cell layer in a patient with temporal lobe epilepsy. Epilepsia 42:960–962
Nitschke F, Wang P, Schmieder P, Girard JM, Awrey DE, Wang T, Israelian J, Zhao X, Turnbull J, Heydenreich M, Kleinpeter E, Steup M, Minassian BA (2013) Hyperphosphorylation of glucosyl C6 carbons and altered structure of glycogen in the neurodegenerative epilepsy Lafora disease. Cell Metab 17:756–767
Odedra BR, Palmer TN (1981) A putative pathway of glyconeogenesis in skeletal muscle. Biosci Rep 1:157–165
Outlaw WH Jr, Lowry OH (1979) Measurement of 10(−7) to 10(−12) mol of potassium by stimulation of pyruvate kinase. Anal Biochem 92:370–374
Oz G, Seaquist ER, Kumar A, Criego AB, Benedict LE, Rao JP, Henry P-G, Moortele P-FVD, Gruetter R (2007) Human brain glycogen content and metabolism: implications on its role in brain energy metabolism. Am J Physiol Endocrinol Metab 292:E946–E951
Palmucci L, Anzil AP, Luh S (1982) Intra-astrocytic glycogen granules and corpora amylacea stain positively for polyglucosans: a cytochemical contribution on the fine structural polymorphism of particulate polysaccharides. Acta Neuropathol 57:99–102
Pedley TA, Fisher RS, Futamachi KJ, Prince DA (1976) Regulation of extracellular potassium concentration in epileptogenesis. Fed Proc 35:1254–1259
Perez EL, Lauritzen F, Wang Y, Lee TS, Kang D, Zaveri HP, Chaudhry FA, Ottersen OP, Bergersen LH, Eid T (2012) Evidence for astrocytes as a potential source of the glutamate excess in temporal lobe epilepsy. Neurobiol Dis 47:331–337
Petroff OA, Errante LD, Rothman DL, Kim JH, Spencer DD (2002) Neuronal and glial metabolite content of the epileptogenic human hippocampus. Ann Neurol 52:635–642
Petroff OA, Errante LD, Kim JH, Spencer DD (2003) N-acetyl-aspartate, total creatine, and myo-inositol in the epileptogenic human hippocampus. Neurology 60:1646–1651
Pfeiffer B, Elmer K, Roggendorf W, Reinhart PH, Hamprecht B (1990) Immunohistochemical demonstration of glycogen phosphorylase in rat brain slices. Histochemistry 94:73–80
Phelps CH (1975) An ultrastructural study of methionine sulphoximine-induced glycogen accumulation in astrocytes of the mouse cerebral cortex. J Neurocytol 4:479–490
Porkka-Heiskanen T, Strecker RE, McCarley RW (2000) Brain site-specificity of extracellular adenosine concentration changes during sleep deprivation and spontaneous sleep: an in vivo microdialysis study. Neuroscience 99:507–517
Qu H, Eloqayli H, Unsgard G, Sonnewald U (2001) Glutamate decreases pyruvate carboxylase activity and spares glucose as energy substrate in cultured cerebellar astrocytes. J Neurosci Res 66:1127–1132
Raben N, Plotz P, Byrne BJ (2002) Acid alpha-glucosidase deficiency (glycogenosis type II, Pompe disease). Curr Mol Med 2:145–166
Radhakrishnan VV, Rao MB, Radhakrishnan K, Thomas SV, Nayak DS, Santoshkumar B, Joseph E, Raghunath B (1999) Pathology of temporal lobe epilepsy: An analysis of 100 consecutive surgical specimens from patients with medically refractory epilepsy. Neurol India 47:196–201
Radhakrishnan A, Radhakrishnan K, Radhakrishnan VV, Mary PR, Kesavadas C, Alexander A, Sarma PS (2007) Corpora amylacea in mesial temporal lobe epilepsy: clinico-pathological correlations. Epilepsy Res 74:81–90
Rakus D, Pasek M, Krotkiewski H, Dzugaj A (2004) Interaction between muscle aldolase and muscle fructose 1,6-bisphosphatase results in the substrate channeling. Biochemistry 43:14948–14957
Ribeiro Mde C, Barbosa-Coutinho L, Mugnol F, Hilbig A, Palmini A, da Costa JC, Paglioli Neto E, Paglioli E (2003) Corpora amylacea in temporal lobe epilepsy associated with hippocampal sclerosis. Arq Neuropsiquiatr 61:942–945
Roach PJ (2011) Are there errors in glycogen biosynthesis and is laforin a repair enzyme? FEBS Lett 585:3216–3218
Roach PJ, Depaoli-Roach AA, Hurley TD, Tagliabracci VS (2012) Glycogen and its metabolism: some new developments and old themes. Biochem J 441:763–787
Robitaille Y, Carpenter S, Karpati G, DiMauro SD (1980) A distinct form of adult polyglucosan body disease with massive involvement of central and peripheral neuronal processes and astrocytes: a report of four cases and a review of the occurrence of polyglucosan bodies in other conditions such as Lafora’s disease and normal ageing. Brain 103:315–336
Rodin EA, Luby ED, Gottlieb JS (1962) The electroencephalogram during prolonged experimental sleep deprivation. Electroencephalogr Clin Neurophysiol 14:544–551
Scharf MT, Naidoo N, Zimmerman JE, Pack AI (2008) The energy hypothesis of sleep revisited. Prog Neurobiol 86:264–280
Schmoll D, Fuhrmann E, Gebhardt R, Hamprecht B (1995) Significant amounts of glycogen are synthesized from 3-carbon compounds in astroglial primary cultures from mice with participation of the mitochondrial phosphoenolpyruvate carboxykinase isoenzyme. Eur J Biochem 227:308–315
Schrader J, Wahl M, Kuschinsky W, Kreutzberg GW (1980) Increase of adenosine content in cerebral cortex of the cat during bicuculline-induced seizure. Pflugers Arch 387:245–251
Seidel JL, Shuttleworth CW (2011) Contribution of astrocyte glycogen stores to progression of spreading depression and related events in hippocampal slices. Neuroscience 192:295–303
Sellinger OZ, Schatz RA, Porta R, Wilens TE (1984) Brain methylation and epileptogenesis: the case of methionine sulfoximine. Ann Neurol 16(Suppl):S115–S120
Sickmann HM, Waagepetersen HS, Schousboe A, Benie AJ, Bouman SD (2012) Brain glycogen and its role in supporting glutamate and GABA homeostasis in a type 2 diabetes rat model. Neurochem Int 60:267–275
Sitoh YY, Tien RD (1998) Neuroimaging in epilepsy. J Magn Reson Imaging 8:277–288
Sonnewald U, Westergaard N, Petersen SB, Unsgard G, Schousboe A (1993) Metabolism of [U-13C]glutamate in astrocytes studied by 13C NMR spectroscopy: incorporation of more label into lactate than into glutamine demonstrates the importance of the tricarboxylic acid cycle. J Neurochem 61:1179–1182
Spuch C, Ortolano S, Navarro C (2012) Lafora progressive myoclonus epilepsy: recent insights into cell degeneration. Recent Pat Endocr Metab Immune Drug Discov 6:99–107
Swanson RA, Yu AC, Sharp FR, Chan PH (1989) Regulation of glycogen content in primary astrocyte culture: effects of glucose analogues, phenobarbital, and methionine sulfoximine. J Neurochem 52:1359–1365
Swanson RA, Yu AC, Chan PH, Sharp FR (1990) Glutamate increases glycogen content and reduces glucose utilization in primary astrocyte culture. J Neurochem 54:490–496
Tagliabracci VS, Girard JM, Segvich D, Meyer C, Turnbull J, Zhao X, Minassian BA, Depaoli-Roach AA, Roach PJ (2008) Abnormal metabolism of glycogen phosphate as a cause for Lafora disease. J Biol Chem 283:33816–33825
Tagliabracci VS, Heiss C, Karthik C, Contreras CJ, Glushka J, Ishihara M, Azadi P, Hurley TD, DePaoli-Roach AA, Roach PJ (2011) Phosphate incorporation during glycogen synthesis and Lafora disease. Cell Metab 13:274–282
Takahashi-Yanaga F (2013) Activator or inhibitor? GSK-3 as a new drug target. Biochem Pharmacol 86:191–199
Tani H, Dulla CG, Huguenard JR, Reimer RJ (2010) Glutamine is required for persistent epileptiform activity in the disinhibited neocortical brain slice. J Neurosci 30:1288–1300
Theodore WH, Newmark ME, Sato S, Brooks R, Patronas N, De La Paz R, DiChiro G, Kessler RM, Margolin R, Manning RG et al (1983) [18F]fluorodeoxyglucose positron emission tomography in refractory complex partial seizures. Ann Neurol 14:429–437
Valles-Ortega J, Duran J, Garcia-Rocha M, Bosch C, Saez I, Pujadas L, Serafin A, Canas X, Soriano E, Delgado-Garcia JM, Gruart A, Guinovart JJ (2011) Neurodegeneration and functional impairments associated with glycogen synthase accumulation in a mouse model of Lafora disease. EMBO Mol Med 3:667–681
van der Hel WS, Notenboom RG, Bos IW, van Rijen PC, van Veelen CW, de Graan PN (2005) Reduced glutamine synthetase in hippocampal areas with neuron loss in temporal lobe epilepsy. Neurology 64:326–333
Van Paesschen W, Revesz T, Duncan JS (1997) Corpora amylacea in hippocampal sclerosis. J Neurol Neurosurg Psychiatry 63:513–515
Vanderhaeghen JJ (1971) Correlation between ultrastructure and histochemistry of Lafora bodies. Acta Neuropathol 17:24–36
Vanderwolf CH (1988) Cerebral activity and behavior: control by central cholinergic and serotonergic systems. Int Rev Neurobiol 30:225–340
Verge V, Hevor TK (1995) Regulation of fructose-1,6-bisphosphatase activity in primary cultured astrocytes. Neurochem Res 20:1049–1056
Vilchez D, Ros S, Cifuentes D, Pujadas L, Valles J, Garcia-Fojeda B, Criado-Garcia O, Fernandez-Sanchez E, Medrano-Fernandez I, Dominguez J, Garcia-Rocha M, Soriano E, Rodriguez de Cordoba S, Guinovart JJ (2007) Mechanism suppressing glycogen synthesis in neurons and its demise in progressive myoclonus epilepsy. Nat Neurosci 10:1407–1413
Walling SG, Rigoulot MA, Scharfman HE (2007) Acute and chronic changes in glycogen phosphorylase in hippocampus and entorhinal cortex after status epilepticus in the adult male rat. Eur J Neurosci 26:178–189
Wang Y, Ma K, Wang P, Baba O, Zhang H, Parent JM, Zheng P, Liu Y, Minassian BA, Liu Y (2013) Laforin prevents stress-induced polyglucosan body formation and lafora disease progression in neurons. Mol Neurobiol 48:49–61
Xu J, Song D, Xue Z, Gu L, Hertz L, Peng L (2013) Requirement of Glycogenolysis for Uptake of Increased Extracellular K(+) in Astrocytes: Potential Implications for K (+) Homeostasis and Glycogen Usage in Brain. Neurochem Res 38:472–485
Yaari Y, Konnerth A, Heinemann U (1986) Nonsynaptic epileptogenesis in the mammalian hippocampus in vitro. II. Role of extracellular potassium. J Neurophysiol 56:424–438
Yanez AJ, Garcia-Rocha M, Bertinat R, Droppelmann C, Concha II, Guinovart JJ, Slebe JC (2004) Subcellular localization of liver FBPase is modulated by metabolic conditions. FEBS Lett 577:154–158
Acknowledgments
The author S.M. thanks the grant KL2 RR033182 from the National Insititute of Health (NIH) to the University of Minnesota Clinical and Translational Science Institute (CTSI) for support.
Disclosure/Conflict of interests
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
DiNuzzo, M., Mangia, S., Maraviglia, B. et al. Does abnormal glycogen structure contribute to increased susceptibility to seizures in epilepsy?. Metab Brain Dis 30, 307–316 (2015). https://doi.org/10.1007/s11011-014-9524-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-014-9524-5