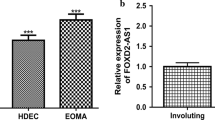
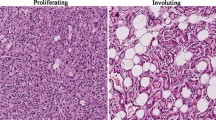
Abstract
Our work aims to investigate long non-coding RNA (lncRNA) N6-methyladenosine (m6A) modification and its role in infantile hemangioma (IH). The mRNA and protein expression levels were assessed using quantitative real-time polymerase chain reaction, western blot and immunohistochemistry. Me-RIP assay was performed to evaluate lncRNA NEAT1 m6A levels. Cell proliferation, migration and invasion were evaluated using cell counting kit-8 assay, transwell migration and invasion assay, respectively. Photo-activatable ribonucleoside-enhanced crosslinking and immunoprecipitation assay was conducted to verify the binding relationship between lncRNA nuclear paraspeckle assembly transcript 1 (NEAT1) and ALKBH5 (an RNA demethylase). The binding relationship between lncRNA NEAT1, microRNA (miR)-378b and FOS-like antigen 1 (FOSL1) was verified using dual-luciferase reporter gene assay and/or RNA immunoprecipitation assay. ALKBH5, lncRNA NEAT1 and FOLS1 expression was elevated in IH tissues, while miR-378b was downregulated. ALKBH5 knockdown suppressed cell proliferation, migration and invasion of IH cells, while promoting cell apoptosis. ALKBH5 promoted lncRNA NEAT1 expression by reducing the m6A modification of lncRNA NEAT1. In addition, miR‐378b was the target of lncRNA NEAT1, and its overexpression reversed the promotion effect of lncRNA NEAT1 overexpression on IH cell tumor-like behaviors. Moreover, FOLS1 was the target of miR-378b, and its overexpression reversed the inhibitory effect of miR-378b overexpression on IH cell tumor-like behaviors in vitro. ALKBH5 might have great potential as therapeutic target for IH, since ALKBH5 silencing suppressed IH progression by regulation of the NEAT1/miR-378b/FOSL1 axis.






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Material availability
All data generated or analyzed during this study are included in this article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ALKBH5:
-
Alkylation repair homolog protein 5
- ANOVA:
-
Analysis of variance
- CCK8:
-
Cell counting kit-8
- ceRNAs:
-
Competing endogenous RNAs
- DMEM:
-
Dulbecco's modified eagle media
- FOSL1:
-
FOS-like antigen 1
- FAR1:
-
Fos-related antigen 1
- IHC:
-
Immunohistochemistry
- IH:
-
Infantile hemangioma
- lncRNA:
-
Long non-coding RNA
- Me-RIP:
-
Methylated RNA binding protein immunoprecipitation
- miR:
-
MicroRNA
- NF1:
-
Neurofibromatosis 1
- ncRNA:
-
Non-coding RNA
- m6A:
-
N6-Methyladenosine
- NEAT1:
-
Nuclear paraspeckle assembly transcript 1
- PAR-CLIP:
-
Photo-activatable ribonucleoside-enhanced crosslinking and immunoprecipitation
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- RIP:
-
RNA immunoprecipitation
- SDS-PAGE:
-
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
- SD:
-
Standard deviation
References
Solman L, Glover M, Beattie PE, Buckley H, Clark S, Gach JE, Giardini A, Helbling I, Hewitt RJ, Laguda B, Langan SM, Martinez AE, Murphy R, Proudfoot L, Ravenscroft J, Shahidullah H, Shaw L, Syed SB, Wells L, Flohr C (2018) Oral propranolol in the treatment of proliferating infantile haemangiomas: British Society for Paediatric Dermatology consensus guidelines. Br J Dermatol 179:582–589. https://doi.org/10.1111/bjd.16779
Léauté-Labrèze C, Harper JI, Hoeger PH (2017) Infantile haemangioma. Lancet 390:85–94. https://doi.org/10.1016/s0140-6736(16)00645-0
Smith CJF, Friedlander SF, Guma M, Kavanaugh A, Chambers CD (2017) Infantile hemangiomas: an updated review on risk factors, pathogenesis, and treatment. Birth Defects Res 109:809–815. https://doi.org/10.1002/bdr2.1023
Roundtree IA, Evans ME, Pan T, He C (2017) Dynamic RNA modifications in gene expression regulation. Cell 169:1187–1200. https://doi.org/10.1016/j.cell.2017.05.045
Nilsen TW (2014) Molecular biology. Internal mRNA methylation finally finds functions. Science 343:1207–1208. https://doi.org/10.1126/science.1249340
Guo T, Liu DF, Peng SH, Xu AM (2020) ALKBH5 promotes colon cancer progression by decreasing methylation of the lncRNA NEAT1. Am J Transl Res 12:4542–4549
Chen S, Zhou L, Wang Y (2020) ALKBH5-mediated m(6)A demethylation of lncRNA PVT1 plays an oncogenic role in osteosarcoma. Cancer Cell Int 20:34. https://doi.org/10.1186/s12935-020-1105-6
Zhao H, Xu Y, Xie Y, Zhang L, Gao M, Li S, Wang F (2021) m6A regulators is differently expressed and correlated with immune response of esophageal cancer. Front Cell Dev Biol 9:650023. https://doi.org/10.3389/fcell.2021.650023
Zhang S, Zhao BS, Zhou A, Lin K, Zheng S, Lu Z, Chen Y, Sulman EP, Xie K, Bögler O, Majumder S, He C, Huang S (2017) m(6)A demethylase ALKBH5 maintains tumorigenicity of glioblastoma stem-like cells by sustaining FOXM1 expression and cell proliferation program. Cancer Cell 31:591-606.e6. https://doi.org/10.1016/j.ccell.2017.02.013
Deng G, Sui G (2013) Noncoding RNA in oncogenesis: a new era of identifying key players. Int J Mol Sci 14:18319–18349. https://doi.org/10.3390/ijms140918319
Chakravarty D, Sboner A, Nair SS, Giannopoulou E, Li R, Hennig S, Mosquera JM, Pauwels J, Park K, Kossai M, MacDonald TY, Fontugne J, Erho N, Vergara IA, Ghadessi M, Davicioni E, Jenkins RB, Palanisamy N, Chen Z, Nakagawa S, Hirose T, Bander NH, Beltran H, Fox AH, Elemento O, Rubin MA (2014) The oestrogen receptor alpha-regulated lncRNA NEAT1 is a critical modulator of prostate cancer. Nat Commun 5:5383. https://doi.org/10.1038/ncomms6383
Chen X, Kong J, Ma Z, Gao S, Feng X (2015) Up regulation of the long non-coding RNA NEAT1 promotes esophageal squamous cell carcinoma cell progression and correlates with poor prognosis. Am J Cancer Res 5:2808–2815
Fu JW, Kong Y, Sun X (2016) Long noncoding RNA NEAT1 is an unfavorable prognostic factor and regulates migration and invasion in gastric cancer. J Cancer Res Clin Oncol 142:1571–1579. https://doi.org/10.1007/s00432-016-2152-1
Yu L, Shu H, Xing L, Lv MX, Li L, Xie YC, Zhang Z, Zhang L, Xie YY (2020) Silencing long non-coding RNA NEAT1 suppresses the tumorigenesis of infantile hemangioma by competitively binding miR-33a-5p to stimulate HIF1α/NF-κB pathway. Mol Med Rep 22:3358–3366. https://doi.org/10.3892/mmr.2020.11409
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297. https://doi.org/10.1016/s0092-8674(04)00045-5
Tan X, Banerjee P, Liu X, Yu J, Gibbons DL, Wu P, Scott KL, Diao L, Zheng X, Wang J, Jalali A, Suraokar M, Fujimoto J, Behrens C, Liu X, Liu CG, Creighton CJ, Wistuba II, Kurie JM (2018) The epithelial-to-mesenchymal transition activator ZEB1 initiates a prometastatic competing endogenous RNA network. J Clin Invest 128:1267–1282. https://doi.org/10.1172/jci97225
Liu H, Deng H, Zhao Y, Li C, Liang Y (2018) LncRNA XIST/miR-34a axis modulates the cell proliferation and tumor growth of thyroid cancer through MET-PI3K-AKT signaling. J Exp Clin Cancer Res 37:279. https://doi.org/10.1186/s13046-018-0950-9
Wang G, Xu G, Wang W (2020) Long noncoding RNA CDKN2B-AS1 facilitates lung cancer development through regulating miR-378b/NR2C2. Onco Targets Ther 13:10641–10649. https://doi.org/10.2147/ott.S261973
Dhillon AS, Tulchinsky E (2015) FRA-1 as a driver of tumour heterogeneity: a nexus between oncogenes and embryonic signalling pathways in cancer. Oncogene 34:4421–4428. https://doi.org/10.1038/onc.2014.374
Wu ZB, Shi SL, Pan FJ, Li L, Chen HY (2021) Propranolol inhibits infantile hemangioma by regulating the miR-424/vascular endothelial growth factor-A (VEGFA) axis. Transl Pediatr 10:1867–1876. https://doi.org/10.21037/tp-21-244
Li D, Li P, Guo Z, Wang H, Pan W (2017) Downregulation of miR-382 by propranolol inhibits the progression of infantile hemangioma via the PTEN-mediated AKT/mTOR pathway. Int J Mol Med 39:757–763. https://doi.org/10.3892/ijmm.2017.2863
Xiao L, Liu D, Zuo S, Zhu X, Wang Y, Dong C (2019) Urea-modulated UT-B urea transporter internalization is clathrin- and caveolae-dependent in infantile hemangioma-derived vascular endothelial cells. J Cell Biochem 120:5128–5136. https://doi.org/10.1002/jcb.27789
Li P, Xiao XE, Xu Q, Guo ZT (2011) Establishment of human infancy hemangioma-derived endothelial cell line XPTS-1 and animal model of human infancy hemangioma. Zhonghua Kou Qiang Yi Xue Za Zhi 46:129–133
Talotta F, Casalino L, Verde P (2020) The nuclear oncoprotein Fra-1: a transcription factor knocking on therapeutic applications’ door. Oncogene 39:4491–4506. https://doi.org/10.1038/s41388-020-1306-4
Ji Y, Chen S, Li K, Li L, Xu C, Xiang B (2014) Signaling pathways in the development of infantile hemangioma. J Hematol Oncol 7:13. https://doi.org/10.1186/1756-8722-7-13
Yi YC, Chen XY, Zhang J and Zhu JS (2020) Novel insights into the interplay between m(6)A modification and noncoding RNAs in cancer. Mol Cancer 19:121. https://doi.org/10.1186/s12943-020-01233-2
He L, Li H, Wu A, Peng Y, Shu G, Yin G (2019) Functions of N6-methyladenosine and its role in cancer. Mol Cancer 18:176. https://doi.org/10.1186/s12943-019-1109-9
Sun T, Wu R, Ming L (2019) The role of m6A RNA methylation in cancer. Biomed Pharmacother 112:108613. https://doi.org/10.1016/j.biopha.2019.108613
Landfors M, Nakken S, Fusser M, Dahl JA, Klungland A, Fedorcsak P (2016) Sequencing of FTO and ALKBH5 in men undergoing infertility work-up identifies an infertility-associated variant and two missense mutations. Fertil Steril 105:1170-1179.e5. https://doi.org/10.1016/j.fertnstert.2016.01.002
Zhu H, Gan X, Jiang X, Diao S, Wu H, Hu J (2019) ALKBH5 inhibited autophagy of epithelial ovarian cancer through miR-7 and BCL-2. J Exp Clin Cancer Res 38:163. https://doi.org/10.1186/s13046-019-1159-2
Schmitt AM, Chang HY (2016) Long noncoding RNAs in cancer pathways. Cancer Cell 29:452–463. https://doi.org/10.1016/j.ccell.2016.03.010
Li MM, Dong CX, Sun B, Lei HZ, Wang YL, Gong YB, Sun LL, Sun ZW (2019) LncRNA-MALAT1 promotes tumorogenesis of infantile hemangioma by competitively binding miR-424 to stimulate MEKK3/NF-κB pathway. Life Sci 239:116946. https://doi.org/10.1016/j.lfs.2019.116946
Zhou L, Jia X, Yang X (2021) LncRNA-TUG1 promotes the progression of infantile hemangioma by regulating miR-137/IGFBP5 axis. Hum Genomics 15:50. https://doi.org/10.1186/s40246-021-00349-w
Zhang M, Weng W, Zhang Q, Wu Y, Ni S, Tan C, Xu M, Sun H, Liu C, Wei P, Du X (2018) The lncRNA NEAT1 activates Wnt/β-catenin signaling and promotes colorectal cancer progression via interacting with DDX5. J Hematol Oncol 11:113. https://doi.org/10.1186/s13045-018-0656-7
Li X, Deng S, Pang X, Song Y, Luo S, Jin L, Pan Y (2019) LncRNA NEAT1 silenced miR-133b promotes migration and invasion of breast cancer cells. Int J Mol Sci. https://doi.org/10.3390/ijms20153616
Zhao D, Zhang Y, Wang N, Yu N (2017) NEAT1 negatively regulates miR-218 expression and promotes breast cancer progression. Cancer Biomark 20:247–254. https://doi.org/10.3233/cbm-170027
Zhang J, Guo S, Piao HY, Wang Y, Wu Y, Meng XY, Yang D, Zheng ZC, Zhao Y (2019) ALKBH5 promotes invasion and metastasis of gastric cancer by decreasing methylation of the lncRNA NEAT1. J Physiol Biochem 75:379–389. https://doi.org/10.1007/s13105-019-00690-8
Mong EF, Akat KM, Canfield J, Lockhart J, VanWye J, Matar A, Tsibris JCM, Wu JK, Tuschl T, Totary-Jain H (2018) Modulation of LIN28B/Let-7 Signaling by propranolol contributes to infantile hemangioma involution. Arterioscler Thromb Vasc Biol 38:1321–1332. https://doi.org/10.1161/atvbaha.118.310908
Pan WK, Li P, Guo ZT, Huang Q, Gao Y (2015) Propranolol induces regression of hemangioma cells via the down-regulation of the PI3K/Akt/eNOS/VEGF pathway. Pediatr Blood Cancer 62:1414–1420. https://doi.org/10.1002/pbc.25453
Sun W, Lan X, Zhang H, Wang Z, Dong W, He L, Zhang T, Zhang P, Liu J, Qin Y (2018) NEAT1_2 functions as a competing endogenous RNA to regulate ATAD2 expression by sponging microRNA-106b-5p in papillary thyroid cancer. Cell Death Dis 9:380. https://doi.org/10.1038/s41419-018-0418-z
Jiang X, Guo S, Zhang Y, Zhao Y, Li X, Jia Y, Xu Y, Ma B (2020) LncRNA NEAT1 promotes docetaxel resistance in prostate cancer by regulating ACSL4 via sponging miR-34a-5p and miR-204-5p. Cell Signal 65:109422. https://doi.org/10.1016/j.cellsig.2019.109422
Mei H, Xian H, Ke J (2021) LncRNA-MCM3AP-AS1 promotes the progression of infantile hemangiomas by increasing miR-138-5p/HIF-1α axis-regulated glycolysis. Front Mol Biosci 8:753218. https://doi.org/10.3389/fmolb.2021.753218
Yang L, Dai J, Li F, Cheng H, Yan D, Ruan Q (2017) The expression and function of miR-424 in infantile skin hemangioma and its mechanism. Sci Rep 7:11846. https://doi.org/10.1038/s41598-017-10674-7
Nix JS, Yuan M, Imada EL, Ames H, Marchionni L, Gutmann DH, Rodriguez FJ (2021) Global microRNA profiling identified miR-10b-5p as a regulator of neurofibromatosis 1 (NF1)-glioma migration. Neuropathol Appl Neurobiol 47:96–107. https://doi.org/10.1111/nan.12641
Vallejo A, Perurena N, Guruceaga E, Mazur PK, Martinez-Canarias S, Zandueta C, Valencia K, Arricibita A, Gwinn D, Sayles LC, Chuang CH, Guembe L, Bailey P, Chang DK, Biankin A, Ponz-Sarvise M, Andersen JB, Khatri P, Bozec A, Sweet-Cordero EA, Sage J, Lecanda F, Vicent S (2017) An integrative approach unveils FOSL1 as an oncogene vulnerability in KRAS-driven lung and pancreatic cancer. Nat Commun 8:14294. https://doi.org/10.1038/ncomms14294
Vallejo A, Valencia K, Vicent S (2017) All for one and FOSL1 for all: FOSL1 at the crossroads of lung and pancreatic cancer driven by mutant KRAS. Mol Cell Oncol 4:e1314239. https://doi.org/10.1080/23723556.2017.1314239
Zhang K, Myllymäki SM, Gao P, Devarajan R, Kytölä V, Nykter M, Wei GH, Manninen A (2017) Oncogenic K-Ras upregulates ITGA6 expression via FOSL1 to induce anoikis resistance and synergizes with αV-class integrins to promote EMT. Oncogene 36:5681–5694. https://doi.org/10.1038/onc.2017.177
Acknowledgements
We would like to give our sincere gratitude to the reviewers for their constructive comments.
Funding
This work was supported by Scientific Research Project of Hunan Provincial Health Commission (No.202206023142).
Author information
Authors and Affiliations
Contributions
KP: Conceptualization; Writing-original draft; Methodology; Formal analysis; RPX: Supervision; Validation; FZ: Data curation; YX: Resources; TDM: Investigation; ML: Software; YF: Visualization; CGZ: Project administration; Funding acquisition; Writing-review and editing. All authors have read and approved the final version of this manuscript to be published.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that there is no conflict of interest.
Ethical approval
This study passed the review of the Ethics Committee of Hunan children's Hospital before enrollment of patients and all participants signed informed consent. The animal studies were approved by Ethics Committee of Hunan children's Hospital.
Informed consent
Informed consent was obtained from study participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Peng, K., Xia, RP., Zhao, F. et al. ALKBH5 promotes the progression of infantile hemangioma through regulating the NEAT1/miR-378b/FOSL1 axis. Mol Cell Biochem 477, 1527–1540 (2022). https://doi.org/10.1007/s11010-022-04388-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04388-2