Abstract
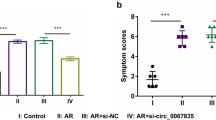
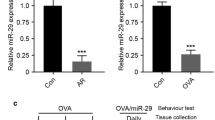
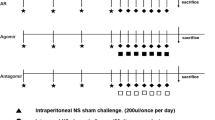
Type 2 innate lymphoid cells (ILC2s) exert an increasingly important influence on the pathological process of allergic rhinitis (AR), which is affected by microRNAs-mediated post-transcriptional regulation. This study aims to investigate the function of miR-150-5p in AR patients and the mouse model of AR. The mouse model of AR was established using the OVA challenge. The expressions of miR-150-5p, ICAM-1, p-p38 and p-GATA-3 were evaluated via RT-qPCR and western blot analysis. The level of ILC2s was examined with flow cytometry. Concentrations of OVA-specific IgE, IL-13 and IL-5 in serum were evaluated using ELISA. Histopathological examination was conducted through H&E staining. The interplay between ICAM-1 and miR-150-5p was determined through the DLR assay. The decreased miR-150-5p expression and increased ICAM-1, p-p38 and p-GATA-3 expressions and ILC2s levels were detected in AR patients and AR mice compared with controls. Treatment with miR-150-5p lentivirus alleviated AR symptoms (sneezing, rubbing, mucosa inflammation, serum type 2 cytokines and OVA-specific IgE) and lowered the ILC2s level in AR mice. MiR-150-5p was found to directly bind to 3′-UTR of ICAM-1 and downregulate ICAM-1 expression, thereby descending the level of p-p38, p-GATA-3 and suppressing ILC2s function to alleviate AR symptoms. Treatment with Lenti-ICAM-1 counteracted these protective effects of miR-150-5p. Upregulation of miR-150-5p repressed the ICAM-1/p38 axis which was vital to ILC2s development and function, thereby alleviating allergic symptoms of AR.







Similar content being viewed by others
Data availability
All datasets for the analysis in the present study are available upon reasonable request to the corresponding author.
Abbreviations
- AR:
-
Allergic rhinitis
- ICAM-1:
-
Intercellular cell adhesion molecule-1
- LFA-1:
-
Leukocyte function-associated molecule
- miRNAs:
-
MicroRNAs
- NALT:
-
Nasal-associated lymphoid tissue
- NC:
-
Negative control
- OVA:
-
Ovalbumin
- PBMCs:
-
Peripheral blood mononuclear cells
- SPF:
-
Specific pathogen-free
- Th2:
-
T-helper type 2
- ILC2s:
-
Type 2 innate lymphoid cells
References
Incorvaia C, Cavaliere C, Frati F, Masieri S (2018) Allergic rhinitis. J Biol Regul Homeost Agents 32(1 Suppl. 1):61–66
Khan DA (2014) Allergic rhinitis and asthma: epidemiology and common pathophysiology. Allergy Asthma Proc 35(5):357–361. https://doi.org/10.2500/aap.2014.35.3794
Bernstein DI, Schwartz G, Bernstein JA (2016) Allergic rhinitis: mechanisms and treatment. Immunol Allergy Clin N Am 36(2):261–278. https://doi.org/10.1016/j.iac.2015.12.004
Scanlon ST, McKenzie AN (2012) Type 2 innate lymphoid cells: new players in asthma and allergy. Curr Opin Immunol 24(6):707–712. https://doi.org/10.1016/j.coi.2012.08.009
Klose CS, Artis D (2016) Innate lymphoid cells as regulators of immunity, inflammation and tissue homeostasis. Nat Immunol 17(7):765–774. https://doi.org/10.1038/ni.3489
Christianson CA, Goplen NP, Zafar I, Irvin C, Good JT Jr, Rollins DR, Gorentla B, Liu W, Gorska MM, Chu H, Martin RJ, Alam R (2015) Persistence of asthma requires multiple feedback circuits involving type 2 innate lymphoid cells and IL-33. J Allergy Clin Immunol 136(1):59-68.e14. https://doi.org/10.1016/j.jaci.2014.11.037
Smith SG, Chen R, Kjarsgaard M, Huang C, Oliveria JP, O’Byrne PM, Gauvreau GM, Boulet LP, Lemiere C, Martin J, Nair P, Sehmi R (2016) Increased numbers of activated group 2 innate lymphoid cells in the airways of patients with severe asthma and persistent airway eosinophilia. J Allergy Clin Immunol 137(1):75-86.e78. https://doi.org/10.1016/j.jaci.2015.05.037
Fan D, Wang X, Wang M, Wang Y, Zhang L, Li Y, Fan E, Cao F, Van Crombruggen K, Zhang L (2016) Allergen-dependent differences in ILC2s frequencies in patients with allergic rhinitis. Allergy Asthma Immunol Res 8(3):216–222. https://doi.org/10.4168/aair.2016.8.3.216
Sun R, Yang Y, Huo Q, Gu Z, Wei P, Tang X (2020) Increased expression of type 2 innate lymphoid cells in pediatric patients with allergic rhinitis. Exp Ther Med 19(1):735–740. https://doi.org/10.3892/etm.2019.8235
Eifan AO, Durham SR (2016) Pathogenesis of rhinitis. Clin Exp Allergy 46(9):1139–1151. https://doi.org/10.1111/cea.12780
Xiao L, Jiang L, Hu Q, Li Y (2018) MiR-302e attenuates allergic inflammation in vitro model by targeting RelA. Biosci Rep. https://doi.org/10.1042/BSR20180025
Zhu Y, Liu Y, Zhu X, Wang Z, Wang M (2020) Upregulation of miR-155 regulates group 2 innate lymphoid cells by targeting c-maf in allergic rhinitis. Eur J Pharmacol 887:173564. https://doi.org/10.1016/j.ejphar.2020.173564
Li Y, Yu J, Wang F, Guo R, Xing H, Chen Y, Chen D, Xie X, Wan D, Jiang Z (2021) MiR-150-5p regulate T cell activation in severe aplastic anemia by targeting Bach2. Cell Tissue Res. https://doi.org/10.1007/s00441-020-03373-9
Chen Z, Wang H, Xia Y, Yan F, Lu Y (2018) Therapeutic potential of mesenchymal cell-derived miRNA-150-5p-expressing exosomes in rheumatoid arthritis mediated by the modulation of MMP14 and VEGF. J Immunol 201(8):2472–2482. https://doi.org/10.4049/jimmunol.1800304
Neamah WH, Singh NP, Alghetaa H, Abdulla OA, Chatterjee S, Busbee PB, Nagarkatti M, Nagarkatti P (2019) AhR activation leads to massive mobilization of myeloid-derived suppressor cells with immunosuppressive activity through regulation of CXCR2 and MicroRNA miR-150-5p and miR-543-3p that target anti-inflammatory genes. J Immunol 203(7):1830–1844. https://doi.org/10.4049/jimmunol.1900291
Wei S, Liu Q (2019) Long noncoding RNA MALAT1 modulates sepsis-induced cardiac inflammation through the miR-150-5p/NF-kappaB axis. Int J Clin Exp Pathol 12(9):3311–3319
Singh PB, Pua HH, Happ HC, Schneider C, von Moltke J, Locksley RM, Baumjohann D, Ansel KM (2017) MicroRNA regulation of type 2 innate lymphoid cell homeostasis and function in allergic inflammation. J Exp Med 214(12):3627–3643. https://doi.org/10.1084/jem.20170545
Hogg N, Patzak I, Willenbrock F (2011) The insider’s guide to leukocyte integrin signalling and function. Nat Rev Immunol 11(6):416–426. https://doi.org/10.1038/nri2986
Lei AH, Xiao Q, Liu GY, Shi K, Yang Q, Li X, Liu YF, Wang HK, Cai WP, Guan YJ, Gabrilovich DI, Zhou J (2018) ICAM-1 controls development and function of ILC2. J Exp Med 215(8):2157–2174. https://doi.org/10.1084/jem.20172359
Hubbard AK, Rothlein R (2000) Intercellular adhesion molecule-1 (ICAM-1) expression and cell signaling cascades. Free Radic Biol Med 28(9):1379–1386. https://doi.org/10.1016/s0891-5849(00)00223-9
Dragoni S, Hudson N, Kenny BA, Burgoyne T, McKenzie JA, Gill Y, Blaber R, Futter CE, Adamson P, Greenwood J, Turowski P (2017) Endothelial MAPKs direct ICAM-1 signaling to divergent inflammatory functions. J Immunol 198(10):4074–4085. https://doi.org/10.4049/jimmunol.1600823
Hoyler T, Klose CS, Souabni A, Turqueti-Neves A, Pfeifer D, Rawlins EL, Voehringer D, Busslinger M, Diefenbach A (2012) The transcription factor GATA-3 controls cell fate and maintenance of type 2 innate lymphoid cells. Immunity 37(4):634–648. https://doi.org/10.1016/j.immuni.2012.06.020
Zhong C, Zhu J (2017) Transcriptional regulators dictate innate lymphoid cell fates. Protein Cell 8(4):242–254. https://doi.org/10.1007/s13238-017-0369-7
Maneechotesuwan K, Xin Y, Ito K, Jazrawi E, Lee KY, Usmani OS, Barnes PJ, Adcock IM (2007) Regulation of Th2 cytokine genes by p38 MAPK-mediated phosphorylation of GATA-3. J Immunol 178(4):2491–2498. https://doi.org/10.4049/jimmunol.178.4.2491
Yao MY, Zhang WH, Ma WT, Liu QH, Xing LH, Zhao GF (2020) Long non-coding RNA MALAT1 exacerbates acute respiratory distress syndrome by upregulating ICAM-1 expression via microRNA-150-5p downregulation. Aging (Albany NY) 12(8):6570–6585. https://doi.org/10.18632/aging.102953
Hoyte FCL, Nelson HS (2018) Recent advances in allergic rhinitis. F1000Res. https://doi.org/10.12688/f1000research.15367.1
Gurram RK, Zhu J (2019) Orchestration between ILC2s and Th2 cells in shaping type 2 immune responses. Cell Mol Immunol 16(3):225–235. https://doi.org/10.1038/s41423-019-0210-8
Ho J, Bailey M, Zaunders J, Mrad N, Sacks R, Sewell W, Harvey RJ (2015) Group 2 innate lymphoid cells (ILC2s) are increased in chronic rhinosinusitis with nasal polyps or eosinophilia. Clin Exp Allergy 45(2):394–403. https://doi.org/10.1111/cea.12462
Bartemes KR, Kephart GM, Fox SJ, Kita H (2014) Enhanced innate type 2 immune response in peripheral blood from patients with asthma. J Allergy Clin Immunol 134(3):671-678.e674. https://doi.org/10.1016/j.jaci.2014.06.024
Doherty TA, Scott D, Walford HH, Khorram N, Lund S, Baum R, Chang J, Rosenthal P, Beppu A, Miller M, Broide DH (2014) Allergen challenge in allergic rhinitis rapidly induces increased peripheral blood type 2 innate lymphoid cells that express CD84. J Allergy Clin Immunol 133(4):1203–1205. https://doi.org/10.1016/j.jaci.2013.12.1086
Dhariwal J, Cameron A, Trujillo-Torralbo MB, Del Rosario A, Bakhsoliani E, Paulsen M, Jackson DJ, Edwards MR, Rana BMJ, Cousins DJ, Hansel TT, Johnston SL, Walton RP, Consortium M-GSA (2017) Mucosal type 2 innate lymphoid cells are a key component of the allergic response to aeroallergens. Am J Respir Crit Care Med 195(12):1586–1596. https://doi.org/10.1164/rccm.201609-1846OC
Kato Y, Akasaki S, Muto-Haenuki Y, Fujieda S, Matsushita K, Yoshimoto T (2014) Nasal sensitization with ragweed pollen induces local-allergic-rhinitis-like symptoms in mice. PLoS ONE 9(8):e103540. https://doi.org/10.1371/journal.pone.0103540
Deshpande DA, Dileepan M, Walseth TF, Subramanian S, Kannan MS (2015) MicroRNA regulation of airway inflammation and airway smooth muscle function: relevance to asthma. Drug Dev Res 76(6):286–295. https://doi.org/10.1002/ddr.21267
Pua HH, Ansel KM (2015) MicroRNA regulation of allergic inflammation and asthma. Curr Opin Immunol 36:101–108. https://doi.org/10.1016/j.coi.2015.07.006
Zhu YQ, Liao B, Liu YH, Wang Z, Zhu XH, Chen XB, Wang MQ (2019) MicroRNA-155 plays critical effects on Th2 factors expression and allergic inflammatory response in type-2 innate lymphoid cells in allergic rhinitis. Eur Rev Med Pharmacol Sci 23(10):4097–4109. https://doi.org/10.26355/eurrev_201905_17911
Wang J, Cui Z, Liu L, Zhang S, Zhang Y, Zhang Y, Su H, Zhao Y (2019) MiR-146a mimic attenuates murine allergic rhinitis by downregulating TLR4/TRAF6/NF-kappaB pathway. Immunotherapy 11(13):1095–1105. https://doi.org/10.2217/imt-2019-0047
Dustin ML, Bivona TG, Philips MR (2004) Membranes as messengers in T cell adhesion signaling. Nat Immunol 5(4):363–372. https://doi.org/10.1038/ni1057
Mukhopadhyay S, Malik P, Arora SK, Mukherjee TK (2014) Intercellular adhesion molecule-1 as a drug target in asthma and rhinitis. Respirology 19(4):508–513. https://doi.org/10.1111/resp.12285
Furusho S, Myou S, Fujimura M, Kita T, Yasui M, Kasahara K, Nakao S, Takehara K, Sato S (2006) Role of intercellular adhesion molecule-1 in a murine model of toluene diisocyanate-induced asthma. Clin Exp Allergy 36(10):1294–1302. https://doi.org/10.1111/j.1365-2222.2006.02568.x
Steiner O, Coisne C, Cecchelli R, Boscacci R, Deutsch U, Engelhardt B, Lyck R (2010) Differential roles for endothelial ICAM-1, ICAM-2, and VCAM-1 in shear-resistant T cell arrest, polarization, and directed crawling on blood-brain barrier endothelium. J Immunol 185(8):4846–4855. https://doi.org/10.4049/jimmunol.0903732
Zhu J (2017) GATA3 regulates the development and functions of innate lymphoid cell subsets at multiple stages. Front Immunol 8:1571. https://doi.org/10.3389/fimmu.2017.01571
Yagi R, Zhong C, Northrup DL, Yu F, Bouladoux N, Spencer S, Hu G, Barron L, Sharma S, Nakayama T, Belkaid Y, Zhao K, Zhu J (2014) The transcription factor GATA3 is critical for the development of all IL-7Ralpha-expressing innate lymphoid cells. Immunity 40(3):378–388. https://doi.org/10.1016/j.immuni.2014.01.012
Furusawa J, Moro K, Motomura Y, Okamoto K, Zhu J, Takayanagi H, Kubo M, Koyasu S (2013) Critical role of p38 and GATA3 in natural helper cell function. J Immunol 191(4):1818–1826. https://doi.org/10.4049/jimmunol.1300379
Chen CH, Zhang DH, LaPorte JM, Ray A (2000) Cyclic AMP activates p38 mitogen-activated protein kinase in Th2 cells: phosphorylation of GATA-3 and stimulation of Th2 cytokine gene expression. J Immunol 165(10):5597–5605. https://doi.org/10.4049/jimmunol.165.10.5597
Funding
This work was supported by the National Natural Science Foundation of China (Grant Number 8197150744).
Author information
Authors and Affiliations
Contributions
All authors gave final approval of the version to be published and agreed to be accountable for all aspects of this work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no commercial or financial conflict of interest.
Ethical approval
The study was approved by the Ethics Committee of The Affiliated Hospital of Shandong University of Traditional Chinese Medicine and obeyed the principles of the Declaration of Helsinki. The study protocol was approved by the Institutional Animal Ethics Committee of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine. The methodologies of this research were carried out following the National Institutes of Health guide for the care and use of laboratory animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, L., Meng, W., Chen, X. et al. MiR-150-5p regulates the functions of type 2 innate lymphoid cells via the ICAM-1/p38 MAPK axis in allergic rhinitis. Mol Cell Biochem 477, 1009–1022 (2022). https://doi.org/10.1007/s11010-021-04346-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-021-04346-4