Abstract
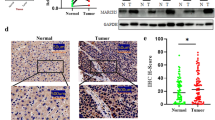
Reprogramming of energy metabolism is a hallmark of cancer which is prevalent worldwide. Octamer transcription factor-1 (OCT1) is a well-known transcription factor. However, the role of OCT1 in metabolism remodeling has not been well defined. In the present study, we found that OCT1 was up-regulated in non-small cell lung cancer (NSCLC) and correlated with poor patient survival. Further data identified that OCT1 increased glycolysis flux, promoting proliferation in lung cancer cells. Mechanistically, OCT1 facilitated the aerobic glycolysis and cell proliferation via up-regulation of hexokinase 2 (HK2), a crucial enzyme of the Warburg effect. Hence, our findings indicate that, in NSCLC, high levels of OCT1 contribute to the Warburg effect through up-regulation of HK2, linking up the OCT1/HK2 axis and cancer progression, which provide a potential biomarker and therapeutic target for NSCLC treatment.





Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Warburg O (1956) On the origin of cancer cells. Science 123:309–314. https://doi.org/10.1126/science.123.3191.309
Warburg O (1956) On respiratory impairment in cancer cells. Science 124:269–270
Vander Heiden MG, Cantley LC, Thompson CB (2009) Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324:1029–1033. https://doi.org/10.1126/science.1160809
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. https://doi.org/10.1016/j.cell.2011.02.013
Vander Heiden MG (2011) Targeting cancer metabolism: a therapeutic window opens. Nat Rev Drug Discov 10:671–684. https://doi.org/10.1038/nrd3504
Galluzzi L, Kepp O, Vander Heiden MG, Kroemer G (2013) Metabolic targets for cancer therapy. Nat Rev Drug Discov 12:829–846. https://doi.org/10.1038/nrd4145
Yeung SJ, Pan J, Lee MH (2008) Roles of p53, MYC and HIF-1 in regulating glycolysis—the seventh hallmark of cancer. Cell Mol Life Sci 65:3981–3999. https://doi.org/10.1007/s00018-008-8224-x
DeBerardinis RJ, Lum JJ, Hatzivassiliou G, Thompson CB (2008) The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab 7:11–20. https://doi.org/10.1016/j.cmet.2007.10.002
Dejure FR, Eilers M (2017) MYC and tumor metabolism: chicken and egg. EMBO J 36:3409–3420. https://doi.org/10.15252/embj.201796438
Herr W, Sturm RA, Clerc RG, Corcoran LM, Baltimore D, Sharp PA, Ingraham HA, Rosenfeld MG, Finney M, Ruvkun G et al (1988) The POU domain: a large conserved region in the mammalian pit-1, oct-1, oct-2, and Caenorhabditis elegans unc-86 gene products. Genes Dev 2:1513–1516. https://doi.org/10.1101/gad.2.12a.1513
Ryan AK, Rosenfeld MG (1997) POU domain family values: flexibility, partnerships, and developmental codes. Genes Dev 11:1207–1225. https://doi.org/10.1101/gad.11.10.1207
Kang J, Gemberling M, Nakamura M, Whitby FG, Handa H, Fairbrother WG, Tantin D (2009) A general mechanism for transcription regulation by Oct1 and Oct4 in response to genotoxic and oxidative stress. Genes Dev 23:208–222. https://doi.org/10.1101/gad.1750709
Kang J, Shakya A, Tantin D (2009) Stem cells, stress, metabolism and cancer: a drama in two Octs. Trends Biochem Sci 34:491–499. https://doi.org/10.1016/j.tibs.2009.06.003
Maddox J, Shakya A, South S, Shelton D, Andersen JN, Chidester S, Kang J, Gligorich KM, Jones DA, Spangrude GJ, Welm BE, Tantin D (2012) Transcription factor Oct1 is a somatic and cancer stem cell determinant. PLoS Genet 8:e1003048. https://doi.org/10.1371/journal.pgen.1003048
Vázquez-Arreguín K, Tantin D (2016) The Oct1 transcription factor and epithelial malignancies: old protein learns new tricks. Biochim Biophys Acta 1859:792–804. https://doi.org/10.1016/j.bbagrm.2016.02.007
Obinata D, Takayama K, Urano T, Murata T, Kumagai J, Fujimura T, Ikeda K, Horie-Inoue K, Homma Y, Ouchi Y, Takahashi S, Inoue S (2012) Oct1 regulates cell growth of LNCaP cells and is a prognostic factor for prostate cancer. Int J Cancer 130:1021–1028. https://doi.org/10.1002/ijc.26043
Jeong SH, Lee YJ, Cho BI, Ha WS, Choi SK, Jung EJ, Ju YT, Jeong CY, Ko GH, Yoo J, Hong SC (2014) OCT-1 overexpression is associated with poor prognosis in patients with well-differentiated gastric cancer. Tumour Biol 35:5501–5509. https://doi.org/10.1007/s13277-014-1724-4
Vázquez-Arreguín K, Maddox J, Kang J, Park D, Cano RR, Factor RE, Ludwig T, Tantin D (2018) BRCA1 through its E3 ligase activity regulates the transcription factor Oct1 and carbohydrate metabolism. Mol Cancer Res 16:439–452. https://doi.org/10.1158/1541-7786.Mcr-17-0364
Vázquez-Arreguín K, Bensard C, Schell JC, Swanson E, Chen X, Rutter J, Tantin D (2019) Oct1/Pou2f1 is selectively required for colon regeneration and regulates colon malignancy. PLoS Genet 15:e1007687. https://doi.org/10.1371/journal.pgen.1007687
Shakya A, Cooksey R, Cox JE, Wang V, McClain DA, Tantin D (2009) Oct1 loss of function induces a coordinate metabolic shift that opposes tumorigenicity. Nat Cell Biol 11:320–327. https://doi.org/10.1038/ncb1840
Qi Q, Liu X, Li S, Joshi HC, Ye K (2013) Synergistic suppression of noscapine and conventional chemotherapeutics on human glioblastoma cell growth. Acta Pharmacol Sin 34:930–938. https://doi.org/10.1038/aps.2013.40
D’Addario G, Früh M, Reck M, Baumann P, Klepetko W, Felip E (2010) Metastatic non-small-cell lung cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 21(Suppl 5):v116–v119. https://doi.org/10.1093/annonc/mdq189
Tantin D, Schild-Poulter C, Wang V, Haché RJ, Sharp PA (2005) The octamer binding transcription factor Oct-1 is a stress sensor. Cancer Res 65:10750–10758. https://doi.org/10.1158/0008-5472.Can-05-2399
Li L, Liang Y, Kang L, Liu Y, Gao S, Chen S, Li Y, You W, Dong Q, Hong T, Yan Z, Jin S, Wang T, Zhao W, Mai H, Huang J, Han X, Ji Q, Song Q, Yang C, Zhao S, Xu X, Ye Q (2018) Transcriptional regulation of the Warburg effect in cancer by SIX1. Cancer Cell 33:368-385.e7. https://doi.org/10.1016/j.ccell.2018.01.010
Koppenol WH, Bounds PL, Dang CV (2011) Otto Warburg’s contributions to current concepts of cancer metabolism. Nat Rev Cancer 11:325–337. https://doi.org/10.1038/nrc3038
Patra KC, Hay N (2013) Hexokinase 2 as oncotarget. Oncotarget 4:1862–1863. https://doi.org/10.18632/oncotarget.1563
Hay N (2016) Reprogramming glucose metabolism in cancer: can it be exploited for cancer therapy? Nat Rev Cancer 16:635–649. https://doi.org/10.1038/nrc.2016.77
Patra KC, Wang Q, Bhaskar PT, Miller L, Wang Z, Wheaton W, Chandel N, Laakso M, Muller WJ, Allen EL, Jha AK, Smolen GA, Clasquin MF, Robey B, Hay N (2013) Hexokinase 2 is required for tumor initiation and maintenance and its systemic deletion is therapeutic in mouse models of cancer. Cancer Cell 24:213–228. https://doi.org/10.1016/j.ccr.2013.06.014
Roberts DJ, Miyamoto S (2015) Hexokinase II integrates energy metabolism and cellular protection: akting on mitochondria and TORCing to autophagy. Cell Death Differ 22:248–257. https://doi.org/10.1038/cdd.2014.173
DeWaal D, Nogueira V, Terry AR, Patra KC, Jeon SM, Guzman G, Au J, Long CP, Antoniewicz MR, Hay N (2018) Hexokinase-2 depletion inhibits glycolysis and induces oxidative phosphorylation in hepatocellular carcinoma and sensitizes to metformin. Nat Commun 9:446. https://doi.org/10.1038/s41467-017-02733-4
Heikkinen S, Pietila M, Halmekyto M, Suppola S, Pirinen E, Deeb SS, Janne J, Laakso M (1999) Hexokinase II-deficient mice. Prenatal death of homozygotes without disturbances in glucose tolerance in heterozygotes. J Biol Chem 274:22517–22523. https://doi.org/10.1074/jbc.274.32.22517
Acknowledgements
The authors are grateful to Dr. Bin Lv (Wenzhou Medical University) for providing clinical lung samples. This research was funded by National Natural Science Foundation of China (81973341, 81902826), Science and Technology Program of Guangzhou (202002030010), and the Fundamental Research Funds for the Central Universities (21620426).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was carried out in accordance with the recommendations of Requirements of the Ethical Review System of Biomedical Research Involving Human by National Health and Family Planning Commission of China, Jinan University and Wenzhou Medical University Ethics Committee with written informed consent from all subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, Z., Su, J., Sun, M. et al. Octamer transcription factor-1 induces the Warburg effect via up-regulation of hexokinase 2 in non-small cell lung cancer. Mol Cell Biochem 476, 3423–3431 (2021). https://doi.org/10.1007/s11010-021-04171-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-021-04171-9