Abstract
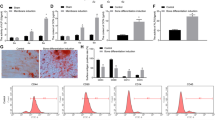
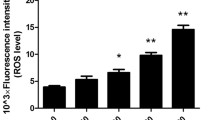
MiR-9 has been found to be involved in the repair of spinal cord injury and regulates the proliferation and differentiation of mesenchymal stem cells. However, the role of miR-9 in repair of bone defects has not been well studied. The current study was designed to investigate its role and potential underlying mechanism in regulating osteoblast differentiation and angiogenesis. After treating the murine pre-osteoblast cell line MC3T3-E1 with BMP2, miR-9 expression was obviously down-regulated. Following transfection with miR-9 mimics, its overexpression enhanced the differentiation of MC3T3-E1 cells into osteoblasts as evidence that miR-9 up-regulated the mRNA levels of osteoblast differentiation-related protein, as well as increased differentiation and mineralization of osteoblasts. Further functional analysis has shown that miR-9 overexpression effectively increased human umbilical vein endothelial cell proliferation. Moreover, miR-9 up-regulation promoted cell migration, VEGF, and VE-cadherin concentrations, as well as tube formation in vitro. The mechanistic assay demonstrated that overexpression of miR-9-induced activation of the AMPK signaling pathway. Taken together, our findings suggested that miR-9 overexpression promoted osteoblast differentiation and angiogenesis via the AMPK signaling pathway, representing a novel and potential therapeutic target for the treatment of bone injury-related diseases.





Similar content being viewed by others
Abbreviations
- BMP2:
-
Bone morphogenetic protein 2
- HUVECs:
-
Human umbilical vein endothelial cells
- VEGF:
-
Vascular endothelial growth factor
- VE-cadherin:
-
Vascular endothelial cadherin
- AMPK:
-
Adenosine 5′-monophosphate (AMP)-activated protein kinase
- iPS:
-
Induced pluripotent stem cells
- MSC:
-
Mesenchymal stem cell
- aMEM:
-
a-Minimal essential medium
- FBS:
-
Fetal bovine serum
- ALP:
-
Alkaline phosphatase
- ARS:
-
Alizarin red staining
- OC:
-
Osteocalcin
- Runx2:
-
Runt-related transcription factor 2
- OSX:
-
Osterix
- p21:
-
Cyclin-dependent kinase inhibitor 1
References
Dong S, Yang B, Guo H, Kang F (2012) MicroRNAs regulate osteogenesis and chondrogenesis. Biochem Biophys Res Commun 418:587–591
Long F, Ornitz DM (2013) Development of the endochondral skeleton. CSH Perspect Biol 5:a008334
Hu N, Jiang D, Huang E, Liu X, Li R, Liang X, Kim SH, Chen X, Gao J-L, Zhang H (2013) BMP9-regulated angiogenic signaling plays an important role in the osteogenic differentiation of mesenchymal progenitor cells. J Cell Sci 126:532–541
Wei J, Shi Y, Zheng L, Zhou B, Inose H, Wang J, Guo XE, Grosschedl R, Karsenty G (2012) miR-34 s inhibit osteoblast proliferation and differentiation in the mouse by targeting SATB2. J Cell Biol 197:509–521
Portal-Nunez S, Lozano D, Esbrit P (2012) Role of angiogenesis on bone formation. Histol Histopathol 27:559–566
Kular J, Tickner J, Chim SM, Xu J (2012) An overview of the regulation of bone remodelling at the cellular level. Clin Biochem 45:863–873
Dirckx N, Hul M, Maes C (2013) Osteoblast recruitment to sites of bone formation in skeletal development, homeostasis, and regeneration. Birth Defects Res C 99:170–191
Chen G, Deng C, Li Y-P (2012) TGF-β and BMP signaling in osteoblast differentiation and bone formation. Int J Biol Sci 8:272
Taipaleenmäki H, Hokland LB, Chen L, Kauppinen S, Kassem M (2012) Mechanisms in endocrinology: micro-RNAs: targets for enhancing osteoblast differentiation and bone formation. Eur J Endocrinol 166:359–371
Izu Y, Sun M, Zwolanek D, Veit G, Williams V, Cha B, Jepsen KJ, Koch M, Birk DE (2011) Type XII collagen regulates osteoblast polarity and communication during bone formation. J Cell Biol 193:1115–1130
Thompson WR, Rubin CT, Rubin J (2012) Mechanical regulation of signaling pathways in bone. Gene 503:179–193
Inoki K, Kim J, Guan K-L (2012) AMPK and mTOR in cellular energy homeostasis and drug targets. Annu Rev Pharmacol 52:381–400
Palacios OM, Carmona JJ, Michan S, Chen KY, Manabe Y, Ward Iii JL, Goodyear LJ, Tong Q (2009) Diet and exercise signals regulate SIRT3 and activate AMPK and PGC-1α in skeletal muscle. Aging (Albany NY) 1:771
Hardie DG, Sakamoto K (2006) AMPK: a key sensor of fuel and energy status in skeletal muscle. Physiology 21:48–60
Chang JT, Wherry EJ, Goldrath AW (2014) Molecular regulation of effector and memory T cell differentiation. Nat Immunol 15:1104–1115
Thompson AM, Martin KA, Rzucidlo EM (2014) Resveratrol induces vascular smooth muscle cell differentiation through stimulation of SirT1 and AMPK. PLoS One 9:e85495
Huntzinger E, Izaurralde E (2011) Gene silencing by microRNAs: contributions of translational repression and mRNA decay. Nat Rev Genet 12:99–110
Poltronieri P, D’Urso PI, Mezzolla V, D’Urso OF (2013) Potential of anti-cancer therapy based on anti-miR-155 oligonucleotides in glioma and brain tumours. Chem Biol Drug Des 81:79–84
Boese AS, Majer A, Saba R, Booth SA (2013) Small RNA drugs for prion disease: a new frontier. Expert Opin Drug Discov 8:1265–1284
Okamoto H, Matsumi Y, Hoshikawa Y, Takubo K, Ryoke K, Shiota G (2012) Involvement of microRNAs in regulation of osteoblastic differentiation in mouse induced pluripotent stem cells. PLoS One 7:e43800
Suomi S, Taipaleenmaki H, Seppanen A, Ripatti T, Vaananen K, Hentunen T, Saamanen AM, Laitala-Leinonen T (2008) MicroRNAs regulate osteogenesis and chondrogenesis of mouse bone marrow stromal cells. Gene Regul Syst Bio 2:177–191
Guo L, Zhao RC, Wu Y (2011) The role of microRNAs in self-renewal and differentiation of mesenchymal stem cells. Exp Hematol 39:608–616
Hermeking H (2012) MicroRNAs in the p53 network: micromanagement of tumour suppression. Nat Rev Cancer 12:613–626
Iorio MV, Croce CM (2012) microRNA involvement in human cancer. Carcinogenesis 33:1126–1133
Genda Y, Arai M, Ishikawa M, Tanaka S, Okabe T, Sakamoto A (2013) microRNA changes in the dorsal horn of the spinal cord of rats with chronic constriction injury: a TaqMan® Low Density Array study. Int J Mol Med 31:129–137
Wu D, Raafat A, Pak E, Clemens S, Murashov AK (2012) Dicer-microRNA pathway is critical for peripheral nerve regeneration and functional recovery in vivo and regenerative axonogenesis in vitro. Exp Neurol 233:555–565
Han R, Kan Q, Sun Y, Wang S, Zhang G, Peng T, Jia Y (2012) MiR-9 promotes the neural differentiation of mouse bone marrow mesenchymal stem cells via targeting zinc finger protein 521. Neurosci Lett 515:147–152
Li H, Xie H, Liu W, Hu R, Huang B, Tan Y-F, Liao E-Y, Xu K, Sheng Z-F, Zhou H-D (2009) A novel microRNA targeting HDAC5 regulates osteoblast differentiation in mice and contributes to primary osteoporosis in humans. J Clin Investig 119:3666
Andris F, Leo O (2015) AMPK in lymphocyte metabolism and function. Int Rev Immunol 34:67–81
Li Y, Peng T, Li L, Wang X, Duan R, Gao H, Guan W, Lu J, Teng J, Jia Y (2014) MicroRNA-9 regulates neural apoptosis in methylmalonic acidemia via targeting BCL2L11. Int J Dev Neurosci 36:19–24
Brandenburger T, Castoldi M, Brendel M, Grievink H, Schlosser L, Werdehausen R, Bauer I, Hermanns H (2012) Expression of spinal cord microRNAs in a rat model of chronic neuropathic pain. Neurosci Lett 506:281–286
Zhuang G, Wu X, Jiang Z, Kasman I, Yao J, Guan Y, Oeh J, Modrusan Z, Bais C, Sampath D, Ferrara N (2012) Tumour-secreted miR-9 promotes endothelial cell migration and angiogenesis by activating the JAK-STAT pathway. EMBO J 31:3513–3523
Chevallier N, Anagnostou F, Zilber S, Bodivit G, Maurin S, Barrault A, Bierling P, Hernigou P, Layrolle P, Rouard H (2010) Osteoblastic differentiation of human mesenchymal stem cells with platelet lysate. Biomaterials 31:270–278
Singh A, Ali S, Mahdi AA, Srivastava RN (2012) MicroRNAs and their role in bone remodeling and pathogenesis. Br J Med Res 2:727–749
Carmeliet P, Lampugnani MG, Moons L, Breviario F, Compernolle V, Bono F, Balconi G, Spagnuolo R, Oosthuyse B, Dewerchin M, Zanetti A, Angellilo A, Mattot V, Nuyens D, Lutgens E, Clotman F, de Ruiter MC, Gittenberger-de Groot A, Poelmann R, Lupu F, Herbert JM, Collen D, Dejana E (1999) Targeted deficiency or cytosolic truncation of the VE-cadherin gene in mice impairs VEGF-mediated endothelial survival and angiogenesis. Cell 98:147–157
Kunimasa K, Ahn M-R, Kobayashi T, Eguchi R, Kumazawa S, Fujimori Y, Nakano T, Nakayama T, Kaji K and Ohta T (2011) Brazilian propolis suppresses angiogenesis by inducing apoptosis in tube-forming endothelial cells through inactivation of survival signal ERK1/2. Evid Based Complement Altern Med 2011. doi:10.1093/ecam/nep024
Zhao X, Zmijewski JW, Lorne E, Liu G, Park Y-J, Tsuruta Y, Abraham E (2008) Activation of AMPK attenuates neutrophil proinflammatory activity and decreases the severity of acute lung injury. Am J Physiol-Lung C 295:L497–L504
Kanazawa I, Yamaguchi T, Yano S, Yamauchi M, Sugimoto T (2008) Metformin enhances the differentiation and mineralization of osteoblastic MC3T3-E1 cells via AMP kinase activation as well as eNOS and BMP-2 expression. Biochem Biophys Res Commun 375:414–419
Nagata D, Mogi M, Walsh K (2003) AMP-activated protein kinase (AMPK) signaling in endothelial cells is essential for angiogenesis in response to hypoxic stress. J Biol Chem 278:31000–31006
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Jining Qu and Daigang Lu have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Qu, J., Lu, D., Guo, H. et al. MicroRNA-9 regulates osteoblast differentiation and angiogenesis via the AMPK signaling pathway. Mol Cell Biochem 411, 23–33 (2016). https://doi.org/10.1007/s11010-015-2565-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2565-1