Abstract
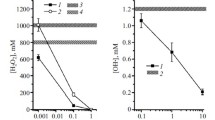
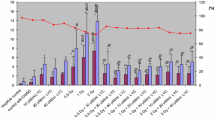
The aim of this study was to investigate protective effects of the lipophilic vitamin C derivative, 6-o-palmitoylascorbate (PlmtVC), against X-ray radiation-induced damages including cell death, DNA double-strand breaks (DSBs), lipid peroxidation, and protein carbonylation in human lymphocytes HEV0082, and the stability of PlmtVC under cell-cultured or cell-free condition. Irradiation with X-ray (1.5 Gy) diminished the cell viability and induced apoptosis, both of which were protected by pre-irradiational administration with PlmtVC. Gamma-H2A.X foci as a hallmark of DSBs were markedly enhanced in the irradiated cells. PlmtVC prevented X-ray-induced DSBs more appreciably than l-ascorbic acid (l-AA). Intracellular ROS production, lipid peroxidation, and protein carbonylation in HEV0082 cells were increased by X-ray at 1.5 Gy, all of which were significantly repressed by PlmtVC. PlmtVC also elevated endogenous reduced glutathione (GSH) in HEV0082 cells, and prevented X-ray-induced GSH depletion that are more appreciably over l-AA. Thus, PlmtVC prevents X-ray-induced cell death through its antioxidative activity. Stability tests showed that after being kept under physiological conditions (pH 7.4, 37 °C) for 14 days, vitamin C residual rates in PlmtVC solutions (62.2–82.0 %) were significantly higher than those in l-AA solutions (20.5–28.7 %). When PlmtVC or l-AA was added to HEV0082 lymphocytes, intracellular vitamin C in l-AA-treated cells was not detectable after 24 h, whereas PlmtVC-treated cells could keep a high level of intracellular vitamin C, suggesting an excellent stability of PlmtVC. Thus, X-ray-induced diverse harmful effects could be prevented by PlmtVC, which was suggested to ensue intrinsically from the persistent enrichment of intracellular vitamin C, resulting in relief to X-ray-caused oxidative stress.











Similar content being viewed by others
References
Tusher VG, Tibshirani R, Chu G (2001) Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA 98:5116–5121
Varshney R, Kale RK (1990) Effects of calmodulin antagonists on radiation-induced lipid peroxidation in microsomes. Int J Radiat Biol 58:733–743
Krisko A, Radman M (2010) Protein damage and death by radiation in Escherichia coli and Deinococcus radiodurans. Proc Natl Acad Sci USA 107:14373–14377. doi:10.1073/pnas.1009312107
Sutherland BM, Bennett PV, Sidorkina O, Laval J (2000) Clustered DNA damages induced in isolated DNA and in human cells by low doses of ionizing radiation. Proc Natl Acad Sci USA 97:103–108
Sokolov M, Neumann R (2013) Lessons learned about human stem cell responses to ionizing radiation exposures: a long road still ahead of us. Int J Mol Sci 14:15695–15723. doi:10.3390/ijms140815695
Thornalley PJ, Vasák M (1985) Possible role for metallothionein in protection against radiation-induced oxidative stress. Kinetics and mechanism of its reaction with superoxide and hydroxyl radicals. Biochim Biophys Acta 827:36–44
Ward JF, Blakely WF, Joner EI (1985) Mammalian cells are not killed by DNA single-strand breaks caused by hydroxyl radicals from hydrogen peroxide. Radiat Res 103:383–392
Cadet J, Douki T, Ravanat JL (2010) Oxidatively generated base damage to cellular DNA. Free Radic Biol Med 49:9–21. doi:10.1016/j.freeradbiomed.2010.03.025
Manda K, Ueno M, Anzai K (2007) AFMK, a melatonin metabolite, attenuates X-ray-induced oxidative damage to DNA, proteins and lipids in mice. J Pineal Res 42:386–393
Corre I, Niaudet C, Paris F (2010) Plasma membrane signaling induced by ionizing radiation. Mutat Res 704:61–67. doi:10.1016/j.mrrev.2010.01.014
Daly MJ, Gaidamakova EK, Matrosova VY, Vasilenko A, Zhai M, Leapman M, Lai B, Ravel B, Li SM, Kemner KM, Fredrickson JK (2007) Protein oxidation implicated as the primary determinant of bacterial radioresistance. PLoS Biol 5:e92. doi:10.1371/journal.pbio.0050092
Haro KJ, Sheth A, Scheinberg DA (2012) Dysregulation of IRP1-mediated iron metabolism causes gamma ray-specific radioresistance in leukemia cells. PLoS ONE 7:e48841. doi:10.1371/journal.pone.0048841
Smutná M, Beňová K, Dvořák P, Nekvapil T, Kopřiva V, Maté D (2013) Protein carbonyls and traditional biomarkers in pigs exposed to low-dose γ-radiation. Res Vet Sci 94:214–218. doi:10.1016/j.rvsc.2012.09.002
Wright EG (2000) Inducible genomic instability: new insights into the biological effects of ionizing radiation. Med Confl Surviv 16:117–130
Vignard J, Mirey G, Salles B (2013) Ionizing-radiation induced DNA double-strand breaks: a direct and indirect lighting up. Radiother Oncol 108:362–369. doi:10.1016/j.radonc.2013.06.013
Lomax ME, Folkes LK, O’Neill P (2013) Biological consequences of radiation-induced DNA damage: relevance to radiotherapy. Clin Oncol 25:578–585. doi:10.1016/j.clon.2013.06.007
Prasad NR, Menon VP, Vasudev V, Pugalendi KV (2005) Radioprotective effect of sesamol on gamma-radiation induced DNA damage, lipid peroxidation and antioxidants levels in cultured human lymphocytes. Toxicology 209:225–235
Buonanno M, de Toledo SM, Pain D, Azzam EI (2011) Long-term consequences of radiation-induced bystander effects depend on radiation quality and dose and correlate with oxidative stress. Radiat Res 175:405–415. doi:10.1667/RR2461.1
Chatterjee A (2013) Reduced glutathione: a radioprotector or a modulator of DNA-repair activity? Nutrients 5:525–542
Krisko A, Leroy M, Radman M, Meselson M (2012) Extreme anti-oxidant protection against ionizing radiation in bdelloid rotifers. Proc Natl Acad Sci USA 109:2354–2357. doi:10.1073/pnas.1119762109
Vorotnikova E, Tries M, Braunhut S (2004) Retinoids and TIMP1 prevent radiation-induced apoptosis of capillary endothelial cells. Radiat Res 161:174–184
Singh VK, Wise SY, Singh PK, Posarac A, Fatanmi OO, Ducey EJ, Bolduc DL, Elliott TB, Seed TM (2013) Alpha-tocopherol succinate-mobilized progenitors improve intestinal integrity after whole body irradiation. Int J Radiat Biol 89:334–345. doi:10.3109/09553002.2013.762137
Rahman K (2007) Studies on free radicals, antioxidants, and co-factors. Clin Interv Aging 2:219–236
Sram RJ, Binkova B, Rossner P Jr (2012) Vitamin C for DNA damage prevention. Mutat Res 733:39–49. doi:10.1016/j.mrfmmm.2011.12.001
Fujii Y, Kato TA, Ueno A, Kubota N, Fujimori A, Okayasu R (2010) Ascorbic acid gives different protective effects in human cells exposed to X-rays and heavy ions. Mutat Res 699:58–61. doi:10.1016/j.mrgentox.2010.04.003
Hata K, Urushibara A, Yamashita S, Shikazono N, Yokoya A, Katsumura Y (2013) Chemical repair of base lesions, AP-sites, and strand breaks on plasmid DNA in dilute aqueous solution by ascorbic acid. Biochem Biophys Res Commun 434:341–345. doi:10.1016/j.bbrc.2013.03.075
Simpson GL, Ortwerth BJ (2000) The non-oxidative degradation of ascorbic acid at physiological conditions. Biochim Biophys Acta 1501:12–24
Miwa N, Yamazaki H (1986) Potentiated susceptibility of ascites tumor to acyl derivatives of ascorbate caused by balanced hydrophobicity in the molecule. Exp Cell Biol 54:245–249
May JM, Qu ZC, Cobb CE (1996) Accessibility and reactivity of ascorbate 6-palmitate bound to erythrocyte membranes. Free Radic Biol Med 21:471–480
Ross D, Mendiratta S, Qu ZC, Cobb CE, May JM (1999) Ascorbate 6-palmitate protects human erythrocytes from oxidative damage. Free Radic Biol Med 26:81–89
Iwakawa M, Goto M, Noda S, Sagara M, Yamada S, Yamamoto N, Kawakami Y, Matsui Y, Miyazawa Y, Yamazaki H, Tsuji H, Ohno T, Mizoe J, Tsujii H, Imai T (2005) DNA repair capacity measured by high throughput alkaline comet assays in EBV-transformed cell lines and peripheral blood cells from cancer patients and healthy volunteers. Mutat Res 588:1–6
Gey KF (1995) Ten-year retrospective on the antioxidant hypothesis of arteriosclerosis: threshold plasma levels of antioxidant micronutrients related to minimum cardiovascular risk. J Nutr Biochem 6:206–236
Srám RJ, Rössner P, Smerhovský Z (2004) Cytogenetic analysis and occupational health in the Czech Republic. Mutat Res 566:21–48
Mottola M, Wilke N, Benedini L, Oliveira RG, Fanani ML (2013) Ascorbyl palmitate interaction with phospholipid monolayers: electrostatic and rheological preponderancy. Biochim Biophys Acta 1828:2496–2505. doi:10.1016/j.bbamem.2013.06.016
Daly MJ (2009) A new perspective on radiation resistance based on Deinococcus radiodurans. Nat Rev Microbiol 7:237–245. doi:10.1038/nrmicro2073
Meves A, Stock SN, Beyerle A, Pittelkow MR, Peus D (2002) Vitamin C derivative ascorbyl palmitate promotes ultraviolet-B-induced lipid peroxidation and cytotoxicity in keratinocytes. J Invest Dermatol 119:1103–1108
Barker CL, Clothier RH (1997) Human keratinocyte cultures as models of cutaneous esterase activity. Toxicol In Vitro 11:637–640
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xiao, L., Tsutsui, T. & Miwa, N. The lipophilic vitamin C derivative, 6-o-palmitoylascorbate, protects human lymphocytes, preferentially over ascorbate, against X-ray-induced DNA damage, lipid peroxidation, and protein carbonylation. Mol Cell Biochem 394, 247–259 (2014). https://doi.org/10.1007/s11010-014-2101-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-2101-8