Abstract
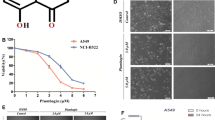
Thymoquinone (TQ) is the active ingredient extracted from the essential oil of Nigella sativa. A number of studies implicated TQ as an antitumor agent. In this study, cytotoxic effects of the oil of N. sativa and TQ were evaluated on human cervical cancer cell line, HeLa cells. IC50 value was ~0.125 μl/ml for N. sativa oil preparations and 12.5 μM for TQ. TQ strongly inhibited wound healing at all concentrations ranging from 12.5 to 100 μM in a scratch wound healing assay. Additionally, induction of apoptosis by TQ was assessed by Giemsa staining and TQ was found to induce apoptosis in cancer cells especially at concentrations of 50 and 100 μM. TQ-mediated transcriptional regulation of 84 genes involved in apoptosis was studied using a PCR array. At low dose (12.5 μM), TQ was found to induce expression of four pro-apoptotic genes: BIK (~22.7-fold), FASL (~2.9-fold), BCL2L10 (~2.1-fold), and CASP1 (~2-fold). TQ was also found to reduce the expression of an anti-apoptotic gene implicated in NF-kappa-B signaling and cancer: RELA (~8-fold). At high dose (100 μM), TQ mediated the expression of 21 genes implicated directly in apoptosis (6 genes), TNF signaling (10 genes), and NF-kappa-B signaling (3 genes) such as BIK, BID, TNFRSF10A, TNFRSF10B, TNF, TRAF3, RELA, and RELB. In conclusion, this study implicates the role of TQ in the inhibition of cancer cell proliferation and migration. At the same time, our results strongly suggest that TQ intervenes with TNF and NF-kappa-B signaling during TQ-mediated induction of apoptosis in cancer cells.





Similar content being viewed by others
References
Mann J (2002) Natural products in cancer chemotherapy: past, present and future. Nat Rev Cancer 2(2):143–148
El-Dakhakhany M (1963) Studies on the chemical constitution of Egyptian N. sativa L. seeds. Planta Med 11:465–470
Woo CC, Kumar AP, Sethi G, Tan KH (2012) Thymoquinone: potential cure for inflammatory disorders and cancer. Biochem Pharmacol 83(4):443–451
Banerjee S, Padhye S, Azmi A, Wang Z, Philip PA, Kucuk O et al (2010) Review on molecular and therapeutic potential of thymoquinone in cancer. Nutr Cancer 62:938–946
Badary OA, Al-Shabanah OA, Nagi MN, Al-Rikabi AC, Elmazar MM (1999) Inhibition of benzo(a)pyrene-induced forestomach carcinogenesis in mice by thymoquinone. Eur J Cancer Prev 8:435–440
Badary OA, Gamal El-Din AM (2001) Inhibitory effects of thymoquinone against 20-methylcholanthrene-induced fibrosarcoma tumorigenesis. Cancer Detect Prev 25:362–368
Gurung RL, Lim SN, Khaw AK, Soon JF, Shenoy K, Mohamed Ali S et al (2010) Thymoquinone induces telomere shortening, DNA damage and apoptosis in human glioblastoma cells. PLoS One 5:e12124
Woo CC, Loo SY, Veronica G, Yap CW, Sethi G, Kumar AP et al (2011) Anticancer activity of thymoquinone in breast cancer cells: possible involvement of PPARg pathway. Biochem Pharmacol 82:464–475
El-Mahdy MA, Zhu Q, Wang QE, Wani G, Wani AA (2005) Thymoquinone induces apoptosis through activation of caspase-8 and mitochondrial events in p53-null myoloblastic leukemia HL-60 cells. Int J Cancer 117:409–417
Alhosin M, Abusnina A, Achour M, Sharif T, Muller C, Peluso J et al (2010) Induction of apoptosis by thymoquinone in lymphoblastic leukemia Jurkat cells is mediated by a p73-dependent pathway which targets the epigenetic integrator UHRF1. Biochem Pharmacol 79:1251–1260
Jafri SH, Glass J, Shi R, Zhang S, Prince M, Kleiner-Hancock H (2010) Thymoquinone and cisplatin as a therapeutic combination in lung cancer: in vitro and in vivo. J Exp Clin Cancer Res 29:87
El-Najjar N, Chatila M, Moukadem H, Vuorela H, Ocker M, Gandesiri M et al (2010) Reactive oxygen species mediate thymoquinone-induced apoptosis and activate ERK and JNK signaling. Apoptosis 15:183–195
Banerjee S, Kaseb AO, Wang Z, Kong D, Mohammad M, Padhye S et al (2009) Antitumor activity of gemcitabine and oxaliplatin is augmented by thymoquinone in pancreatic cancer. Cancer Res 69:5575–5583
Roepke M, Diestel A, Bajbouj K, Walluscheck D, Schonfeld P, Roessner A et al (2007) Lack of p53 augments thymoquinone-induced apoptosis and caspase activation in human osteosarcoma cells. Cancer Biol Ther 6:160–169
Kaseb AO, Chinnakannu K, Chen D, Sivanandam A, Tejwani S, Menon M et al (2007) Androgen receptor and E2F-1 targeted thymoquinone therapy for hormone refractory prostate cancer. Cancer Res 67:7782–7788
Gali-Muhtasib H, Diab-Assaf M, Boltze C, Al-Hmaira J, Hartig R, Roessner A et al (2004) Thymoquinone extracted from black seed triggers apoptotic cell death in human colorectal cancer cells via a p53-dependent mechanism. Int J Oncol 25:857–866
Arafa el-SA, Zhu Q, Shah ZI, Wani G, Barakat BM, Racoma I, Racoma I et al (2011) Thymoquinone up-regulates PTEN expression and induces apoptosis in doxorubicin resistant human breast cancer cells. Mutat Res 706:28–35
Sethi G, Ahn KS, Aggarwal BB (2008) Targeting nuclear factor-kappa-B activation pathway by thymoquinone: role in suppression of antiapoptotic gene products and enhancement of apoptosis. Mol Cancer Res 6:1059–1070
Mohamed A, Afridi DM, Garani O, Tucci M (2005) Thymoquinone inhibits the activation of NF-kappa-B in the brain and spinal cord of experimental autoimmune encephalomyelitis. Biomed Sci Instrum 41:388–393
Connelly L, Barham W, Onishko HM, Sherrill T, Chodosh LA, Blackwell TS, Yull FE (2011) Inhibition of NF-kappa-B activity in mammary epithelium increases tumor latency and decreases tumor burden. Oncogene 30(12):1402–1412
Tekeoglu I, Dogan A, Ediz L, Budancamanak M, Demirel A (2007) Effects of thymoquinone (volatile oil of black cumin) on rheumatoid arthritis in rat models. Phytother Res 21(9):895–897
Chehl N, Chipitsyna G, Gong Q, Yeo CJ, Arafat HA (2009) Anti-inflammatory effects of the Nigella sativa seed extract, thymoquinone, in pancreatic cancer cells. HPB (Oxford) 11(5):373–381
Ravindran J, Nair HB, Sung B, Prasad S, Tekmal RR, Aggarwal BB (2010) Thymoquinone poly (lactide-co-glycolide) nanoparticles exhibit enhanced anti-proliferative, anti-inflammatory, and chemosensitization potential. Biochem Pharmacol 79(11):1640–1647
Boyd JM, Gallo GJ, Elangovan B, Houghton AB, Malstrom S, Avery BJ et al (1995) BIK, a novel death-inducing protein shares a distinct sequence motif with BCL-2 family proteins and interacts with viral and cellular survival-promoting proteins. Oncogene 11:1921–1928
Mathai JP, Germain M, Marcellus RC, Shore GC (2002) Induction and endoplasmic reticulum location of BIK/NBK in response to apoptotic signaling by E1A and p53. Oncogene 21:2534–2544
Tong Y, Yang Q, Vater C, Venkatesh LK, Custeau D, Chittenden T et al (2001) The pro-apoptotic protein, BIK, exhibits potent antitumor activity that is dependent on its BH3 domain. Mol Cancer Ther 1:95–102
Germain M, Mathai JP, Shore GC (2002) BH-3-only BIK functions at the endoplasmic reticulum to stimulate cytochrome c release from mitochondria. J Biol Chem 277:18053–18060
Gillissen B, Essmann F, Graupner V, Starck L, Radetzki S, Dorken B et al (2003) Induction of cell death by the BH3-only BCL-2 homolog NBK/BIK is mediated by an entirely BAX-dependent mitochondrial pathway. EMBO J 22:3580–3590
Theodorakis P, Lomonosova E, Chinnadurai G (2002) Critical requirement of BAX for manifestation of apoptosis induced by multiple stimuli in human epithelial cancer cells. Cancer Res 62:3373–3376
Guicciardi ME, Gores GJ (2009) Life and death by death receptors. FASEB J 23(6):1625–1637
Madrid LV, Mayo MW, Reuther JY, Baldwin AS Jr (2001) AKT stimulates the transactivation potential of the RelA/p65 Subunit of NF-kappa B through utilization of the Ikappa-B kinase and activation of the mitogen-activated protein kinase p38. J Biol Chem 276(22):18934–18940
Karin M, Cao Y, Greten FR, Li ZW (2002) NF-kappa-B in cancer: from innocent bystander to major culprit. Nat Rev Cancer 2(4):301–310
Pommier Y, Sordet O, Antony S, Hayward RL, Kohn KW (2004) Apoptosis defects and chemotherapy resistance: molecular interaction maps and networks. Oncogene 23(16):2934–2949
Levkau B, Scatena M, Giachelli CM, Ross R, Raines EW (1999) Apoptosis overrides survival signals through a caspase-mediated dominant-negative NF-kappa-B loop. Nat Cell Biol 1(4):227–233
Acknowledgments
We thank Dr. Nazmiye Bitgen for her technical support in Giemsa staining of cancer cells and assessment of apoptotic cells. This work was supported by Erciyes University Scientific Research Fund (EU-BAP), Grant Number: CAP-12-4044 (C. SAKALAR).
Conflict of Interest
All of the authors declare that they have no conflict of interest and no financial relationship with any commercial entity that has an interest in the subject of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakalar, C., Yuruk, M., Kaya, T. et al. Pronounced transcriptional regulation of apoptotic and TNF–NF-kappa-B signaling genes during the course of thymoquinone mediated apoptosis in HeLa cells. Mol Cell Biochem 383, 243–251 (2013). https://doi.org/10.1007/s11010-013-1772-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1772-x