Abstract
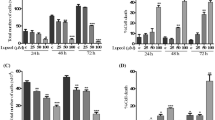
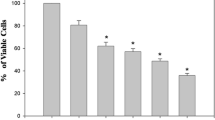
Gonolobus condurango plant extract is used as an anticancer drug in some traditional systems of medicine including homeopathy, but it apparently lacks any scientific validation. Further, no detailed study is available to suggest whether condurango-glycoside-A (CGA), a major ingredient of condurango serves as a potent anticancer compound. Therefore, we investigated apoptosis-inducing ability of CGA against cervix carcinoma cells (HeLa). β-galactosidase-activity and DNA damage were critically studied at different time points; while induced DNA-damage was observed at 9–12th hours, senescence of cells appeared at a later stage (18th hour after CGA treatment), implicating thereby a possible role of DNA damage in inducing pre-mature cell senescence. Concurrently, the number of cells undergoing apoptosis increased along with increase in reactive oxygen species (ROS) generation. Expression of p53 was also up-regulated, indicating that apoptosis could have been mediated through p53 pathway. DCHFDA (4′,6-Diamidino-2-phenylindole dihydrochloride) assay, acridine orange/ethidium bromide staining and annexin V/PI assay results collectively confirmed that apoptosis was induced by increased ROS generation. Reduction in proliferation of cells was further evidenced by the cell cycle arrest at G0/G1 stage. Expression profiles of certain relevant genes and proteins like p53, Akt, Bcl-2, Bax, cytochrome c and caspase 3 also provided evidence of ROS mediated p53 up-regulation and further boost in Bax expression and followed by cytochrome c release and activation of caspase 3. Overall results suggest that CGA initiates ROS generation, promoting up-regulation of p53 expression, thus resulting in apoptosis and pre-mature senescence associated with DNA damage.




Similar content being viewed by others
References
Abbott BJ, Leiter J, Hartwell JL, Caldwell ME, Schepartz SA (1966) Screening data from the cancer chemotherapy national service center screening laboratories. XXXIII. Plant extracts. Cancer Res 26:587–743
Boericke W (2004) Condurango Pocket manual of homeopathic material medica and repertory B. Jain Publishers, Delhi, pp 229–230
de las Heras B, Slowing K, Benedí J, Carretero E, Ortega T, Toledo C, Bermejo P, Iglesias I, Abad MJ, Gómez-Serranillos P, Liso PA, Villar A, Chiriboga X (1998) Antiinflammatory and antioxidant activity of plants used in traditional medicine in Ecuador. J Ethnopharmacol 61:161–166
Hayashi K, Wada K, Mitsuhashi H, Bando H, Takase M, Terada S, Koide Y, Aiba T, Narita T, Mizuno D (1980) Antitumor active glycosides from condurango cortex. Chem Pharm Bull (Tokyo) 28:1954–1958
Takase M, Terada S, Yamamoto H, Narita T, Kimura M, Hayashi K, Mitsuhashi H (1982) Studies on the constituents of Asclepiadaceae plants. XLIX. Confirmation of the structures of antitumor-active glycosides in condurango cortex. Chemical transformation of the aglycone moiety. Chem Pharm Bull 30:2429–2432
Berger S, Junior P, Kopanski L (1988) Structural revision of pregnane ester glycosides from condurango cortex and new compounds. Phytochemistry 27:1451–1458
Umehara K, Endoh M, Miyase T, Kuroyanagi M, Ueno A (1994) Studies on differentiation inducers. IV. Pregnane derivatives from condurango cortex. Chem Pharm Bull 42:611–616
Fulda S, Debatin KM (2006) Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene 25:4798–4811
Green JA (2011) Clinical trials in cancer. Br J Cancer 104:1521–1522
Lemarie A, Grimm S (2011) Mitochondrial respiratory chain complexes: apoptosis sensors mutated in cancer. Oncogene 300:3985–4003
Colado M, Blasco MA, Serrano M (2007) Cellular senescence in cancer and aging. Cell 130:223–233
Sappino A-P, Buser R, Seguin Q, Fernet M, Lesne L, Gumy-Pause F, Reith W, Favaudon V, Mandriota SJ (2012) The CEACAM1 tumour suppressor is an ATM and p53-regulated gene required for the induction of cellular senescence by DNA damage. Oncogenesis. doi:10.1038/oncsis.2012.7
Sikdar S, Mukherjee A, Boujedaini N, Khuda-Bukhsh AR (2013) Ethanolic extract of condurango (Marsdenia condurango) used in traditional systems of medicine including homeopathy against cancer can induce DNA damage and apoptosis in non-small lung cancer cells, A549 and H522, in vitro. Int J Genuine Tradit Med 3:e9. doi:10.5667/tang.2012.0044
Bishayee K, Mukherjee A, Paul A, Khuda-Bukhsh AR (2012) Homeopathic mother tincture of Conium initiates reactive oxygen species mediated DNA damage and makes HeLa cells prone to apoptosis. Int J Genuine Tradit Med 2:e26. doi:10.5667/tang.2012.0018
Liang CC, Park AY, Guan JL (2007) In vitro scratch assay: a conventional and inexpensive method for analysis of cell migration in vitro. Nat Protoc 2:329–333
Mauro MO, Sartori D, Oliveira RJ, Ishii PL, Mantovani MS, Ribeiro LR (2011) Activity of selenium on cell proliferation, cytotoxicity, and apoptosis and on the expression of CASP9, BCL-XL and APC in intestinal adenocarcinoma cells. Mutat Res 715:7–12
Guthrie HD, Welch GR (2006) Determination of intracellular reactive oxygen species and high mitochondrial membrane potential in Percoll-treated viable boar sperm using fluorescence-activated flow cytometry. J Anim Sci 84:2089–2100
Bishayee K, Ghosh S, Mukherjee A, Sadhukhan R, Mondal J, Khuda-Bukhsh AR (2013) Quercetin induces cytochrome-c release and ROS accumulation to promote apoptosis and arrest the cell cycle in G2/M, in cervical carcinoma: signal cascade and drug–DNA interaction. Cell Prolif 46:153–163
Paul A, Bishayee K, Ghosh S, Mukherjee A, Sikdar S, Chakraborty D, Boujedaini N, Khuda-Bukhsh AR (2012) Chelidonine isolated from ethanolic extract of Chelidonium majus promotes apoptosis in HeLa cells through p38–p53 and PI3K/AKT signalling pathways. Zhong Xi Yi Jie He Xue Bao 10:1025–1038
Bishayee K, Chakraborty D, Ghosh S, Boujedaini N, Khuda-Bukhsh AR (2013) Lycopodine triggers apoptosis by modulating 5-lipoxygenase, and depolarizing mitochondrial membrane potential in androgen sensitive and refractory prostate cancer cells without modulating p53 activity: signaling cascade and drug–DNA interaction. Eur J Pharmacol 698:110–121
Massague J (2004) G1 cell-cycle control and cancer. Nature 432:298–306
Meng Q, Xia C, Fang J, Rojanasakul Y, Jiang BH (2006) Role of PI3K and Akt specific isoforms in ovarian cancer cell migration, invasion and proliferation through the p70S6K1 pathway. Cell Signal 18:2262–2271
Vavrova J, Rezacova M (2009) Apoptosis and senescence—main mechanisms of accelerated aging of haematopoietic cells after irradiation. Acta Vet Brno 78:205–217
Lopez-Lazaro M (2007) Dual role of hydrogen peroxide in cancer: possible relevance to cancer chemoprevention and therapy. Cancer Lett 252:1–8
Yip NC, Fombon IS, Liu P, Brown S, Kannappan V, Armesilla AL, Xu B, Cassidy J, Darling JL, Wang W (2011) Disulfiram modulated ROS-MAPK and NFkB pathway and targeted breast cancer cells with cancer stem cell-like properties. Br J Cancer 104:1564–1574
Gottlieb TM, Leal JFM, Seger R, Taya Y, Oren M (2002) Cross-talk between Akt, p53 and Mdm2: possible implications for the regulation of apoptosis. Oncogene 21:1299–1303
Tsuruta F, Masuyama N, Gotoh Y (2002) The phosphatidylinositol 3-kinase (PI3K)-Akt pathway suppresses Bax translocation to mitochondria. J Biol Chem 277:14040–14047
Vento MT, Zazzu VZ, Loffreda A, Cross JR, Downward J, Stoppelli MP, Iaccarino I (2010) Praf2 is a novel Bcl-xL/Bcl-2 interacting protein with the ability to modulate survival of cancer cells. PLoS ONE 5:1–13
Tsujimoto Y, Shimizu S (2000) Bcl-2 family: life-or-death switch. FEBS Lett 466:6–10
Acknowledgments
This work was financially supported by a grant no. nil sanctioned to Prof. A. R. Khuda-Bukhsh, Department of Zoology, University of Kalyani, by Boiron Laboratories, Lyon, France.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bishayee, K., Paul, A., Ghosh, S. et al. Condurango-glycoside-A fraction of Gonolobus condurango induces DNA damage associated senescence and apoptosis via ROS-dependent p53 signalling pathway in HeLa cells. Mol Cell Biochem 382, 173–183 (2013). https://doi.org/10.1007/s11010-013-1732-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1732-5