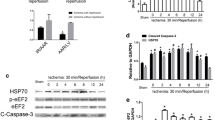
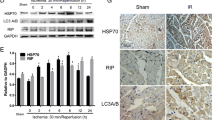
Abstract
Heat shock transcription factor 1 (HSF1), which has been identified as an endogenous cardioprotective factor, possesses considerable anti-inflammatory effects and the ability against oxidative stress. However, the mechanisms have not been fully characterized yet. In this study, we investigated the effects of HSF1-regulated HMGB1 on cardiomyocyte death. Cultured cardiomyocytes were transfected with empty vector or HSF1 plasmid before the exposure to H2O2. Cell death was assessed by cell staining. Additionally, the levels of intracellular and extracellular HMGB1 as well as its subcellular location were detected, and the expression of heat shock proteins (HSP27 and HSP90) in the cardiomyocytes was also determined. Not only did H2O2 significantly increase cell death, but also elevated the levels of intracellular and extracellular HMGB1 and induced its translocation, whereas, as expected, HSF1 overexpression effectively attenuated cell death. Furthermore, HSF1 inhibited the intracellular expression of HMGB1 at early stage of oxidative stress and subsequently, HSF1 did negatively regulate the extracellular levels and the translocation of HMGB1 at late stage. Besides, the expression of HSP27 and HSP90 was significantly increased. These results suggested HSF1-attenuated cardiomyocyte death via reducing intracellular and extracellular levels of HMGB1 as well as preventing its translocation, which was partially associated with HSP27 and HSP90 up-regulated by HSF1 overexpression.






Similar content being viewed by others
References
Rauvala H, Rouhiainen A (2010) Physiological and pathophysiological outcomes of the interactions of HMGB1 with cell surface receptors. Biochim Biophys Acta 1799:164–170
Andrassy M, Volz HC, Igwe JC et al (2008) High mobility group box-1 in ischemia–reperfusion injury of the heart. Circulation 117:3216–3226
Kim HP, Wang X, Zhang J et al (2005) Heat shock protein-70 mediates the cytoprotective effect of carbon monoxide: involvement of p38βMAPK and heat shock factor-1. J Immunol 175:2622–2629
Villa J, Ribeiro SP, Mullen JB et al (1994) Induction of the heat shock response reduces mortality rate and organ damage in sepsis-induced acute lung injury model. Crit Care Med 22:914–921
Shi Y, Tu Z, Tang D et al (2006) The inhibition of LPS-induced production of inflammatory cytokines by HSP70 involves inactivation of the NF-κB pathway but not the MAPK pathways. Shock 26:277–284
Zou Y, Zhu W, Sakamoto M et al (2003) Heat shock transcription factor 1 protects cardiomyocytes from ischemia/reperfusion injury. Circulation 108:3024–3030
Harrison EM, Sharpe E, Bellamy CO et al (2008) Heat shock protein 90-binding agents protect renal cells from oxidative stress and reduce kidney ischemia–reperfusion injury. Am J Physiol Renal Physiol 295:F397–F405
Sakamoto M, Minamino T, Toko H et al (2006) Upregulation of heat shock transcription factor 1 plays a critical role in adaptive cardiac hypertrophy. Circ Res 99:1411–1418
Toko H, Minamino T, Komuro I (2008) Role of heat shock transcriptional factor 1 and heat shock proteins in cardiac hypertrophy. Trends Cardiovasc Med 18:88–93
Tang D, Kang R, Xiao W et al (2007) Nuclear heat shock protein 72 as a negative regulator of oxidative stress (hydrogen peroxide) –induced HMGB1 cytoplasmic translocation and release. J Immunol 178:7376–7384
Pasumarthi KB, Kardami E, Cattini PA (1996) High and low molecular weight fibroblast growth factor-2 increase proliferation of neonatal rat cardiac myocytes have differential effects on binucleation and nuclear morphology. Evidence for both paracrine and intracrine actions of fibroblast growth factor-2. Circ Res 78:126–136
Tang D, Kang R, Xiao W et al (2007) The anti-inflammatory effects of heat shock protein 72 involve inhibition of high-mobility-group box 1 release and proinflammatory function in macrophages. J Immunol 179:1236–1244
Tang D, Shi Y, Kang R et al (2007) Hydrogen Peroxide stimulates macrophages and monocytes to actively release HMGB1. J Leukoc Biol 81:741–747
Tuner NA, Xia F, Azhar G et al (1998) Oxidative stress induces DNA fragmentation and caspase activation via the c-Jun NH2-terminal kinase pathway in H9c2 cardiac muscle cells. J Mol Cell Cardiol 30:1789–1801
Ulloa L, Tracy KJ (2005) The “cytokine profile”: a code for sepsis. Trends Mol Med 11:56–63
Ulloa L, Messmer D (2006) High-mobility group box 1 (HMGB1) protein: friend and foe. Cytokine Growth Factor Rev 17:189–201
Tzeng HP, Fan J, Vallejo JG et al (2008) Negative inotropic effects of high-mobility group box 1 protein in isolated contracting cardiac myocytes. Am J Physiol Heart Circ Physiol 294:H1490–H1496
Yang J, Huang C, Yang J et al (2010) Statins attenuate high mobility group box-1 protein induced vascular endothelial activation: a key role fro TLR4/NF-kappa B signaling pathway. Mol Cell Biochem 345:189–195
Xu H, Yao Y, Su Z et al (2011) Endogenous HMGB1 contributes to ischemia/reperfusion-induced myocardial apoptosis by potentiating the effect of TNFa/JNK. Am J Physiol Heart Circ Physiol 300:H913–H921
Hu X, Zhou X, He B et al (2010) Minocycline protects against myocardial ischemia and reperfusion injury by inhibiting high mobility group box 1 protein in rats. Eur J Pharmacol 638:84–89
Tsung A, Sahai R, Tanaka H et al (2005) The nuclear factor HMGB1 mediates hepatic injury after murine liver ischemia-reperfusion. J Exp Med 201:1135–1143
Liu K, Mori S, Takahashi HK et al (2007) Anti-high mobility group box 1 monoclonal antibody ameliorates brain infarction induced by transient ischemia in rats. FASEB J 21:3904–3916
Li J, Gong Q, Zhong S et al (2011) Neutralization of the extracellular HMGB1 released by ischaemic-damaged renal cells protects against renal ischaemia–reperfusion injury. Nephrol Dial Transplant 26:469–478
Creaqh-Brown BC, Quinlan GJ, Evans TW et al (2010) The RAGE axis in systemic inflammation, acute lung injury and myocardial dysfunction: an important therapeutic target? Intensive Care Med 36:1644–1656
Zhang L, Jiang H, Gao X et al (2011) Heat shock transcription factor-1 inhibits H2O2-induced apoptosis via down-regulation of reactive oxygen species in cardiac myocytes. Mol Cell Biochem 347:21–28
Acknowledgments
This work was supported by National Natural Science Foundation of China (No. 30871073) and Science and Technology Commission of Shanghai Municipality (No. 09540703500). We thank Dr. Issei Komuro (Chiba University Graduate School of Medicine, Japan) for kindly providing HSF1 plasmids.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, Y., Liu, M., Zhang, L. et al. Heat shock transcription factor 1 inhibits H2O2-induced cardiomyocyte death through suppression of high-mobility group box 1. Mol Cell Biochem 364, 263–269 (2012). https://doi.org/10.1007/s11010-012-1226-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-012-1226-x