Abstract
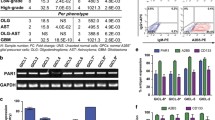
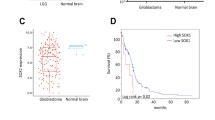
CD133 is extensively used as a surface marker to identify and isolate glioma-initiating cells (GICs) from malignant brain tumors; however, instances of CD133− cells exhibiting similar properties have also been reported. To clarify the availability of CD133 as the GIC marker, we first evaluated the ratio of CD133+ cells and malignancy of glioma spheroids GIC1 and GIC2, respectively. GIC1, which showed a lower percentage of CD133+ cells, exhibited a highly aggressive behavior in comparison with GIC2. The following experiments demonstrated that tumor suppressor PTEN was lost in GIC1, resulting in the activation of AKT pathway. Overexpression of recombinant PTEN in GIC1 suppressed its proliferation and self-renew without significant effect on CD133 expression level, indicating that the inconsistence between the ratio of CD133+ cells and proliferation and self-renewal capacity of GIC1 and GIC2 was caused by PTEN deficiency. To further validate our conclusion, a series of GICs were analyzed and the percentages of CD133+ cells could not reflect the degrees of cell proliferation and self-renewal characteristics in the PTEN deficient GICs, suggesting that the application of CD133 as the GIC maker was restricted by PTEN loss. Furthermore, down-regulation of PTEN in the PTEN-expressing GICs could break the positive correlation between the ratio of CD133+ cells and proliferation and self-renewal capacity. Our results demonstrated that PTEN status is related to cell proliferation and self-renewal independent of CD133 phenotype in the glioma-initiating cells, resulting in the limitations of CD133 as a biomarker for PTEN deficient GICs.





Similar content being viewed by others
References
Hallahan D, Geng L, Qu S, Scarfone C, Giorgio T et al (2003) Integrin-mediated targeting of drug delivery to irradiated tumor blood vessels. Cancer Cell 3:63–74
Dick JE (2008) Stem cell concepts renew cancer research. Blood 112:4793–4807
Bao S, Wu Q, McLendon RE, Hao Y, Shi Q et al (2006) Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 444:756–760
Fan X, Salford LG, Widegren B (2007) Glioma stem cells: evidence and limitation. Semin Cancer Biol 17:214–218
Lesniak MS (2006) Targeted therapy for malignant glioma: neural stem cells. Expert Rev Neurother 6:1–3
Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J et al (2004) Identification of human brain tumour initiating cells. Nature 432:396–401
Miraglia S, Godfrey W, Yin AH, Atkins K, Warnke R et al (1997) A novel five-transmembrane hematopoietic stem cell antigen: isolation, characterization, and molecular cloning. Blood 90:5013–5021
Yu Y, Flint A, Dvorin EL, Bischoff J (2002) AC133–2, a novel isoform of human AC133 stem cell antigen. J Biol Chem 277:20711–20716
Singh SK, Clarke ID, Terasaki M, Bonn VE, Hawkins C, Squire J, Dirks PB (2003) Identification of a cancer stem cell in human brain tumors. Cancer Res 63:5821–5828
Shu Q, Wong KK, Su JM, Adesina AM, Yu LT et al (2008) Direct orthotopic transplantation of fresh surgical specimen preserves CD133+ tumor cells in clinically relevant mouse models of medulloblastoma and glioma. Stem Cells 26:1414–1424
Wu A, Oh S, Wiesner SM, Ericson K, Chen L et al (2008) Persistence of CD133+ cells in human and mouse glioma cell lines: detailed characterization of GL261 glioma cells with cancer stem cell-like properties. Stem Cells Dev 17:173–184
Cheng JX, Liu BL, Zhang X (2009) How powerful is CD133 as a cancer stem cell marker in brain tumors? Cancer Treat Rev 35:403–408
Pollard SM, Yoshikawa K, Clarke ID, Danovi D, Stricker S et al (2009) Glioma stem cell lines expanded in adherent culture have tumor-specific phenotypes and are suitable for chemical and genetic screens. Cell Stem Cell 4:568–580
Beier D, Hau P, Proescholdt M, Lohmeier A, Wischhusen J et al (2007) CD133(+) and CD133(−) glioblastoma-derived cancer stem cells show differential growth characteristics and molecular profiles. Cancer Res 67:4010–4015
Joo KM, Kim SY, Jin X, Song SY, Kong DS et al (2008) Clinical and biological implications of CD133-positive and CD133-negative cells in glioblastomas. Lab Invest 88:808–815
Wang J, Sakariassen PO, Tsinkalovsky O, Immervoll H, Boe SO et al (2008) CD133 negative glioma cells form tumors in nude rats and give rise to CD133 positive cells. Int J Cancer 122:761–768
Clement V, Dutoit V, Marino D, Dietrich PY, Radovanovic I (2009) Limits of CD133 as a marker of glioma self-renewing cells. Int J Cancer 125:244–248
Alexiou GA, Voulgaris S (2010) The role of the PTEN gene in malignant gliomas. Neurol Neurochir Pol 44:80–86
Duerr EM, Rollbrocker B, Hayashi Y, Peters N, Meyer-Puttlitz B et al (1998) PTEN mutations in gliomas and glioneuronal tumors. Oncogene 16:2259–2264
Furnari FB, Fenton T, Bachoo RM, Mukasa A, Stommel JM et al (2007) Malignant astrocytic glioma: genetics, biology, and paths to treatment. Genes Dev 21:2683–2710
Maehama T, Dixon JE (1998) The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J Biol Chem 273:13375–13378
Radu A, Neubauer V, Akagi T, Hanafusa H, Georgescu MM (2003) PTEN induces cell cycle arrest by decreasing the level and nuclear localization of cyclin D1. Mol Cell Biol 23:6139–6149
Stiles B, Gilman V, Khanzenzon N, Lesche R, Li A et al (2002) Essential role of AKT-1/protein kinase B alpha in PTEN-controlled tumorigenesis. Mol Cell Biol 22:3842–3851
Hu TH, Huang CC, Lin PR, Chang HW, Ger LP et al (2003) Expression and prognostic role of tumor suppressor gene PTEN/MMAC1/TEP1 in hepatocellular carcinoma. Cancer 97:1929–1940
Izumoto S, Ohnishi T, Kanemura H, Arita N, Maruno M et al (2001) PTEN mutations in malignant gliomas and their relation with meningeal gliomatosis. J Neurooncol 53:21–26
Galli R, Binda E, Orfanelli U, Cipelletti B, Gritti A et al (2004) Isolation and characterization of tumorigenic, stem-like neural precursors from human glioblastoma. Cancer Res 64:7011–7021
Koller E, Propp S, Murray H, Lima W, Bhat B et al (2006) Competition for RISC binding predicts in vitro potency of siRNA. Nucleic Acids Res 34:4467–4476
Molofsky AV, Pardal R, Iwashita T, Park IK, Clarke MF, Morrison SJ (2003) Bmi-1 dependence distinguishes neural stem cell self-renewal from progenitor proliferation. Nature 425:962–967
Reynolds BA, Weiss S (1992) Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255:1707–1710
Reynolds BA, Weiss S (1996) Clonal and population analyses demonstrate that an EGF-responsive mammalian embryonic CNS precursor is a stem cell. Dev Biol 175:1–13
Gilbert CA, Daou MC, Moser RP, Ross AH (2010) Gamma-secretase inhibitors enhance temozolomide treatment of human gliomas by inhibiting neurosphere repopulation and xenograft recurrence. Cancer Res 70:6870–6879
Singec I, Knoth R, Meyer RP, Maciaczyk J, Volk B et al (2006) Defining the actual sensitivity and specificity of the neurosphere assay in stem cell biology. Nat Methods 3:801–806
Groszer M, Erickson R, Scripture-Adams DD, Lesche R, Trumpp A et al (2001) Negative regulation of neural stem/progenitor cell proliferation by the Pten tumor suppressor gene in vivo. Science 294:2186–2189
Zeppernick F, Ahmadi R, Campos B, Dictus C, Helmke BM et al (2008) Stem cell marker CD133 affects clinical outcome in glioma patients. Clin Cancer Res 14:123–129
Liu G, Yuan X, Zeng Z, Tunici P, Ng H et al (2006) Analysis of gene expression and chemoresistance of CD133+ cancer stem cells in glioblastoma. Mol Cancer 5:67
Hambardzumyan D, Squatrito M, Holland EC (2006) Radiation resistance and stem-like cells in brain tumors. Cancer Cell 10:454–456
Piccirillo SG, Reynolds BA, Zanetti N, Lamorte G, Binda E et al (2006) Bone morphogenetic proteins inhibit the tumorigenic potential of human brain tumour-initiating cells. Nature 444:761–765
Clark PA, Treisman DM, Ebben J, Kuo JS (2007) Developmental signaling pathways in brain tumor-derived stem-like cells. Dev Dyn 236:3297–3308
Lasky JL, Liau LM (2006) Targeting stem cells in brain tumors. Technol Cancer Res Treat 5:251–260
Rebetz J, Tian D, Persson A, Widegren B, Salford LG et al (2008) Glial progenitor-like phenotype in low-grade glioma and enhanced CD133-expression and neuronal lineage differentiation potential in high-grade glioma. PLoS One 3:e1936
Tong QS, Zheng LD, Tang ST, Ruan QL, Liu Y et al (2008) Expression and clinical significance of stem cell marker CD133 in human neuroblastoma. World J Pediatr 4:58–62
Kong DS, Kim MH, Park WY, Suh YL, Lee JI et al (2008) The progression of gliomas is associated with cancer stem cell phenotype. Oncol Rep 19:639–643
Murat A, Migliavacca E, Gorlia T, Lambiv WL, Shay T et al (2008) Stem cell-related “self-renewal” signature and high epidermal growth factor receptor expression associated with resistance to concomitant chemoradiotherapy in glioblastoma. J Clin Oncol 26:3015–3024
Keniry M, Parsons R (2008) The role of PTEN signaling perturbations in cancer and in targeted therapy. Oncogene 27:5477–5485
Altomare DA, Tanno S, De Rienzo A, Klein-Szanto AJ, Tanno S et al (2002) Frequent activation of AKT2 kinase in human pancreatic carcinomas. J Cell Biochem 87:470–476
Lee DH, Szczepanski MJ, Lee YJ (2009) Magnolol induces apoptosis via inhibiting the EGFR/PI3K/Akt signaling pathway in human prostate cancer cells. J Cell Biochem 106:1113–1122
Hambardzumyan D, Becher OJ, Rosenblum MK, Pandolfi PP, Manova-Todorova K, Holland EC (2008) PI3K pathway regulates survival of cancer stem cells residing in the perivascular niche following radiation in medulloblastoma in vivo. Genes Dev 22:436–448
Eyler CE, Foo WC, LaFiura KM, McLendon RE, Hjelmeland AB, Rich JN (2008) Brain cancer stem cells display preferential sensitivity to Akt inhibition. Stem Cells 26:3027–3036
Chugh P, Bradel-Tretheway B, Monteiro-Filho CM, Planelles V, Maggirwar SB, Dewhurst S, Kim B (2008) Akt inhibitors as an HIV-1 infected macrophage-specific anti-viral therapy. Retrovirology 5:11
Martelli AM, Tazzari PL, Tabellini G, Bortul R, Billi AM et al (2003) A new selective AKT pharmacological inhibitor reduces resistance to chemotherapeutic drugs, TRAIL, all-trans-retinoic acid, and ionizing radiation of human leukemia cells. Leukemia 17:1794–1805
Mandal M, Kim S, Younes MN, Jasser SA, El-Naggar AK, Mills GB, Myers JN (2005) The Akt inhibitor KP372–1 suppresses Akt activity and cell proliferation and induces apoptosis in thyroid cancer cells. Br J Cancer 92:1899–1905
Groszer M, Erickson R, Scripture-Adams DD, Dougherty JD, Le Belle J et al (2006) PTEN negatively regulates neural stem cell self-renewal by modulating G0–G1 cell cycle entry. Proc Natl Acad Sci USA 103:111–116
Fan X, Matsui W, Khaki L, Stearns D, Chun J, Li YM, Eberhart CG (2006) Notch pathway inhibition depletes stem-like cells and blocks engraftment in embryonal brain tumors. Cancer Res 66:7445–7452
Platet N, Liu SY, Atifi ME, Oliver L, Vallette FM, Berger F, Wion D (2007) Influence of oxygen tension on CD133 phenotype in human glioma cell cultures. Cancer Lett 258:286–290
Chen R, Nishimura MC, Bumbaca SM, Kharbanda S, Forrest WF et al (2010) A hierarchy of self-renewing tumor-initiating cell types in glioblastoma. Cancer Cell 17:362–375
Ogden AT, Waziri AE, Lochhead RA, Fusco D, Lopez K et al (2008) Identification of A2B5+ CD133− tumor-initiating cells in adult human gliomas. Neurosurgery 62:505–514
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cheng, RB., Ma, RJ., Wang, ZK. et al. PTEN status is related to cell proliferation and self-renewal independent of CD133 phenotype in the glioma-initiating cells. Mol Cell Biochem 349, 149–157 (2011). https://doi.org/10.1007/s11010-010-0669-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0669-1