Abstract
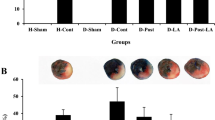
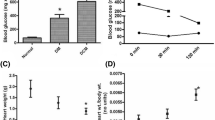
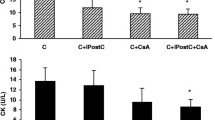
Ischemic preconditioning (IPC) produces cardioprotection by phosphorylation of glycogen synthase kinase-3β (GSK-3β) that inhibits the opening of mitochondrial permeability transition pore (MPTP). The activity of glycogen GSK-3β is elevated during diabetes mellitus (DM). This study investigated the role of GSK-3β in attenuation of cardioprotective effect of IPC in diabetic rat. DM was induced by single administration of streptozotocin (STZ, 50 mg/kg, i.p.). Isolated perfused heart was subjected to 30 min of ischemia followed by 120 min of reperfusion. Myocardial infarct size was estimated by triphenyltetrazolium chloride (TTC) staining and lactate dehydrogenase (LDH) and creatine kinase-MB (CK-MB) was analyzed in coronary effluent. IPC significantly decreased myocardial infarct size and release of LDH and CK-MB from normal rat heart. The cardioprotective effect of IPC was significantly attenuated in diabetic rat. Four episodes of preconditioning by either of GSK-3β inhibitors, lithium chloride (LiCl, 20 mM), indirubin-3 monooxime (1 μM), and SB216763 (3 μM) significantly reduced the LDH and CK-MB release and decreased infarct size in diabetic rat heart. Perfusion of atractyloside, an opener of MPTP, significantly attenuated, the cardioprotective effect of IPC in normal rat heart, and of GSK-3β inhibitor induced preconditioning in the DM rat heart. Our results suggest that preconditioning with GSK-3β inhibitors in diabetic rat heart may provide a more consistent cardioprotection, as compared to IPC. Also, the mechanism of diabetes mellitus-induced attenuation of cardioprotective effect of IPC involves activation of GSK-3β, due to impaired protective upstream signaling pathways and opening of MPTP during reperfusion.



Similar content being viewed by others
References
Murry CJ, Lopez AD (1997) Alternative projections of mortality and disability by cause 1990–2020 global burden of disease study. Lancet 349:1498–1504
Topol EJ, Calliff RM, Vandormael M, Grines CL, George BS, Sanz ML, Wall T, O’Brien M, Schwaiger M, Aguirre FV, Young S, Pompa JJ, Sigman KN, Lee KL, Ellis SG (1992) The thrombolysis and angioplasty in myocardial infarction. A randomized trial of late reperfusion therapy for acute myocardial infarction. Circulation 85:2090–2099
Baxter GF, Ebrahim Z (2002) Role of bradykinin in preconditioning and protection of the ischaemic myocardium. Br J Pharmacol 135:843–854
Piper HM, Abdullah Y, Schafer (2004) The first minutes of reperfusion: a window of opportunity for cardioprotection. Cardiovasc Res 16:365–371
Murry CE, Jennings JB, Reimer KA (1986) Preconditioning with ischemia: a delay of lethal injury in ischemic myocardium. Circulation 74:1124–1136
Nakamura M, Wang N, Zhao Z, Wilcox J, Thourani V, Guyton R, Johansen J (2000) Preconditioning decreases Bax expression, PMN accumulation and apoptosis in reperfused rat heart. Cardiovasc Res 45:661–670
Hausenloy DJ, Duchen MR, Yellon DM (2003) Inhibiting mitochondrial permeability transition pore opening at reperfusion protects against ischaemia–reperfusion injury. Cardiovasc Res 60:617–625
DeFily DV, Chilian WM (1993) Preconditioning protects coronary arteriolar endothelium from ischemia-reperfusion injury. Am J Physiol 265:H700–H706
Bouchard JF, Chouinard J, Lamontagne D (1998) Role of kinins in endothelial protective effect of ischemic preconditioning. Br J Pharmacol 123:413–420
Fryer RM, Pratt PF, Hsu AK, Gross GJ (2001) Differential activation of extracellular signal regulated kinase isoforms in preconditioning and opioid-induced cardioprotection. J Pharmacol Exp Ther 296:642–649
Iliodromitis KE, Lazou A, Kremastinos DT (2007) Ischemic preconditioning: protection against myocardial necrosis and apoptosis. Vasc Health Risk Manag 3:629–637
Ferdinandy P, Szilvassy Z, Baxter GF (1998) Adaptation to myocardial stress in disease states: is preconditioning a healthy heart phenomenon. Trends Pharmacol Sci 19:223–229
Ferdinandy P (2003) Myocardial ischaemia/reperfusion injury and preconditioning: effects of hypercholesterolaemia/hyperlipidaemia. Br J Pharmacol 138:283–285
Abete P, Ferrara N, Cioppa A, Ferrara P, Bianco S, Calabrese C, Cacciatore F, Longobardi G, Rengo F (1996) Preconditioning does not prevent postischemic dysfunction in aging heart. J Am Coll Cardiol 27:1777–1786
Liu P, Xu B, Cavalieri TA, Hock CE (2004) Attenuation of antioxidative capacity enhances reperfusion injury. In aged rat myocardium after MI/R. Am J Physiol Heart Circ Physiol 287:H2719–H2727
Snoeckx L, van der Vusse GJ, Coumans WA, Willemsen P, van der Nagpal P, Reneman RS (1986) Myocardial function in normal and spontaneously hypertensive rats during reperfusion after a period of global ischaemia. Cardiovasc Res 20:67–75
Snoeckx LH, van der Vusse GJ, Coumans WA, Willemsen PH, Reneman RS (1993) Differences in ischaemia tolerance between hypertrophied hearts of adult and aged spontaneously hypertensive rats. Cardiovasc Res 27:874–881
Sasaki H, Ogawa K, Shimizu M, Mori C, Takatsuka H, Okazaki F, Kawai M, Taniguchi I, Mochizuki S (2007) The insulin sensitizer pioglitazone improves the deterioration of ischemic preconditioning in type 2 diabetes mellitus rats. Int Heart J 48:623–635
Giricz Z, Lalu MM, Csonka C, Bencsik P, Shulz R, Ferdinandy P (2006) Hyperlipidaemia attenuate the infarct size-limitng effect of preconditioning: role of matrix metalloprotenase inhibition. J Pharmacol ExpTher 316:154–161
Ungi I, Ungi T, Ruzsa Z, Nagy E, Zimmermann Z, Csont T, Ferdinandy P (2005) Hypercholesterolemia attenuate the anti-ischemic effect of preconditioning durining coronary angioplasty. Chest 128:1623–1628
Kannel WB, McGee DL (1979) Diabetes and cardiovascular risk factor: the Framingham study. Circulation 59:8–13
Abbott RD, Donahue RP, Kannel WB, Wilson PW (1998) The impact of diabetes on survival following myocardial infarction in men vs women: the Framingham study. JAMA 260:3456–3460
Herlitz J, Wognsen GB, Emanuelsson H, Haglid M, Karlsson T, Albertsson P, Westberg S (1996) Mortality and morbidity in diabetic and nondiabetic patients during a 2-year period after coronary artery bypass grafting. Diabetes Care 19:698–703
Liu Y, Thornton JD, Cohen MV, Downey JM, Schaffer SW (1993) Streptozotocin induced non insulin dependent diabetes protect the heart from infarction. Circulation 88:1273–1278
Tatsumi T, Matoba S, Kobara M, Keira N, Kawahara A, Tsuruyuma K, Tanaka T, Katamura M, Nakagawa C, Ohta B, Yamahara Y, Asayama J, Nakagawa M (1998) Energy metabolism after ischemic preconditioning in streptozotocin induced diabetic rat hearts. J Am Coll Cardiol 31:707–715
Thirunavukkarasu M, Varma S, Penumathsa KS, Juhasz B, Zhan L, Otani H, Bagchi D, Das DK, Maulik N (2007) Resveratrol alleviates cardiac dysfunction in streptozotocin-induced diabetes: role of nitric oxide, thioredoxin, and heme oxygenase. Free Radic Biol Med 43:720–729
Tosaki A, Engelman DT, Engelman RM, Das DK (1996) The evolution of diabetic response to ischemia/reperfusion and preconditioning in isolated working rat hearts. Cardiovasc Res 31:526–536
Kersten JR, Toller WG, Gross ER, Pagel PS, Warltier DC (2000) Diabetes abolishes ischemic preconditioning: role of glucose, insulin and osmolarity. Am J Physiol Heart Circ Physiol 278:H1218–H1224
Ravingerova T, Stetka R, Volkovova K, Pancza D, Dzurba A, Ziegelhoffer A, Styk J (2000) Acute diabetes modulates response to ischemia in isolated rat heart. Mol Cell Biochem 210:143–151
Nieszner E, Posa I, Kocsis G, Preda I, Koltai MZ (2002) Influence of diabetic state of different sulfonylureas on the size of myocardial infarction with and without ischemic preconditioning in rabbits. Exp Clin Endocrinol Diabetes 110:212–218
Del Valle HF, Lascano EC, Negroni JA (2002) Ischemic preconditioning protection against stunning in conscious diabetic sheep: role of glucose, insulin, sarcolemmal and mitochondrial KATP channels. Cardiovasc Res 55:642–659
Del Valle HF, Lascano EC, Negroni JA, Crottogini AJ (2003) Absence of ischemic preconditioning protection in diabetic sheep hearts: role of sarcolemmal KATP channel dysfunction. Mol Cell Biochem 249:21–30
Hunter DR, Haworth RA (1979) The Ca-induced membrane transition in mitochondria. The protective mechanisms. Arch Biochem Biophys 195:453–459
Griffiths EJ, Halestrap AP (1995) Mitochondrial non-specific pores remain closed during cardiac ischaemia, but open upon reperfusion. Biochem J 307:93–98
Kroemer G, Dallaporta B, Resche-Rigon M (1998) The mitochondrial death/life regulator in apoptosis and necrosis. Annu Rev Physiol 60:619–642
Doble BW, Woodgett JR (2003) GSK-3: tricks of the trade for a multi-tasking kinase. J Cell Sci 16:1175–1186
Beurel E, Jope RS (2006) The paradoxical pro- and anti-apoptotic actions of GSK3 in the intrinsic and extrinsic apoptosis signaling pathways. Prog Neurobiol 79:173–189
Hausenloy DJ, Maddock HL, Baxter GF, Yellon DM (2002) Inhibiting mitochondrial permeability transition pore opening: a new paradigm for myocardial preconditioning. Cardiovasc Res 55:534–543
Juhaszaova M, Zorov DB, Kim SH, SFuQ Pepe, Fishbein KW, Ziman BD, Wang S, Ytrehus K, Antos CL, Olson EN, Sollott SJ (2004) Glycogen synthase kinase-3beta mediates convergence of protection signaling to inhibit the mitochondrial permeability transition pore. J Clin Invest 113:1535–1549
Feng J, Lucchinetti E, Ahuja P, Pasch T, Perriard JC, Zaugg M (2005) Isoflurane postconditioning prevents opening of the mitochondrial permeability transition pore through inhibition of glycogen synthase kinase 3. Anesthesiology 103:987–995
Elder-Finkelman H, Krebs EG (1999) Increased glycogen synthase kinase-3 activity in diabetes- and obesity prone-C57BL/6J mice. Diabetes 48:1662–1666
Gross ER, Hsu AK, Gross GJ (2004) Opioid-induced cardioprotection occurs via glycogensynthase kinase beta inhibition during reperfusion in intact rat hearts. Circ Res 94:960–966
Pearce NJ, Arch JR, Clapham JC, Coghlam MP, Corcoran SL, Lister CA, Llano A, Moore GB, Murphy GJ, Smith SA, Taylor CM, Yates JW, Morrison AD, Harper AJ, Roxbee-Cox L, Abuin A, Wargent E, Holder HC (2004) Development of glucose intolerance in male transgenic mice overexpressing human glycogen synthase kinase-3 beta on a muscle-specific promoter. Metabolism 53:1322–1330
Gross ER, Hsu AK, Gross GJ (2007) Diabetes abolished morphine-induced cardioprotection via multiple pathways upstream of glycogen synthase kinase-3β. Diabetes 56:127–136
Ozansoy G, Akin FB (2004) Effects of gemfibrozil treatment on vascular reactivity of streptozotocin- diabetic rat aorta. J Pharma Pharmacol 56:241–246
Trinder P (1969) Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Ann Clin Biochem 6:24
Lott J, Turner K (1975) Evaluation of trinder’s glucose oxidase method for measuring glucose in serum and urine. Clin Chem 21:1754–1760
Chopra K, Singh M, Kaul N, Andrabi KJ, Ganguly NK (1992) Decrease of myocardial infarct size with dessferrioxamine. Possible role of oxygen free radicals in its ameliorative effect. Mol Cell Biochem 113:71–76
Parikh V, Singh M (1997) Resident cardiac mast cells and the cardioprotective effect of ischemic preconditioning in isolated rat heart. J Cardiovasc Pharmacol 30:149–156
King JA (1959) A routine method for estimation of lactate dehydrogenase activity. J Med Lab Tech 16:265–272
Hughes BP (1961) A method for the estimation of serum creatine kinase and aldose activity in the normal and pathological sera. Clin Chim Acta 7:597–603
Fishbein MC, Meerbaum S, Rit J, Lando U, Kanmatsuse K, Merair JC, Corday E, Ganz W (1981) Early phase acute myocardial infarct size quantification: validation of the triphenyltetrazolium chloride tissue enzyme staining technique. Am Heart J 101:593–600
Javadov SA, Clarke S, Das M, Griffiths EJ, Lim KH, Halestrap AP (2003) Ischaemic preconditioning inhibits opening of mitochondrial permeability transition pores in the reperfused rat heart. J Physiol 549:513–524
Griffiths EJ, Halestrap AP (1993) Protection by cyclosporin A of ischemia reperfusion-induced damage in isolated rat hearts. J Mol Cell Cardiol 25:1461–1469
Xi Jinkum, Wang H, Robert A, Mueller Edward A, Xu Norfleet Zhelong (2009) Mechanism of resveratrol-induced cardioprotection against reperfusion injury involves glycogen synthase kinase 3β and mitochondrial permeability transition pore. Eur J Pharmacol 604:111–116
Park SS, Zhao H, Jang Y, Mueller RA, Xu Z (2006) N6-(3-Iodobenzyl)-adenosine-5′-N- methylcarboxamide confers cardioprotection at reperfusion by inhibiting mitochondrial permeability transition pore opening via glycogen synthase kinase 3b. J Pharmacol Exp Ther 318:124–131
Hausenloy DJ, Yellon DM (2007) Preconditioning and postconditioning; united at reperfusion. Pharmacol Therap 116:173–191
Faghihi M, Mirershadi F, Dehpour AR, Bazargan M (2008) Preconditioning with acute and chronic lithium administration reduces ischemia/reperfusion injury mediated by cyclooxygenase not nitric oxide synthase pathway in isolated rat heart. Eur J Pharmacol 597:57–63
Kaga S, Zhan L, Altaf E, Maulik N (2006) Glycogen synthase kinase-3β/β-catenin promotes angiogenic and anti-apoptotic signaling through the induction of VEGF, Bcl-2 and survivin expression in rat ischemic preconditioned myocardium. J Mol Cell Cardiol 40:138–147
Barillas R, Friehs I, Cao-Danh H, Martinez JF, del Nido PJ (2007) Inhibition of glycogen synthase kinase-3β improves tolerance to ischemia in hypertrophied hearts. Ann Throac Surg 84:126–133
Desrois M, Sidell RJ, Gauguier D, King LM, Radda GK, Clarke K (2004) Initial steps of insulin signaling and glucose transport are defective in the type 2 diabetic rat heart. Cardiovasc Res 61:288–296
Huisamen B (2003) Protein kinase B in the diabetic heart. Mol Cell Biochem 249:31–38
Acknowledgments
This study is dedicated to the memory of our esteemed colleague Prof. Manjeet Singh, who expired on 30 March, 2009, while this study was in progress. We are grateful to Mr. Praveen Garg, Chairman, ISF College of Pharmacy, Moga for his support and encouragement during the conduct of this study.
Funding source
No external source of funding was used in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yadav, H.N., Singh, M. & Sharma, P.L. Involvement of GSK-3β in attenuation of the cardioprotective effect of ischemic preconditioning in diabetic rat heart. Mol Cell Biochem 343, 75–81 (2010). https://doi.org/10.1007/s11010-010-0500-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0500-z