Abstract
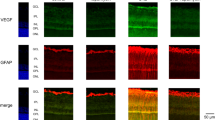
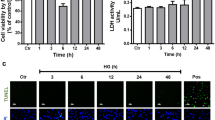
Proliferation of human retinal endothelial cells (HRECs) is an important event in the development of diabetic retinopathy. Glucose fluctuations are strong predictor of diabetic vascular complications. In this study we have investigated the effect of intermittent high glucose on proliferation and expression of vascular endothelial growth factor (VEGF) in HRECs. The possible involvement of mitochondrial reactive oxygen species (ROS) was assessed. HRECs were incubated for 72 h in media containing different glucose concentrations: 5, 25, 5 mmol/l alternating with 25 mmol/l glucose, with or without Mn(III)tetrakis(4-benzoic acid) porphyrin chloride (MnTBAP) and thenoyltri-fluoroacetone (TTFA). The cell proliferation, VEGF expression, mitochondrial ROS, nitrotyrosine and 8-hydroxydeoxyguanosine (8-OHdG) were measured. In cultured HRECs, treatment with constant or intermittent high glucose significantly increased [3H]thymidine incorporation in a time-dependent manner. Treatment with constant high glucose for 48 h resulted in significant increases in [3H]thymidine incorporation, mRNA and protein levels of VEGF compared with HRECs treated with the normal glucose, which were markedly enhanced in cells exposed to intermittent high glucose. The levels of mitochondrial ROS, nitrotyrosine and 8-OhdG were significantly elevated under both intermittent and constant high glucose conditions, the effect being greater under intermittent high glucose. In addition, the antioxidants MnTBAP or TTFA can effectively prevent cell proliferation and overexpression of VEGF, as well as overproduction of mitochondrial ROS, nitrotyrosine and 8-OhdG in HRECs induced by constant or intermittent high glucose. Intermittent high glucose enhances cell proliferation and overexpression of VEGF through reactive oxygen species (ROS) overproduction at the mitochondrial transport chain level in HRECs, indicating that glycemic variability have important pathological effects on the development of diabetic retinopathy dependent of mitochondrial ROS.





Similar content being viewed by others
References
Klein R, Klein BE, Moss SE, Davis MD, DeMets DL (1984) The Wisconsin epidemiologic study of diabetic retinopathy. II. Prevalence and risk of diabetic retinopathy when age at diagnosis is less than 30 years. Arch Ophthalmol 102:520–526
Sjolie AK, Stephenson J, Aldington S, Kohner E, Janka H, Stevens L, Fuller J (1997) Retinopathy and vision loss in insulin-dependent diabetes in Europe. The EURODIAB IDDM complications study. Ophthalmology 104:252–260
Miller JW, Adamis AP, Aiello LP (1997) Vascular endothelial growth factor in ocular neovascularization and proliferative diabetic retinopathy. Diabetes Metab Rev 13:37–50
Simorre-Pinatel V, Guerrin M, Chollet P, Penary M, Clamens S, Malecaze F, Plouet J (1994) Vasculotropin-VEGF stimulates retinal capillary endothelial cells through an autocrine pathway. Invest Ophthalmol Vis Sci 35:3393–3400
Duh E, Aiello LP (1999) Vascular endothelial growth factor and diabetes: the agonist versus antagonist paradox. Diabetes 48:1899–1906
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414:813–820
Nishikawa T, Edelstein D, Du XL, Yamagishi S, Matsumura T, Kaneda Y, Yoerk MA, Beebe D, Oates PJ, Hammes HP et al (2000) Normalizing mitochondrial superoxide production blocks three pathways of hyperglycemic damage. Nature 404:787–790
Kowluru RA, Tang J, Kern TS (2001) Abnormalities of retinal metabolism in diabetes and experimental galactosemia. VII. Effect of long-term administration of antioxidants on the development of retinopathy. Diabetes 50:1938–1942
Kowluru RA, Engerman RL, Kern TS (2000) Abnormalities of retinal metabolism in diabetes or experimental galactosemia VIII. Prevention by aminoguanidine. Curr Eye Res 21:814–819
Kuroki M, Voest EE, Amano S, Beerepoot LV, Takashima S, Tolentino M, Kim RY, Rohan RM, Colby KA, Yeo KT, Adamis AP (1996) Reactive oxygen intermediates increase vascular endothelial growth factor expression in vitro and in vivo. J Clin Invest 98:1667–1675
Obrosova IG, Minchenko AG, Marinescu V, Fathallah L, Kennedy A, Stockert CM, Frank RN, Stevens MJ (2001) Antioxidants attenuate early up regulation of retinal vascular endothelial growth factor in streptozotocin-diabetic rats. Diabetologia 44:1102–1110
Klein R (1995) Hyperglycemia and microvascular and macrovascular disease in diabetes. Diabetes Care 18:258–268
Monnier L, Mas E, Ginet C, Michel F, Villon L, Cristol JP, Colette C (2006) Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA 295:1681–1687
Gimeno-Orna JA, Castro-Alonso FJ, Boned-Juliani B, Lou-Arnal LM (2003) Fasting plasma glucose variability as a risk factor of retinopathy in type 2 diabetic patients. J Diabetes Complicat 17:78–81
Quagliaro L, Piconi L, Assaloni R (2003) Intermittent high glucose enhances apoptosis related to oxidative stress in human umbilical vein endothelial cells: the role of protein kinase C and NAD(P)H-oxidase activation. Diabetes 52:2795–2804
Xia P, Aiello LP, Ishii H, Jiang ZY, Park DJ, Robinson GS, Jiang ZY, Park DJ, Robinson GS, Jiang ZY et al (1996) Characterization of vascular endothelial growth factor’s effect on the activation of protein kinase C, its isoforms, and endothelial cell growth. J Clin Invest 98:2018–2026
Sheetz MJ, King GL (2002) Molecular understanding of hyperglycemia’s adverse effects for diabetic complications. JAMA 288:2579–2588
King GL, Brownlee M (1996) The cellular and molecular mechanisms of diabetic complications. Endocrinol Metab Clin North Am 25:255–270
De Vriese AS, Verbeuren TJ, Van de Voorde J, Lameire NH, Vanhoutte PM (2000) Endothelial dysfunction in diabetes. Br J Pharmacol 130:963–974
Hink U, Li H, Mollnau H, Oelze M, Matheis E, Hartmann M, Skatchkov M, Thaiss F, Stahl RA, Warnholtz A et al (2001) Mechanisms underlying endothelial dysfunction in diabetes mellitus. Circ Res 88:E14–E22
Williamson JR, Kilo C (1983) Capillary basement membrane in diabetes. Diabetes 32:96–100
D’Amore PA (1994) Mechanisms of retinal and choroidal neovascularization. Invest Ophthalmol Vis Sci 35:3974–3979
Sholley MM, Ferguson GP, Seibel HR, Montour JL, Wilson JD (1984) Mechanisms of neovascularization. Vascular sprouting can occur without proliferation of endothelial cells. Lab Invest 51:624–634
Witmer AN, Vrensen GF, Van Noorden CJ, Schlingemann RO (2003) Vascular endothelial growth factors and angiogenesis in eye disease. Prog Retin Eye Res 22:1–29
Gariano RF, Gardner TW (2005) Retinal angiogenesis in development and disease. Nature 438:960–966
Premanand C, Rema M, Sameer MZ, Sujatha M, Balasubramanyam M (2006) Effect of curcumin on proliferation of human retinal endothelial cells under in vitro conditions. Invest Ophthalmol Vis Sci 47:2179–2184
Kilpatrick ES, Rigby AS, Atkin SL (2009) The effect of glucose variability on the long-term risk of microvascular complications in type 1 diabetes. Diabetes Care 32:901–1903
Kilpatrick ES, Rigby AS, Atkin SL (2008) A1C variability and the risk of microvascular complications in type 1 diabetes: data from the Diabetes Control and Complications Trial. Diabetes Care 31:L2198–L2202
Jones SC, Saunders HJ, Qi W, Pollock CA (1999) Intermittent high glucose enhances cell growth and collagen synthesis in cultured human tubulointerstitial cells. Diabetologia 42:1113–1119
Takeuchi A, Throckmorton DC, Brogden AP (1995) Periodic high extracellular glucose enhances production of collagens III and IV by mesangial cells. Am J Physiol 268:F13–F19
Singleton JR, Smith AG, Russell JW, Feldman EL (2003) Microvascular complications of impaired glucose tolerance. Diabetes 52:2867–2873
Ceriello A (1998) The emerging role of post-prandial hyperglycaemic spikes in the pathogenesis of diabetic complications. Diabet Med 15:188–193
Chakrabarti S, Cukiernik M, Hileeto D, Evans T, Chen S (2000) Role of vasoactive factors in the pathogenesis of early changes in diabetic retinopathy. Diabetes Metab Res Rev 16:393–407
Sanchez-Elsner T, Botella LM, Velasco B, Corbi A, Attisano L, Bernabeu C (2001) Synergistic cooperation between hypoxia and transforming growth factor-beta pathways on human vascular endothelial growth factor gene expression. J Biol Chem 276:38527–38535
Hammes HP, Lin J, Bretzel RG, Brownlee M, Breier G (1998) Upregulation of the vascular endothelial growth experimental background diabetic retinopathy of the rat. Diabetes 47:401–406
Gilbert RE, Vranes D, Berka JL, Kelly DJ, Cox A, Wu LL, Stacker SA, Cooper ME (1998) Vascular endothelial growth factor and its receptors in control and diabetic rat eyes. Lab Invest 78:1017–1027
Balasubramanyam M, Rema M, Premanand C (2002) Biochemical and molecular mechanisms of diabetic retinopathy. Curr Sci 83:1506–1514
El-Osta A, Brasacchio D, Yao D, Pocai A, Jones PL, Roeder RG, Cooper ME, Brownlee M (2008) Transient high glucose causes persistent epigenetic changes and altered gene expression during subsequent normoglycemia. J Exp Med 205:2409–2417
Siegelaar SE, Holleman F, Hoekstra JB, DeVries JH (2010) Glucose variability: does it matter? Endocr Rev 31:171–182
Nalysnyk L, Hernandez-Medina M, Krishnarajah G (2010) Glycaemic variability and complications in patients with diabetes mellitus: evidence from a systematic review of the literature. Diabetes Obes Metab 12:288–298
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, J., Xu, Y., Sun, S. et al. Intermittent high glucose enhances cell proliferation and VEGF expression in retinal endothelial cells: the role of mitochondrial reactive oxygen species. Mol Cell Biochem 343, 27–35 (2010). https://doi.org/10.1007/s11010-010-0495-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0495-5