Abstract
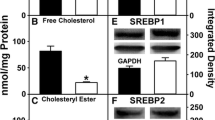
Although studies performed in vitro and with transfected cells in culture suggest a role for liver fatty acid binding protein (L-FABP) in regulating fatty acid oxidation and fat deposition, the physiological significance of this possibility is not completely clear. To begin to address this question, the effect of L-FABP gene ablation on phenotype of standard rodent chow-fed male mice was examined with increasing age up to 18 months. While young (2–3 months old) L-FABP null mice displayed no visually obvious phenotype, with increasing age >9 months the L-FABP null mice were visibly larger, exhibiting increased body weight due to increased fat and lean tissue mass. Liver lipid concentrations were unaffected by L-FABP gene ablation with the exception of triacylglycerol, which was decreased by 74% in the livers of 3-month-old mice. Likewise, serum lipid levels were not altered in L-FABP null mice with the exception of triacylglycerol, which was increased in the serum of 18-month-old mice. Increased body weight, fat tissue mass, and lean tissue mass in 18-month-old L-FABP null mice were accompanied by increased hepatic levels of low-density lipoprotein (LDL) receptor, peroxisome proliferator-activated receptor (PPAR) α, and PPARα-regulated proteins such as fatty acid transport protein (FATP), fatty acid translocase (FAT/CD36), carnitine palmitoyl transferase I (CPT I), and lipoprotein lipase (LPL). A key enzyme in cholesterol biosynthesis, 3-hydroxy-3-methylglutaryl Coenzyme A (HMG-CoA) reductase, was down-regulated in L-FABP null mice. These findings were consistent with a proposed role for L-FABP as an important physiological regulator of PPARα.





Similar content being viewed by others
References
Schroeder F, Petrescu AD, Huang H, Atshaves BP, McIntosh AL, Martin GG, Hostetler HA, Vespa A, Landrock K, Landrock D, Payne HR, Kier AB (2008) Role of fatty acid binding proteins and long chain fatty acids in modulating nuclear receptors and gene transcription. Lipids 43:1–17. doi:10.1007/s11745-007-3111-z
Desvergne B, Michalik L, Wahli W (2004) Be fit or be sick: peroxisome proliferator-activated receptors are down the road. Mol Endocrinol 18:1321–1332. doi:10.1210/me.2004-0088
Frederiksen KS, Wulf EM, Wassermann K, Sauerberg P, Fleckner J (2003) Identification of hepatic transcriptional changes in insulin-resistant rats treated with peroxisome proliferator activated receptor α agonists. J Mol Endocrinol 30:317–329. doi:10.1677/jme.0.0300317
Francis GA, Fayard E, Picard F, Auwerx J (2003) Nuclear receptors and the control of metabolism. Annu Rev Physiol 65:261–311. doi:10.1146/annurev.physiol.65.092101.142528
Lin Q, Ruuska SE, Shaw NS, Dong D, Noy N (1999) Ligand selectivity of the peroxisome proliferator-activated receptor α. Biochemistry 38:185–190. doi:10.1021/bi9816094
Hostetler HA, Petrescu AD, Kier AB, Schroeder F (2005) Peroxisome proliferator activated receptor α (PPARα) interacts with high affinity and is conformationally responsive to endogenous ligands. J Biol Chem 280:18667–18682. doi:10.1074/jbc.M412062200
Hostetler HA, Kier AB, Schroeder F (2006) Very-long-chain and branched-chain fatty acyl CoAs are high affinity ligands for the peroxisome proliferator-activated receptor α (PPARα). Biochemistry 45:7669–7681. doi:10.1021/bi060198l
Jorgensen C, Krogsdam A-M, Kratchamarova I, Willson TM, Knudsen J, Mandrup S, Kristiansen K (2002) Opposing effects of fatty acids and acyl-CoA esters on conformation and cofactor recruitment of peroxisome proliferator activated receptors. Ann N Y Acad Sci USA 967:431–439
Elholm M, Dam I, Jorgenesen C, Krogsdam A-M, Holst D, Kratchamarova I, Gottlicher M, Gustafsson JA, Berge RK, Flatmark T, Knudsen J, Mandrup S, Kristiansen K (2001) Acyl CoA esters antagonize the effects of ligands on peroxisome proliferator activated receptor α conformation, DNA binding, and interaction with cofactors. J Biol Chem 276:21410–21416. doi:10.1074/jbc.M101073200
Gottlicher M, Demoz A, Svensson D, Tollet P, Berge RK, Gustafsson JA (1993) Structural and metabolic requirements for activators of the peroxisome proliferator-activated receptor. Biochem Pharmacol 46:2177–2184. doi:10.1016/0006-2952(93)90607-X
Forman BM, Chen J, Evans RM (1997) Hypolipidemic drugs, polyunsaturated fatty acids, and eicosanoids are ligands for peroxisome proliferator-activated receptors α and δ. Proc Natl Acad Sci USA 94:4312–4317. doi:10.1073/pnas.94.9.4312
Huang H, Starodub O, McIntosh A, Kier AB, Schroeder F (2002) Liver fatty acid binding protein targets fatty acids to the nucleus: real-time confocal and multiphoton fluorescence imaging in living cells. J Biol Chem 277:29139–29151. doi:10.1074/jbc.M202923200
Huang H, Starodub O, McIntosh A, Atshaves BP, Woldegiorgis G, Kier AB, Schroeder F (2004) Liver fatty acid binding protein colocalizes with peroxisome proliferator receptor α and enhances ligand distribution to nuclei of living cells. Biochemistry 43:2484–2500. doi:10.1021/bi0352318
Faergeman NJ, Knudsen J (1997) Role of long-chain fatty acyl-CoA esters in the regulation of metabolism and in cell signalling. Biochem J 323:1–12
Knudsen J, Jensen MV, Hansen JK, Faergeman NJ, Neergard T, Gaigg B (1999) Role of acyl CoA binding protein in acyl CoA transport, metabolism, and cell signaling. Mol Cell Biochem 192:95–103. doi:10.1023/A:1006830606060
Lawrence JW, Kroll DJ, Eacho PI (2000) Ligand dependent interaction of hepatic fatty acid binding protein with the nucleus. J Lipid Res 41:1390–1401
Schroeder F, Atshaves BP, Starodub O, Boedeker AL, Smith R, Roths JB, Foxworth WB, Kier AB (2001) Expression of liver fatty acid binding protein alters growth and differentiation of embryonic stem cells. Mol Cell Biochem 219:127–138. doi:10.1023/A:1010851130136
Escher P, Wahli W (2000) Peroxisome proliferator activated receptors: insights into multiple cellular functions. Mutat Res 448:121–138. doi:10.1016/S0027-5107(99)00231-6
Kersten S (2002) Peroxisome proliferator activated receptors and obesity. Eur J Pharm 440:223–234. doi:10.1016/S0014-2999(02)01431-0
Kersten S, Seydoux J, Peters JM, Gonzalex FJ, Desvergne B, Wahli W (1999) PPARα mediates the adaptive response to fasting. J Clin Invest 103:1489–1498. doi:10.1172/JCI6223
Leone TC, Weinheimer CJ, Kelly DP (1999) A critical role of for the peroxisome proliferator activated receptor α (PPARα) in the cellular fasting response: the PPARα-null mouse as a model for fatty acid oxidation disorders. Proc Natl Acad Sci USA 96:7473–7478. doi:10.1073/pnas.96.13.7473
Hashimoto T, Cook WS, Qi C, Yeldandi AV, Reddy JK, Rao MS (2000) Defect in peroxisome proliferator activated receptor α-inducible fatty acid oxidation determines the severity of hepatic steatosis in response to fasting. J Biol Chem 275:28918–28928. doi:10.1074/jbc.M910350199
Costet P, Legendre C, More J, Edgar A, Galtier P, Pineau T (1998) PPARα deficiency leads to progressive dyslipidemia with sexually dimorphic obesity and steatosis. J Biol Chem 273:29577–29585. doi:10.1074/jbc.273.45.29577
Keller H, Dreyer C, Medin J, Mahfoudi A, Ozato K, Wahli W (1993) Fatty acids and retinoids control lipid metabolism through activation of peroxisome proliferator activated receptor-retinoid X receptor heterodimers. Proc Natl Acad Sci USA 90:2160–2164. doi:10.1073/pnas.90.6.2160
Erol E, Kumar LS, Cline GW, Shulman GI, Kelly DP, Binas B (2004) Liver fatty acid-binding protein is required for high rates of hepatic fatty acid oxidation but not for the action of PPARα in fasting mice. FASEB J 18:347–349
Newberry EP, Xie Y, Kennedy S, Buhman KK, Luo J, Gross RW, Davidson NO (2003) Decreased hepatic triglyceride accumulation and altered fatty acid uptake in mice with deletion of the liver fatty acid binding protein gene. J Biol Chem 278:51664–51672. doi:10.1074/jbc.M309377200
Atshaves BP, McIntosh AL, Lyuksyutova OI, Zipfel WR, Webb WW, Schroeder F (2004) Liver fatty acid binding protein gene ablation inhibits branched-chain fatty acid metabolism in cultured primary hepatocytes. J Biol Chem 279:30954–30965. doi:10.1074/jbc.M313571200
Martin GG, Atshaves BP, McIntosh AL, Mackie JT, Kier AB, Schroeder F (2005) Liver fatty acid binding protein (L-FABP) gene ablation alters liver bile acid metabolism in male mice. Biochem J 391:549–560. doi:10.1042/BJ20050296
Atshaves BP, McIntosh AL, Payne HR, Mackie J, Kier AB, Schroeder F (2005) Effect of branched-chain fatty acid on lipid dynamics in mice lacking liver fatty acid binding protein gene. Am J Physiol 288:C543–C558. doi:10.1152/ajpcell.00359.2004
Martin GG, Atshaves BP, McIntosh AL, Mackie JT, Kier AB, Schroeder F (2006) Liver fatty acid binding protein (L-FABP) gene ablation potentiates hepatic cholesterol accumulation in cholesterol-fed female mice. Am J Physiol 290:G36–G48
Atshaves BP, Petrescu A, Starodub O, Roths J, Kier AB, Schroeder F (1999) Expression and intracellular processing of the 58 kDa sterol carrier protein 2/3-oxoacyl-CoA thiolase in transfected mouse L-cell fibroblasts. J Lipid Res 40:610–622
Atshaves BP, Payne HR, McIntosh AL, Tichy SE, Russell D, Kier AB, Schroeder F (2004) Sexually dimorphic metabolism of branched chain lipids in C57BL/6J mice. J Lipid Res 45:812–830. doi:10.1194/jlr.M300408-JLR200
Martin GG, Danneberg H, Kumar LS, Atshaves BP, Erol E, Bader M, Schroeder F, Binas B (2003) Decreased liver fatty acid binding capacity and altered liver lipid distribution in mice lacking the liver fatty acid binding protein (L-FABP) gene. J Biol Chem 278:21429–21438. doi:10.1074/jbc.M300287200
Atshaves BP, McIntosh AL, Landrock D, Payne HR, Mackie J, Maeda N, Ball JM, Schroeder F, Kier AB (2007) Effect of SCP-x gene ablation on branched-chain fatty acid metabolism. Am J Physiol 292:939–951
Banks WA, Clever CMFCL (2000) Partial saturation and regional variation in the blood to brain transport of leptin in normal weight mice. Am J Physiol Endocrinol Metab 278:E1158–E1165
Banks WA, Farrell CL (2003) Impaired transport of leptin across the blood brain barrier in obesity is acquired and reversible. Am J Physiol Endocrinol Metab 285:E10–E15
Gallegos AM, Atshaves BP, Storey SM, Starodub O, Petrescu AD, Huang H, McIntosh A, Martin G, Chao H, Kier AB, Schroeder F (2001) Gene structure, intracellular localization, and functional roles of sterol carrier protein-2. Prog Lipid Res 40:498–563. doi:10.1016/S0163-7827(01)00015-7
Yang F, Vought BW, Satterlee JS, Walkder AK, Jim Sun Z-Y, Watts JL, DeBeaumont R, Saito RM, Hyberts SG, Yang S, Macol C, Tjian R, van den Heuvel S, Hart AC, Wagner G, Naar AM (2006) An ARC/Mediator subunit required fro SREBP control of cholesterol and lipid homeostasis. Nature 442:700–704. doi:10.1038/nature04942
Zitzer H, Wente W, Brenner MB, Sewing S, Gromada BJ, Efanov AM (2006) Sterol regulatory element binding protein-1 mediates liver X-receptor β increases in insulin secretion and insulin messenger ribonuclei acid levels. Endocrinology 147:3898–3905. doi:10.1210/en.2005-1483
McArthur MJ, Atshaves BP, Frolov A, Foxworth WD, Kier AB, Schroeder F (1999) Cellular uptake and intracellular trafficking of long chain fatty acids. J Lipid Res 40:1371–1383
Luxon BA, Weisiger RA (1993) Sex differences in intracellular fatty acid transport: role of cytoplasmic binding proteins. Am J Physiol 265:G831–G841
Weisiger RA (2005) Cytosolic fatty acid binding proteins catalyze two distinct steps in intracellular transport of their ligands. Mol Cell Biochem 239:35–42. doi:10.1023/A:1020550405578
Murphy EJ (1998) L-FABP and I-FABP expression increase NBD-stearate uptake and cytoplasmic diffusion in L-cells. Am J Physiol 275:G244–G249
Atshaves BP, Storey S, Huang H, Schroeder F (2004) Liver fatty acid binding protein expression enhances branched-chain fatty acid metabolism. Mol Cell Biochem 259:115–129. doi:10.1023/B:MCBI.0000021357.97765.f2
Jolly CA, Hubbell T, Behnke WD, Schroeder F (1997) Fatty acid binding protein: stimulation of microsomal phosphatidic acid formation. Arch Biochem Biophys 341:112–121. doi:10.1006/abbi.1997.9957
Jolly CA, Murphy EJ, Schroeder F (1998) Differential influence of rat liver fatty acid binding protein isoforms on phospholipid fatty acid composition: phosphatidic acid biosynthesis and phospholipid fatty acid remodeling. Biochim Biophys Acta 1390:258–268
Schroeder F, Jolly CA, Cho TH, Frolov AA (1998) Fatty acid binding protein isoforms: structure and function. Chem Phys Lipids 92:1–25. doi:10.1016/S0009-3084(98)00003-6
Fuchs M, Hafer A, Muench C, Kannenberg F, Teichmann S, Scheibner J, Stange EF, Seedorf U (2001) Disruption of the sterol carrier protein 2 gene in mice impairs biliary lipid and hepatic cholesterol metabolism. J Biol Chem 276:48058–48065
Hafer A, Katzberg N, Muench C, Scheibner J, Stange EF, Seedorf U, and Fuchs M (2000) Studies with sterol carrier protein-2 (SCP-2) gene knockout mice identify liver fatty acid binding protein (FABP1) as intracellular cholesterol transporter contributing to biliary cholesterol hypersecretion and gallstone formation. Gastroenterology 118(4, part 1, suppl 2), 135
Desvergne B, Wahli W (1999) Peroxisome proliferator activated receptors: nuclear control of metabolism. Endocr Rev 20:649–688. doi:10.1210/er.20.5.649
Fex M, Lucas S, Winzell MS, Ahren B, Holm C, Mulder H (2006) β-Cell lipases and insulin secretion. Diabetes 55:S24–S31. doi:10.2337/db06-S004
Tansey JT, Sztalryd C, Gruia-Gray J, Roush DL, Zee JV, Gavrilova O, Reitman ML, Deng C-X, Li C, Kimmel AR, Londos C (2001) Perilipin ablation results in a lean mouse with aberrant adipocyte lipolysis, enhanced leptin production, and resistance to diet-induced obesity. Proc Natl Acad Sci USA 98:6494–6499. doi:10.1073/pnas.101042998
Martinez-Botas J, Anderson JB, Tessier D, Lapillonne A, Chang BHJ, Quast MJ, Gorenstein D, Chen K-H, Chan L (2000) Absence of perilipin results in leanness and reverses obesity in Leprdb/db mice. Nat Genet 26:474–479. doi:10.1038/82630
Lubbers ME, van den Bos R, Spruijt BM (2007) Mu opioid receptor knockout mice in the Morris Water Maze: a learning or motivation deficit? Behav Brain Res 180:107–111. doi:10.1016/j.bbr.2007.02.021
Martin GG, Huang H, Atshaves BP, Binas B, Schroeder F (2003) Ablation of the liver fatty acid binding protein gene decreases fatty acyl CoA binding capacity and alters fatty acyl CoA pool distribution in mouse liver. Biochemistry 42:11520–11532. doi:10.1021/bi0346749
Murphy EJ, Barcelo-Coblijn G, Binas B, Glatz JFC (2004) Heart fatty acid uptake is decreased in heart fatty acid binding protein gene-ablated mice. J Biol Chem 279:34481–34488. doi:10.1074/jbc.M314263200
Binas B, Danneberg H, McWhir J, Mullins L, Clark AJ (1999) Requirement for the heart-type fatty acid binding protein in cardiac fatty acid utilization. FASEB J 13:805–812
Vassileva G, Huwyler L, Poirer K, Agellon LB, Toth MJ (2000) The intestinal fatty acid binding protein is not essential for dietary fat absorption in mice. FASEB J 14:2040–2046. doi:10.1096/fj.99-0959com
Hotamisligl GS, Johnson RS, Distel RJ, Ellis RF, Papaioannou VE, Spiegelman BM (1996) Uncoupling of obesity from insulin resistance through a targeted mutation in aP2, the adipocyte fatty acid binding protein. Science 274:1377–1379. doi:10.1126/science.274.5291.1377
Schaap FG, Binas B, Danneberg H, Van der Vusse GJ, Glatz JF (1999) Impaired long-chain fatty acid utilization by cardiac myocytes isolated from mice lacking the heart type fatty acid binding protein gene. Circ Res 85:329–337
Owada Y, Suzuki I, Noda T, Kondo H (2002) Analysis on the phenotype of E-FABP gene knockout mice. Mol Cell Biochem 239:83–86. doi:10.1023/A:1020524621933
Atshaves BP, Foxworth WB, Frolov AA, Roths JB, Kier AB, Oetama BK, Piedrahita JA, Schroeder F (1998) Cellular differentiation and I-FABP protein expression modulate fatty acid uptake and diffusion. Am J Physiol 274:C633–C644
Walkey CJ, Spiegelman BM (2008) A functional peroxisome proliferator-activated receptor γ ligand binding domina is not required for adipogenesis. J Biol Chem 283:24290–24294. doi:10.1074/jbc.C800139200
Acknowledgments
This work was supported in part by the United States Public Health Service National Institutes of Health grants DK41402 (FS and ABK), GM31651 (FS and ABK), and DK70965 (BPA).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martin, G.G., Atshaves, B.P., McIntosh, A.L. et al. Liver fatty acid binding protein gene ablation enhances age-dependent weight gain in male mice. Mol Cell Biochem 324, 101–115 (2009). https://doi.org/10.1007/s11010-008-9989-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-008-9989-9