Abstract
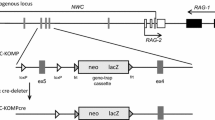
Protein kinase CK2 is a highly conserved and ubiquitous serine–threonine kinase. It is a tetrameric enzyme that is made up of two regulatory CK2β subunits and two catalytic subunits, either CK2α/CK2α, CK2α/CK2α′, or CK2α′/CK2α′. Although the two catalytic subunits diverge in their C termini, their enzymatic activities are similar. To identify the specific function of the two catalytic subunits in development, we have deleted them individually from the mouse genome by homologous recombination. We have previously reported that CK2α′ is essential for male germ cell development, and we now demonstrate that CK2α has an essential role in embryogenesis, as mice lacking CK2α die in mid-embryogenesis, with cardiac and neural tube defects.









Similar content being viewed by others
References
Seldin DC, Leder P (1995) Casein kinase II alpha transgene-induced murine lymphoma: relation to theileriosis in cattle. Science 267:894–897. doi:10.1126/science.7846532
Landesman-Bollag E, Romieu-Mourez R, Song DH et al (2001) Protein kinase CK2 in mammary gland tumorigenesis. Oncogene 20:3247–3257. doi:10.1038/sj.onc.1204411
Song DH, Dominguez I, Mizuno J et al (2003) CK2 phosphorylation of the armadillo repeat region of beta-catenin potentiates Wnt signaling. J Biol Chem 278:24018–24025. doi:10.1074/jbc.M212260200
Song DH, Sussman DJ, Seldin DC (2000) Endogenous protein kinase CK2 participates in Wnt signaling in mammary epithelial cells. J Biol Chem 275:23790–23797. doi:10.1074/jbc.M909107199
Dominguez I, Mizuno J, Wu H et al (2004) Protein kinase CK2 is required for dorsal axis formation in Xenopus embryos. Dev Biol 274:110–124. doi:10.1016/j.ydbio.2004.06.021
Wang S, Jones KA (2006) CK2 controls the recruitment of Wnt regulators to target genes in vivo. Curr Biol 16:2239–2244. doi:10.1016/j.cub.2006.09.034
Gao Y, Wang HY (2006) Casein kinase 2 is activated and essential for Wnt/beta-catenin signaling. J Biol Chem 281(27):18394–18400. doi:10.1074/jbc.M601112200
Dominguez I, Mizuno J, Wu H et al (2005) A role for CK2alpha/beta in Xenopus early embryonic development. Mol Cell Biochem 274:125–131. doi:10.1007/s11010-005-3073-5
Buchou T, Vernet M, Blond O et al (2003) Disruption of the regulatory beta subunit of protein kinase CK2 in mice leads to a cell-autonomous defect and early embryonic lethality. Mol Cell Biol 23:908–915. doi:10.1128/MCB.23.3.908-915.2003
Xu X, Toselli PA, Russell LD et al (1999) Globozoospermia in mice lacking the casein kinase II alpha’ catalytic subunit. Nat Genet 23:118–121. doi:10.1038/12729
Lou DY, Dominguez I, Toselli P et al (2008) The alpha catalytic subunit of protein kinase CK2 is required for mouse embryonic development. Mol Cell Biol 28:131–139. doi:10.1128/MCB.01119-07
Mansour SL, Thomas KR, Capecchi MR (1988) Disruption of the proto-oncogene int-2 in mouse embryo-derived stem cells: a general strategy for targeting mutations to non-selectable genes. Nature 336:348–352. doi:10.1038/336348a0
Deng CX, Wynshaw-Boris A, Shen MM et al (1994) Murine FGFR-1 is required for early postimplantation growth and axial organization. Genes Dev 8:3045–3057. doi:10.1101/gad.8.24.3045
Toselli P, Faris B, Sassoon D et al (1992) In-situ hybridization of tropoelastin mRNA during the development of the multilayered neonatal rat aortic smooth muscle cell culture. Matrix 12:321–332
Litchfield DW, Arendt A, Lozeman FJ et al (1990) Synthetic phosphopeptides are substrates for casein kinase II. FEBS Lett 261:117–120. doi:10.1016/0014-5793(90)80650-8
Seldin DC, Landesman-Bollag E, Farago M et al (2005) CK2 as a positive regulator of Wnt signalling and tumourigenesis. Mol Cell Biochem 274:63–67. doi:10.1007/s11010-005-3078-0
Luscher B, Litchfield DW (1994) Biosynthesis of casein kinase II in lymphoid cell lines. Eur J Biochem 220:521–526. doi:10.1111/j.1432-1033.1994.tb18651.x
Zhang C, Vilk G, Canton DA et al (2002) Phosphorylation regulates the stability of the regulatory CK2beta subunit. Oncogene 21:3754–3764. doi:10.1038/sj.onc.1205467
Huelsken J, Vogel R, Brinkmann V et al (2000) Requirement for beta-catenin in anterior-posterior axis formation in mice. J Cell Biol 148:567–578. doi:10.1083/jcb.148.3.567
Liebner S, Cattelino A, Gallini R et al (2004) Beta-catenin is required for endothelial-mesenchymal transformation during heart cushion development in the mouse. J Cell Biol 166:359–367. doi:10.1083/jcb.200403050
Xu C, Liguori G, Persico MG et al (1999) Abrogation of the Cripto gene in mouse leads to failure of postgastrulation morphogenesis and lack of differentiation of cardiomyocytes. Development 126:483–494
Xu C, Liguori G, Adamson ED et al (1998) Specific arrest of cardiogenesis in cultured embryonic stem cells lacking Cripto-1. Dev Biol 196:237–247. doi:10.1006/dbio.1998.8862
Morkel M, Huelsken J, Wakamiya M et al (2003) Beta-catenin regulates Cripto- and Wnt3-dependent gene expression programs in mouse axis and mesoderm formation. Development 130:6283–6294. doi:10.1242/dev.00859
Ding J, Yang L, Yan YT et al (1998) Cripto is required for correct orientation of the anterior-posterior axis in the mouse embryo. Nature 395:702–707. doi:10.1038/27215
Nusse R, Varmus HE (1992) Wnt genes. Cell 69:1073–1087. doi:10.1016/0092-8674(92)90630-U
Mastick GS, Fan CM, Tessier-Lavigne M et al (1996) Early deletion of neuromeres in Wnt-1−/− mutant mice: evaluation by morphological and molecular markers. J Comp Neurol 374:246–258. doi:10.1002/(SICI)1096-9861(19961014)374:2<246::AID-CNE7>3.0.CO;2-2
McMahon AP, Bradley A (1990) The Wnt-1 (int-1) proto-oncogene is required for development of a large region of the mouse brain. Cell 62:1073–1085. doi:10.1016/0092-8674(90)90385-R
McMahon AP, Joyner AL, Bradley A et al (1992) The midbrain-hindbrain phenotype of Wnt-1-/Wnt-1-mice results from stepwise deletion of engrailed-expressing cells by 9.5 days postcoitum. Cell 69:581–595. doi:10.1016/0092-8674(92)90222-X
Takada S, Stark KL, Shea MJ et al (1994) Wnt-3a regulates somite and tailbud formation in the mouse embryo. Genes Dev 8:174–189. doi:10.1101/gad.8.2.174
Hamblet NS, Lijam N, Ruiz-Lozano P et al (2002) Dishevelled 2 is essential for cardiac outflow tract development, somite segmentation and neural tube closure. Development 129:5827–5838. doi:10.1242/dev.00164
Galceran J, Hsu SC, Grosschedl R (2001) Rescue of a Wnt mutation by an activated form of LEF-1: regulation of maintenance but not initiation of Brachyury expression. Proc Natl Acad Sci USA 98:8668–8673. doi:10.1073/pnas.151258098
Acknowledgments
We acknowledge highly skilled technical assistance in carrying out these studies from Jessica Murray and Julie Cha, Patrick Hogan who maintains the mouse colony, and Greg Martin of the Transgenic Core at Boston University Medical Center. We are grateful to T. Yamaguchi for providing plasmids used for in situ hybridization. This work was supported by N.I.H. R01 CA71796 to David C. Seldin as well as Project 2 of P01 ES011624 (G. Sonenshein, P.I.), a Scientist Development Award from the American Heart Association (0735521T) to Isabel Dominguez, a pre-doctoral fellowship to David Y. Lou through N.I.H. T32 CA064070 (Oncobiology Training Program at Boston University School of Medicine), and a Department of Medicine Pilot Grant to Isabel Dominguez.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seldin, D.C., Lou, D.Y., Toselli, P. et al. Gene targeting of CK2 catalytic subunits. Mol Cell Biochem 316, 141–147 (2008). https://doi.org/10.1007/s11010-008-9811-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-008-9811-8