Abstract
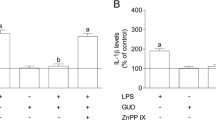
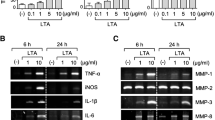
Astrocyte activation has been implicated in the pathogenesis of many neurological diseases. These reactive astrocytes are capable of producing a variety of proinflammatory mediators and potentially neurotoxic compounds, such as nitric oxide (NO), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and interleukin-1β (IL-1β). In this study, we examined the suppressive effects of Tetrandrine (TET) on astrocyte activation induced by lipopolysaccharide (LPS) in vitro. We found that TET decreased the release of NO, TNF-α, IL-6 and IL-1β in LPS-activated astrocytes. Also mRNA expression levels of inducible nitric oxide synthase (iNOS), macrophage inflammatory protein-1α (MIP-1α) and vascular cell adhesion molecule-1 (VCAM-1) were inhibited in TET pretreated astrocytes. Such suppressive effects might be resulted from the inhibition of nuclear factor kappa B (NF-κB) activation through downregulating IκB kinases (IKKs) phosphoration, which decreased inhibitor of nuclear factor-κB-α (IκBα) phosphoration and degradation. Our results suggest that TET acted to regulate astrocyte activation through inhibiting IKKs-IκBα-NF-κB signaling pathway.






Similar content being viewed by others
References
Ambrosini E, Aloisi F (2004) Chemokines and glial cells: a complex network in the central nervous system. Neurochem Res 29:1017–1038. doi:10.1023/B:NERE.0000021246.96864.89
Tuppo EE, Arias HR (2005) The role of inflammation in Alzheimer’s disease. Int J Biochem Cell Biol 37:289–305. doi:10.1016/j.biocel.2004.07.009
Holley JE, Gveric D, Newcombe J, Cuzner ML, Gutowski NJ (2003) Astrocyte characterization in the multiple sclerosis glial scars. Neuropathol Appl Neurobiol 29:434–444. doi:10.1046/j.1365-2990.2003.00491.x
Volterra A, Meldoles J (2005) Astrocytes, from brain glue to communication elements: the revolution continues. Nat Rev Neurosci 6:626–640. doi:10.1038/nrn1722
Li S, Wang L, Berman MA, Zhang Y, Dorf ME (2006) RNAi screen in mouse astrocytes identifies phosphatases that regulate NF-kappaB signaling. Mol Cell 24:497–509. doi:10.1016/j.molcel.2006.10.015
Bal-Price A, Brown GC (2000) Nitric-oxide-induced necrosis and apoptosis in PC12 cells mediated by mitochondria. J Neurochem 75:1455–1464. doi:10.1046/j.1471-4159.2000.0751455.x
Moynagh PN (2005) The interleukin-1 signaling pathway in astrocytes: a key contributor to inflammation in the brain. J Anat 207:265–269. doi:10.1111/j.1469-7580.2005.00445.x
Gimenez MA, Sim JE, Russell JH (2004) TNFR1-dependent VCAM-1 expression by astrocytes exposes the CNS to destructive inflammation. J Neuroimmunol 151:116–125. doi:10.1016/j.jneuroim.2004.02.012
Jang BC, Lim KJ, Paik JH, Cho JW, Baek WK, Suh MH et al (2004) Tetrandrine-induced apoptosis is mediated by activation of caspases and PKC-delta in U937 cells. Biochem Pharmacol 67:1819–1829. doi:10.1016/j.bcp.2004.01.018
Wu SJ, Ng LT (2007) Tetrandrine inhibits proinflammatory Cytokines, iNOS and COX-2 expression in human monocytic cells. Biol Pharm Bull 30:59–62. doi:10.1248/bpb.30.59
Chen F, Sun S, Kuhn DC, Lu Y, Gaydos LJ, Shi X et al (1997) Tetrandrine inhibits signal-induced NF-kappa B activation in rat alveolar macrophages. Biochem Biophys Res Commun 231:99–102. doi:10.1006/bbrc.1997.6057
Ho LJ, Juan TY, Chao P, Wu WL, Chang DM, Chang SY et al (2004) Plant alkaloid tetrandrine downregulates IkappaBalpha kinases-IkappaBalpha-NF-kappaB signaling pathway in human peripheral blood T cell. Br J Pharmacol 143:919–927. doi:10.1038/sj.bjp.0706000
Ho LJ, Chang DM, Lee TC, Chang ML, Lai JH (1999) Plant alkaloid Tetrandrine downregulates protein kinase C-dependent signaling pathway in T cells. Eur J Pharmacol 367:389–398. doi:10.1016/S0014-2999(98)00941-8
Xue Y, Wang Y, Feng DC, Xiao BG, Xu LY (2008) Tetrandrine suppresses lipopolysaccharide-induced microglial activation by inhibiting NF-kappaB pathway. Acta Pharmacol Sin 29:245–251. doi:10.1111/j.1745-7254.2008.00734.x
Chung IY, Benveniste EN (1990) Tumor necrosis factor-alpha production by astrocytes: induction by lipopolysaccharide, IFN-gamma, and IL-1 beta. J Immunol 144:2999–3007
Monje ML, Toda H, Palmer TD (2003) Inflammatory blockade restores adult hippocampal neurogenesis. Science 302:1760–1765. doi:10.1126/science.1088417
Xu LY, Yang JS, Xiao BG (2002) TGF-beta1-conditioned glial cell-derived dendritic cells inhibit expansion of MBP-reactive T cells in vitro. Neuroreport 13:35–39. doi:10.1097/00001756-200201210-00012
Dong Y, Benveniste EN (2001) Immune function of astrocytes. Glia 36:180–190. doi:10.1002/glia.1107
Karin M, Lin A (2002) NF-κB at the crossroads of life and death. Nat Immunol 3:221–227. doi:10.1038/ni0302-221
Ghosh S, Karin M (2002) Missing pieces in the NF-κB puzzle. Cell 109(Suppl):S81–S96. doi:10.1016/S0092-8674(02)00703-1
van Loo G, De Lorenzi R, Schmidt H, Huth M, Mildner A, Schmidt-Supprian M et al (2006) Inhibition of transcription factor NF-κB in the central nervous system ameliorates autoimmune encephalomyelitis in mice. Nat Immunol 7:954–961. doi:10.1038/ni1372
Maragakis NJ, Rothstein JD (2006) Mechanisms of Disease: astrocytes in neurodegenerative disease. Nat Clin Pract Neurol 2:679–689. doi:10.1038/ncpneuro0355
Pahan K, Sheikh FG, Khan M, Namboodiri AM, Singh I (1998) Sphingomyelinase and ceramide stimulate the expression of inducible nitric-oxide synthase in rat primary astrocytes. J Biol Chem 273:2591–2600. doi:10.1074/jbc.273.5.2591
Satoh J, Kastrukoff LF, Kim SU (1991) Cytokine-induced expression of intercellular adhesion molecule-1 (ICAM-1) in cultured human oligodendrocytes and astrocytes. J Neuropathol Exp Neurol 50:215–226. doi:10.1097/00005072-199105000-00004
Hayashi M, Luo Y, Laning J, Strieter RM, Dorf ME (1995) Production and function of monocyte chemoattractant protein-1 and other beta-chemokines in murine glial cells. J Neuroimmunol 60:143–150. doi:10.1016/0165-5728(95)00064-9
Hurwitz AA, Lyman WD, Berman JW (1995) Tumor necrosis factor alpha and transforming growth factor beta upregulate astrocyte expression of monocyte chemoattractant protein-1. J Neuroimmunol 57:193–198. doi:10.1016/0165-5728(95)00011-P
Louis JC, Magal E, Takayama S, Varon S (1993) CNTF protection of oligodendrocytes against natural and tumor necrosis factor-induced death. Science 259:689–692. doi:10.1126/science.8430320
Klein MA, Moller JC, Jones LL, Bluethmann H, Kreutzberg GW, Raivich G (1997) Impaired neuroglial activation in interleukin-6-deficient mice. Glia 19:227–233. doi:10.1002/(SICI)1098-1136(199703)19:3 < 227::AID-GLIA5 > 3.0.CO;2-W
Abbott NJ (2002) Astrocyte–endothelial interactions and blood-brain barrier permeability. J Anat 200:629–638. doi:10.1046/j.1469-7580.2002.00064.x
Ajuebor MN, Das AM, Virag L, Szabo C, Perretti M (1999) Regulation of macrophage inflammatory protein-1 alpha expression and function by endogenous interleukin-10 in a model of acute inflammation. Biochem Biophys Res Commun 255:279–282. doi:10.1006/bbrc.1999.0196
Olson TS, Ley K (2002) Chemokines and chemokine receptors in leukocyte trafficking. Am J Physiol Regul Integr Comp Physiol 283:R7–R28
Shiao YJ, Lin YL, Sun YH, Chi CW, Chen CF, Wang CN (2005) Falcarindiol impairs the expression of inducible nitric oxide synthase by abrogating the activation of IKK and JAK in rat primary astrocytes. Br J Pharmacol 144:42–51. doi:10.1038/sj.bjp.0706022
Brambilla R, Bracchi-Ricard V, Hu WH, Frydel B, Bramwell A, Karmally S et al (2005) Inhibition of astroglial nuclear factor kappa B reduces inflammation and improves functional recovery after spinal cord injury. J Exp Med 202:145–156. doi:10.1084/jem.20041918
Zhang W, Potrovita I, Tarabin V, Herrmann O, Beer V, Weih F et al (2005) Neuronal activation of NF-kappaB contributes to cell death i/n cerebral ischemia. J Cereb Blood Flow Metab 25:30–40. doi:10.1038/sj.jcbfm.9600004
Bhat NR, Zhang P, Lee JC, Hogan EL (1998) Extracellular signal-regulated kinase and p38 subgroups of mitogen-activated protein kinases regulate inducible nitric oxide synthase and tumor necrosis factor-alpha gene expression in endotoxin-stimulated primary glial cultures. J Neurosci 18:1633–1641
Schumann RR, Pfeil D, Freyer D, Buerger W, Lamping N, Kirschning CJ et al (1998) Lipopolysaccharide and pneumococcal cell wall components activate the mitogen activated protein kinases (MAPK) erk-1, erk-2, and p38 in astrocytes. Glia 22:295–305. doi:10.1002/(SICI)1098-1136(199803)22:3 < 295::AID-GLIA8 > 3.0.CO;2-4
Sun LL, Cheng C, Liu HO, Shen CC, Xiao F, Qin J et al (2007) Src suppressed C kinase substrate regulates the lipopolysaccharide-induced TNF-alpha biosynthesis in rat astrocytes. J Mol Neurosci 32:16–24. doi:10.1007/s12031-007-0003-x
Acknowledgments
This work was supported by grants from the Science and Technology Commission of Shanghai Municipality (04DZ14902), Shanghai Leading Academic Discipline Project (T0206), and the Knowledge Innovation Program of the Chinese Academy of Sciences (J0171-1905).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, ST., Wang, Y., Xue, Y. et al. Tetrandrine suppresses LPS-induced astrocyte activation via modulating IKKs-IκBα-NF-κB signaling pathway. Mol Cell Biochem 315, 41–49 (2008). https://doi.org/10.1007/s11010-008-9787-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-008-9787-4