Abstract
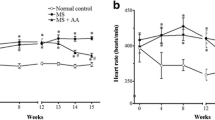
Two of the most potent vasoconstrictors, endothelin-1 (ET-1) and angiotensin II (Ang II), are upregulated in fructose hypertensive rats. It is unknown whether an interrelationship exists between these peptides that may contribute to the development of fructose-induced hypertension. The objective of this study was to investigate the existence of an interaction between the endothelin and renin angiotensin systems that may play a role in the development of fructose-induced hypertension. High fructose feeding and treatment with either bosentan, a dual endothelin receptor antagonist, or with L-158,809, an angiotensin type 1 receptor antagonist, were initiated simultaneously in male Wistar rats. Systolic blood pressure, fasted plasma parameters, insulin sensitivity, plasma Ang II, and vascular ET-1-immunoreactivity were determined following 6 weeks of high fructose feeding. Rats fed with a high fructose diet exhibited insulin resistance, hyperinsulinemia, hypertriglyceridemia, hypertension, and elevated plasma Ang II. Treatment with either bosentan or L-158,809 significantly attenuated the rise in blood pressure with no effect on insulin levels or insulin sensitivity in fructose-fed rats. Bosentan treatment significantly reduced plasma Ang II levels, while L-158,809 treatment significantly increased vascular ET-1-immunoreactivity in fructose-fed rats. Thus, treatment with the endothelin receptor antagonist prevented the development of fructose-induced hypertension and decreased plasma Ang II levels. These data suggest that ET-1 contributes to the development of fructose-induced hypertension through modulation of Ang II levels.






Similar content being viewed by others
References
Reaven GM (1988) Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 37:1595–1607. doi:10.2337/diabetes.37.12.1595
Ginsberg HN (2000) Insulin resistance and cardiovascular disease. J Clin Invest 106:453–458. doi:10.1172/JCI10762
Reaven GM (1991) Insulin resistance, hyperinsulinemia, and hypertriglyceridemia in the etiology and clinical course of hypertension. Am J Med 90:7S–12S. doi:10.1016/0002-9343(91)90028-V
Hwang IS, Ho H, Hoffman BB, Reaven GM (1987) Fructose-induced insulin resistance and hypertension in rats. Hypertension 10:512–516
Verma S, Bhanot S, McNeill JH (1995) Effect of chronic endothelin blockade in hyperinsulinemic hypertensive rats. Am J Physiol 269:H2017–H2021
Verma S, Skarsgard P, Bhanot S, Yao L, Laher I, McNeill JH (1997) Reactivity of mesenteric arteries from fructose hypertensive rats to endothelin-1. Am J Hypertens 10:1010–1019. doi:10.1016/S0895-7061(97)00107-6
Juan CC, Fang VS, Hsu YP, Huang YJ, Hsia DB, Yu PC, Kwok CF, Ho LT (1998) Overexpression of vascular endothelin-1 and endothelin-A receptors in a fructose-induced hypertensive rat model. J Hypertens 16:1775–1782. doi:10.1097/00004872-199816120-00010
Erlich Y, Rosenthal T (1995) Effect of angiotensin-converting enzyme inhibitors on fructose induced hypertension and hyperinsulinaemia in rats. Clin Exp Pharmacol Physiol Suppl 22:S347–S349. doi:10.1111/j.1440-1681.1995.tb02949.x
Iyer SN, Katovich MJ (1996) Effect of acute and chronic losartan treatment on glucose tolerance and insulin sensitivity in fructose-fed rats. Am J Hypertens 9:662–668. doi:10.1016/0895-7061(96)00035-0
Higashiura K, Ura N, Takada T, Li Y, Torii T, Togashi N, Takada M, Takizawa H, Shimamoto K (2000) The effects of an angiotensin-converting enzyme inhibitor and an angiotensin II receptor antagonist on insulin resistance in fructose-fed rats. Am J Hypertens 13:290–297. doi:10.1016/S0895-7061(99)00174-0
Galipeau D, Arikawa E, Sekirov I, McNeill JH (2001) Chronic thromboxane synthase inhibition prevents fructose-induced hypertension. Hypertension 38:872–876
Rossi GP, Sacchetto A, Cesari M, Pessina AC (1999) Interactions between endothelin-1 and the renin-angiotensin-aldosterone system. Cardiovasc Res 43:300–307. doi:10.1016/S0008-6363(99)00110-8
Yanagisawa M, Kurihara H, Kimura S, Tomobe Y, Kobayashi M, Mitsui Y, Yazaki Y, Goto K, Masaki T (1988) A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 332:411–415. doi:10.1038/332411a0
Widdop RE, Jones ES, Hannan RE, Gaspari TA (2003) Angiotensin AT2 receptors: cardiovascular hope or hype? Br J Pharmacol 140:809–824. doi:10.1038/sj.bjp.0705448
Yoshida K, Yasujima M, Kohzuki M, Kanazawa M, Yoshinaga K, Abe K (1992) Endothelin-1 augments pressor response to angiotensin II infusion in rats. Hypertension 20:292–297
Ezra-Nimni O, Ezra D, Peleg E, Munter K, Rosenthal T (2003) Trandolapril and endothelin antagonist LU-135252 in the treatment of the fructose-induced hypertensive, hyperinsulinemic, hypertriglyceridemic rat. Am J Hypertens 16:324–328. doi:10.1016/S0895-7061(03)00003-7
Clozel M, Breu V, Gray GA, Kalina B, Loffler BM, Burri K, Cassal JM, Hirth G, Muller M, Neidhart W et al (1994) Pharmacological characterization of bosentan, a new potent orally active nonpeptide endothelin receptor antagonist. J Pharmacol Exp Ther 270:228–235
Gillies LK, Werstiuk ES, Lee RM (1998) Cross-over study comparing effects of treatment with an angiotensin converting enzyme inhibitor and an angiotensin II type 1 receptor antagonist on cardiovascular changes in hypertension. J Hypertens 16:477–486. doi:10.1097/00004872-199816040-00009
Tamura T, Said S, Andersen SM, McCune SA, Mochizuki S, Gerdes AM (2002) Temporal regression of myocyte hypertrophy in hypertensive, heart failure-prone rats treated with an AT1-receptor antagonist. J Card Fail 8:43–47. doi:10.1054/jcaf.2002.32030
Matsuda M, DeFronzo RA (1999) Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22:1462–1470. doi:10.2337/diacare.22.9.1462
Gomez-Alamillo C, Juncos LA, Cases A, Haas JA, Romero JC (2003) Interactions between vasoconstrictors and vasodilators in regulating hemodynamics of distinct vascular beds. Hypertension 42:831–836. doi:10.1161/01.HYP.0000088854.04562.DA
Massart PE, Hodeige DG, Van Mechelen H, Charlier AA, Ketelslegers JM, Heyndrickx GR, Donckier JE (1998) Angiotensin II and endothelin-1 receptor antagonists have cumulative hypotensive effects in canine Page hypertension. J Hypertens 16:835–841. doi:10.1097/00004872-199816060-00015
Pollock DM, Derebail VK, Yamamoto T, Pollock JS (2000) Combined effects of AT(1) and ET(A) receptor antagonists, candesartan, and A-127722 in DOCA-salt hypertensive rats. Gen Pharmacol 34:337–342. doi:10.1016/S0306-3623(00)00079-3
Ikeda T, Ohta H, Okada M, Kawai N, Nakao R, Siegl PK, Kobayashi T, Miyauchi T, Nishikibe M (2000) Antihypertensive effects of a mixed endothelin-A- and -B-receptor antagonist, J-104132, were augmented in the presence of an AT1 -receptor antagonist, MK-954. J Cardiovasc Pharmacol 36:S337–S341
Bohlender J, Gerbaulet S, Kramer J, Gross M, Kirchengast M, Dietz R (2000) Synergistic effects of AT(1) and ET(A) receptor blockade in a transgenic, angiotensin II-dependent, rat model. Hypertension 35:992–997
d’Uscio LV, Moreau P, Shaw S, Takase H, Barton M, Luscher TF (1997) Effects of chronic ETA-receptor blockade in angiotensin II-induced hypertension. Hypertension 29:435–441
Herizi A, Jover B, Bouriquet N, Mimran A (1998) Prevention of the cardiovascular and renal effects of angiotensin II by endothelin blockade. Hypertension 31:10–14
Mortensen LH, Fink GD (1992) Captopril prevents chronic hypertension produced by infusion of endothelin-1 in rats. Hypertension 19:676–680
Jiang J, Tran L, Vasudevan H, Xia Z, Yuen VG, McNeill JH (2007) Endothelin-1 blockade prevents COX2 induction and TXA2 production in the fructose hypertensive rat. Can J Physiol Pharmacol 85:422–429. doi:10.1139/Y06-088
Lariviere R, Moreau C, Rodrigue ME, Lebel M (2004) Thromboxane blockade reduces blood pressure and progression of renal failure independent of endothelin-1 in uremic rats. Prostaglandins Leukot Essent Fatty Acids 71:103–109. doi:10.1016/j.plefa.2003.12.021
Pollock DM (2005) Endothelin, angiotensin, and oxidative stress in hypertension. Hypertension 45:477–480. doi:10.1161/01.HYP.0000158262.11935.d0
Callera GE, Touyz RM, Teixeira SA, Muscara MN, Carvalho MH, Fortes ZB, Nigro D, Schiffrin EL, Tostes RC (2003) ETA receptor blockade decreases vascular superoxide generation in DOCA-salt hypertension. Hypertension 42:811–817. doi:10.1161/01.HYP.0000088363.65943.6C
Callera GE, Tostes RC, Yogi A, Montezano AC, Touyz RM (2006) Endothelin-1-induced oxidative stress in DOCA-salt hypertension involves NADPH-oxidase-independent mechanisms. Clin Sci (Lond) 110:243–253. doi:10.1042/CS20050307
Delbosc S, Paizanis E, Magous R, Araiz C, Dimo T, Cristol JP, Cros G, Azay J (2005) Involvement of oxidative stress and NADPH oxidase activation in the development of cardiovascular complications in a model of insulin resistance, the fructose-fed rat. Atherosclerosis 179:43–49. doi:10.1016/j.atherosclerosis.2004.10.018
Si X, Webb RC, Richey JM (1999) Bezafibrate, an anti-hypertriglyceridemic drug, attenuates vascular hyperresponsiveness and elevated blood pressure in fructose-induced hypertensive rats. Can J Physiol Pharmacol 77:755–762. doi:10.1139/cjpp-77-10-755
Navarro-Cid J, Maeso R, Perez-Vizcaino F, Cachofeiro V, Ruilope LM, Tamargo J, Lahera V (1995) Effects of losartan on blood pressure, metabolic alterations, and vascular reactivity in the fructose-induced hypertensive rat. Hypertension 26:1074–1078
Chen S, Noguchi Y, Izumida T, Tatebe J, Katayama S (1996) A comparison of the hypotensive and hypoglycaemic actions of an angiotensin converting enzyme inhibitor, an AT1a antagonist and troglitazone. J Hypertens 14:1325–1330. doi:10.1097/00004872-199611000-00011
Galipeau D, Verma S, McNeill JH (2002) Female rats are protected against fructose-induced changes in metabolism and blood pressure. Am J Physiol Heart Circ Physiol 283:H2478–H2484
Fujioka Y, Masai M, Tsuboi S, Okumura T, Morimoto S, Tsujino T, Ohyanagi M, Iwasaki T (2003) Troglitazone reduces activity of the Na+/H+ exchanger in fructose-fed borderline hypertensive rats. Hypertens Res 26:111–116. doi:10.1291/hypres.26.111
Barton M, Carmona R, Ortmann J, Krieger JE, Traupe T (2003) Obesity-associated activation of angiotensin and endothelin in the cardiovascular system. Int J Biochem Cell Biol 35:826–837. doi:10.1016/S1357-2725(02)00307-2
Barton M, Carmona R, Krieger JE, Goettsch W, Morawietz H, d’Uscio LV, Lattmann T, Luscher TF, Shaw S (2000) Endothelin regulates angiotensin-converting enzyme in the mouse kidney. J Cardiovasc Pharmacol 36:S244–S247
Nussdorfer GG, Rossi GP, Belloni AS (1997) The role of endothelins in the paracrine control of the secretion and growth of the adrenal cortex. Int Rev Cytol 171:267–308. doi:10.1016/S0074-7696(08)62590-5
Sung CP, Arleth AJ, Storer BL, Ohlstein EH (1994) Angiotensin type 1 receptors mediate smooth muscle proliferation and endothelin biosynthesis in rat vascular smooth muscle. J Pharmacol Exp Ther 271:429–437
Emori T, Hirata Y, Ohta K, Shichiri M, Marumo F (1989) Secretory mechanism of immunoreactive endothelin in cultured bovine endothelial cells. Biochem Biophys Res Commun 160:93–100. doi:10.1016/0006-291X(89)91625-2
Acknowledgements
The authors thank Drs. G Kargacin, U Kumar, and L Marzban for consultation in the immunohistochemistry and Dr. CB Verchere for use of the microscope. Thanks to Drs. M Clozel and M Iglarz from Actelion Pharmaceuticals Ltd. and Dr. R Schwartz from Merck Research Laboratories for the generous gift of bosentan and L-158,809, respectively. The technical assistance of Dr. L Yao, P Rajput, K Potter, and VG Yuen is gratefully acknowledged. This project was supported by the Heart and Stroke Foundation of British Columbia and Yukon. LTT was a recipient of a Graduate Research Scholarship in Pharmacy from the Health Research Foundation of Canada’s Research-Based Pharmaceutical Companies and the Canadian Institute for Health Research and a Pacific Century Graduate Scholarship from the University of British Columbia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tran, L.T., MacLeod, K.M. & McNeill, J.H. Endothelin-1 modulates angiotensin II in the development of hypertension in fructose-fed rats. Mol Cell Biochem 325, 89–97 (2009). https://doi.org/10.1007/s11010-008-0023-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-008-0023-z