Abstract
Objective
Hypercholesterolemia (HC) has been associated with impairment of vascular and myocardial functions. As HC could generate an alteration in the oxidative status, we studied the effects of a 1-month cholesterol diet on cardiovascular oxidative stress.
Methods and Results
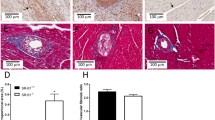
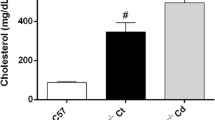
New Zealand rabbits received cholesterol (1%) or normal chow for 1 month. At 30 days, superoxide anion levels, assessed by ESR spectroscopy, NAD(P)H oxidase (NOX) activity, and dihydroethidium (DHE) staining of aortas were higher in the cholesterol-fed (CF) group compared with control (respectively, 4.0 ± 0.6 Arbitrary Units/mg (AU/mg) vs. 2.6 ± 0.3, p < 0.05; 4231 ± 433 vs. 2931 ± 373 AU/mg, p < 0.05; 21.4 ± 1.2 vs. 12.9 ± 1.7% fluorescence/mm2, p < 0.001). NOX gp91phox and p67phox expression in the aortas were higher in the CF group vs. control (1.5 ± 0.2 vs. 0.5 ± 0.2, p < 0.001; 0.9 ± 0.2 vs. 0.3 ± 0.2, p < 0.05). The endothelium-dependent relaxation evaluated on the iliac arteries was higher in control than in the CF group (64.8 ± 10.1 vs. 13.1 ± 3.70%, p < 0.001). The cardiac diastolic pressure estimated on isolated hearts was higher in the CF group than in control (21.1 ± 4.1 vs. 10.3 ± 1.4 mmHg, p < 0.05) after 60 min of ischemia.
Conclusions
Hypercholesterolemia induced increased levels of superoxide in the aortas and a higher expression of NOX subunits, associated with altered vasorelaxation. The increased diastolic pressure observed in hearts, consistent with a post-ischemic contractile dysfunction might be mediated by the production of superoxide.
Similar content being viewed by others
References
Griendling KK, Sorescu D, Ushio-Fukai M (2000) NAD(P)H oxidase: role in cardiovascular biology and disease Circ Res 86: 494–501
Sorescu D, Szocs K, Griendling KK (2001) NAD(P)H oxidases and their relevance to atherosclerosis Trends Cardiovasc Med 11: 124–131
Ohara Y, Peterson TE, Harrison DG (1993) Hypercholesterolemia increases endothelial superoxide anion production J Clin Invest 91: 2546–2551
Sorescu D, Weiss D, Lassegue B, Clempus RE, Szocs K, Sorescu GP, Valppu L, Quinn MT, Lambeth JD, Vega JD, Taylor WR, Griendling KK (2002) Superoxide production and expression of nox family proteins in human atherosclerosis Circulation 105: 1429–1435
Azumi H, Inoue N, Takeshita S, Rikitake Y, Kawashima S, Hayashi Y, Itoh H, Yokoyama M (1999) Expression of NADH/NADPH oxidase p22phox in human coronary arteries Circulation 100: 1494–1498
Barry-Lane PA, Patterson C, van der Merwe M, Hu Z, Holland SM, Yeh ET, Runge MS (2001) p47phox is required for atherosclerotic lesion progression in ApoE(−/−) mice J Clin Invest 108: 1513–1522
Pagano PJ, Clark JK, Cifuentes-Pagano ME, Clark SM, Callis GM, Quinn MT (1997) Localization of a constitutively active, phagocyte-like NADPH oxidase in rabbit aortic adventitia: enhancement by angiotensin II Proc Natl Acad Sci USA 94: 14,483–14,488
Dikalov S, Skatchkov M, Bassenge E (1997) Spin trapping of superoxide radicals and peroxynitrite by 1-hydroxy-3-carboxy-pyrrolidine and 1-hydroxy-2,2,6, 6-tetramethyl-4-oxo-piperidine and the stability of corresponding nitroxyl radicals towards biological reductants Biochem Biophys Res Commun 231: 701–704
Munzel T, Afanas’ev IB, Kleschyov AL, Harrison DG (2002) Detection of superoxide in vascular tissue Arterioscler Thromb Vasc Biol 22: 1761–1768
Warnholtz A, Nickenig G, Schulz E, Macharzina R, Brasen JH, Skatchkov M, Heitzer T, Stasch JP, Griendling KK, Harrison DG, Bohm M, Meinertz T, Munzel T (1999) Increased NADH-oxidase-mediated superoxide production in the early stages of atherosclerosis: evidence for involvement of the renin-angiotensin system Circulation 99: 2027–2033
Balcerczyk A, Soszynski M, Rybaczek D, Przygodzki T, Karowicz-Bilinska A, Maszewski J, Bartosz G (2005) Induction of apoptosis and modulation of production of reactive oxygen species in human endothelial cells by diphenyleneiodonium Biochem Pharmacol 69: 1263–1273
Cai H, Griendling KK, Harrison DG (2003) The vascular NAD(P)H oxidases as therapeutic targets in cardiovascular diseases Trends Pharmacol Sci 24: 471–478
D’Annunzio V, Donato M, Saban M, Sanguinetti SM, Wikinski RL, Gelpi RJ (2005) Hypercholesterolemia attenuates postischemic ventricular dysfunction in the isolated rabbit heart Mol Cell Biochem 273: 137–143
Ma XL, Yue TL, Lopez BL, Barone FC, Christopher TA, Ruffolo RR Jr, Feuerstein GZ (1996) Carvedilol, a new beta adrenoreceptor blocker and free radical scavenger, attenuates myocardial ischemia-reperfusion injury in hypercholesterolemic rabbits J Pharmacol Exp Ther 277: 128–136
Golino P, Maroko PR, Carew TE (1987) The effect of acute hypercholesterolemia on myocardial infarct size and the no-reflow phenomenon during coronary occlusion-reperfusion Circulation 75: 292–298
Jung O, Jung W, Malinski T, Wiemer G, Schoelkens BA, Linz W (2000) Ischemic preconditioning and infarct mass: the effect of hypercholesterolemia and endothelial dysfunction Clin Exp Hypertens 22: 165–179
Vergely C, Maupoil V, Benderitter M, Rochette L (1998) Influence of the severity of myocardial ischemia on the intensity of ascorbyl free radical release and on postischemic recovery during reperfusion Free Radic Biol Med 24: 470–479
Vergely C, Maupoil V, Clermont G, Bril A, Rochette L (2003) Identification and quantification of free radicals during myocardial ischemia and reperfusion using electron paramagnetic resonance spectroscopy Arch Biochem Biophys 420: 209–216
Bolli R (1998) Causative role of oxyradicals in myocardial stunning: a proven hypothesis. A brief review of the evidence demonstrating a major role of reactive oxygen species in several forms of postischemic dysfunction Basic Res Cardiol 93: 156–162
Hoshida S, Nishida M, Yamashita N, Igarashi J, Hori M, Kamada T, Kuzuya T, Tada M (1996) Amelioration of severity of myocardial injury by a nitric oxide donor in rabbits fed a cholesterol-rich diet J Am Coll Cardiol 27: 902–909
Rodriguez-Porcel M, Lerman A, Herrmann J, Schwartz RS, Sawamura T, Condorelli M, Napoli C, Lerman LO (2003) Hypertension exacerbates the effect of hypercholesterolemia on the myocardial microvasculature Cardiovasc Res 58: 213–221
Mohazzab HK, Kaminski PM, Wolin MS (1997) Lactate and PO2 modulate superoxide anion production in bovine cardiac myocytes: potential role of NADH oxidase Circulation 96: 614–620
Souren JE, Van Der Mast C, Van Wijk R (1997) NADPH-oxidase-dependent superoxide production by myocyte-derived H9c2 cells: influence of ischemia, heat shock, cycloheximide and cytochalasin D J Mol Cell Cardiol 29: 2803–2812
Kim KS, Takeda K, Sethi R, Pracyk JB, Tanaka K, Zhou YF, Yu ZX, Ferrans VJ, Bruder JT, Kovesdi I, Irani K, Goldschmidt-Clermont P, Finkel T (1998) Protection from reoxygenation injury by inhibition of rac1 J Clin Invest 101: 1821–1826
Harada H, Hines IN, Flores S, Gao B, McCord J, Scheerens H, Grisham MB (2004) Role of NADPH oxidase-derived superoxide in reduced size liver ischemia and reperfusion injury Arch Biochem Biophys 423: 103–108
Walder CE, Green SP, Darbonne WC, Mathias J, Rae J, Dinauer MC, Curnutte JT, Thomas GR (1997) Ischemic stroke injury is reduced in mice lacking a functional NADPH oxidase Stroke 28: 2252–2258
Kato K, Yin H, Agata J, Yoshida H, Chao L, Chao J (2003) Adrenomedullin gene delivery attenuates myocardial infarction and apoptosis after ischemia and reperfusion Am J Physiol Heart Circ Physiol 285: H1506–H1514
Ikeda Y, Young LH, Scalia R, Ross CR, Lefer AM (2001) PR-39, a proline/arginine-rich antimicrobial peptide, exerts cardioprotective effects in myocardial ischemia-reperfusion Cardiovasc Res 49: 69–77
Wang Q, Tompkins KD, Simonyi A, Korthuis RJ, Sun AY, Sun GY: Apocynin protects against global cerebral ischemia-reperfusion-induced oxidative stress and injury in the gerbil hippocampus. Brain Res 1090(1): 182–189, 2006
Wang QD, Pernow J, Sjoquist PO, Ryden L (2002) Pharmacological possibilities for protection against myocardial reperfusion injury Cardiovasc Res 55: 25–37
Wright DG, Lefer DJ (2005) Statin mediated protection of the ischemic myocardium Vascul Pharmacol 42: 265–270
Cai H, Harrison DG (2000) Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress Circ Res 87: 840–844
Mugge A, Elwell JH, Peterson TE, Hofmeyer TG, Heistad DD, Harrison DG (1991) Chronic treatment with polyethylene-glycolated superoxide dismutase partially restores endothelium-dependent vascular relaxations in cholesterol-fed rabbits Circ Res 69: 1293–1300
Griendling KK, Minieri CA, Ollerenshaw JD, Alexander RW (1994) Angiotensin II stimulates NADH and NADPH oxidase activity in cultured vascular smooth muscle cells Circ Res 74: 1141–1148
Mohazzab KM, Kaminski PM, Wolin MS (1994) NADH oxidoreductase is a major source of superoxide anion in bovine coronary artery endothelium Am J Physiol 266: H2568–H2572
Ishikawa M, Stokes KY, Zhang JH, Nanda A, Granger DN (2004) Cerebral microvascular responses to hypercholesterolemia: roles of NADPH oxidase and P-selectin Circ Res 94: 239–244
Hathaway CA, Heistad DD, Piegors DJ, Miller FJ Jr (2002) Regression of atherosclerosis in monkeys reduces vascular superoxide levels Circ Res 90: 277–283
Paravicini TM, Gulluyan LM, Dusting GJ, Drummond GR (2002) Increased NADPH oxidase activity, gp91phox expression, and endothelium-dependent vasorelaxation during neointima formation in rabbits Circ Res 91: 54–61
Laursen JB, Somers M, Kurz S, McCann L, Warnholtz A, Freeman BA, Tarpey M, Fukai T, Harrison DG (2001) Endothelial regulation of vasomotion in apoE-deficient mice: implications for interactions between peroxynitrite and tetrahydrobiopterin Circulation 103: 1282–1288
Guzik TJ, Sadowski J, Guzik B, Jopek A, Kapelak B, Przybylowski P, Wierzbicki K, Korbut R, Harrison DG, Channon KM (2006) Coronary artery superoxide production and nox isoform expression in human coronary artery disease Arterioscler Thromb Vasc Biol 26: 333–339
O’Donnell RW, Johnson DK, Ziegler LM, DiMattina AJ, Stone RI, Holland JA (2003) Endothelial NADPH oxidase: mechanism of activation by low-density lipoprotein Endothelium 10: 291–297
Li JM, Mullen AM, Yun S, Wientjes F, Brouns GY, Thrasher AJ, Shah AM (2002) Essential role of the NADPH oxidase subunit p47(phox) in endothelial cell superoxide production in response to phorbol ester and tumor necrosis factor-alpha Circ Res 90: 143–150
Ago T, Kitazono T, Ooboshi H, Iyama T, Han YH, Takada J, Wakisaka M, Ibayashi S, Utsumi H, Iida M (2004) Nox4 as the major catalytic component of an endothelial NAD(P)H oxidase Circulation 109: 227–233
Kalinina N, Agrotis A, Tararak E, Antropova Y, Kanellakis P, Ilyinskaya O, Quinn MT, Smirnov V, Bobik A (2002) Cytochrome b558-dependent NAD(P)H oxidase-phox units in smooth muscle and macrophages of atherosclerotic lesions Arterioscler Thromb Vasc Biol 22: 2037–2043
Namiki M, Kawashima S, Yamashita T, Ozaki M, Hirase T, Ishida T, Inoue N, Hirata K, Matsukawa A, Morishita R, Kaneda Y, Yokoyama M (2002) Local overexpression of monocyte chemoattractant protein-1 at vessel wall induces infiltration of macrophages and formation of atherosclerotic lesion: synergism with hypercholesterolemia Arterioscler Thromb Vasc Biol 22: 115–120
Clubb FJ, Cerny JL, Deferrari DA, Butler-Aucoin MM, Willerson JT, Buja LM (2001) Development of atherosclerotic plaque with endothelial disruption in Watanabe heritable hyperlipidemic rabbit aortas Cardiovasc Pathol 10: 1–11
Lassegue B, Clempus RE (2003) Vascular NAD(P)H oxidases: specific features, expression, and regulation Am J Physiol Regul Integr Comp Physiol 285: R277–R297
Szocs K, Lassegue B, Sorescu D, Hilenski LL, Valppu L, Couse TL, Wilcox JN, Quinn MT, Lambeth JD, Griendling KK (2002) Upregulation of Nox-based NAD(P)H oxidases in restenosis after carotid injury Arterioscler Thromb Vasc Biol 22: 21–27
Esposito LA, Melov S, Panov A, Cottrell BA, Wallace DC (1999) Mitochondrial disease in mouse results in increased oxidative stress Proc Natl Acad Sci USA 96: 4820–4825
Wallace DC (1992) Mitochondrial genetics: a paradigm for aging and degenerative diseases? Science 256: 628–632
Madamanchi NR, Vendrov A, Runge MS (2005) Oxidative stress and vascular disease Arterioscler Thromb Vasc Biol 25: 29–38
Puddu P, Puddu GM, Galletti L, Cravero E, Muscari A (2005) Mitochondrial dysfunction as an initiating event in atherogenesis: a plausible hypothesis Cardiology 103: 137–141
Acknowledgments
The authors gratefully acknowledge l’Association de Cardiologie de Bourgogne, le Commissariat à l’Energie Atomique, Philip Bastable (editorial assistance), Professor Philip Gambert (Biochemistry Department, University Hospital of Dijon) and Franck Ménétrier (Histology Department, Faculty of medicine of Dijon) for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Collin, B., Busseuil, D., Zeller, M. et al. Increased superoxide anion production is associated with early atherosclerosis and cardiovascular dysfunctions in a rabbit model. Mol Cell Biochem 294, 225–235 (2007). https://doi.org/10.1007/s11010-006-9263-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-006-9263-y