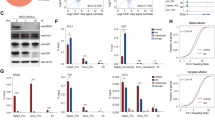
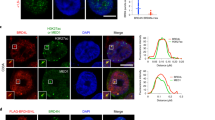
Abstract
Brd2 is a novel protein kinase and plays a role in cell cycle-responsive transcription. Recent studies show that Brd2 contributes to E2F-1 regulated cell cycle progression. In this process, Brd2 exhibits scaffold or transcriptional adapter functions and mediates recruitment of both E2F-1 transcription factors and chromatin-remodelling activity to the E2F-1-resposive promoter. In the present study, we show that Brd2 is also a TBP-associated protein and a 26 amino acids peptide in the first bromodomain of Brd2 is essential for Brd2-TBP interaction. We found that serum stimulation of serum starved NIH/3T3 cells efficiently induces the formation of the Brd2-E2F-1-TBP complex in vivo. In this process, Brd2 plays a pivotal role in the recruitment of TBP into a E2F-1 transcriptional complex, as tested in overexpression assay and at the endogenous level. Furthermore, the 26 amino acid peptide that mediates Brd2-TBP interaction is proved to be critical for Brd2-dependent transactivation on E2F-1-responsive promoters, and moreover, Brd2 and E2F-1 may cooperatively participate in various serum-induced transactivation processes in Luciferase-reporter assays. Thus taken together, because Brd2 may recruit a HAT in its transactivational complex and E2F-1 has been found to stimulate transcription by recruiting acetyltransferase and cofactors GCN5, we predict that Brd2 and E2F-1 may act in a cooperative way to introduce an optimal environment for TBP binding to the TATA-element of gene promoters.
Similar content being viewed by others
References
Berk AJ: Activation of RNA polymerase II transcription. Curr Opin Cell Biol 11: 330–335, 1999
Orphanides G, Lagrange T, Reinberg D: The general transcription factors of RNA polymerase II. Genes Dev 10: 2657–2683, 1996
Beverly ME: Specificity of gene regulation. Cell 109: 267–270, 2002
Lee TI, Young RA: Transcription of eukaryotic protein-coding genes. Annu Rev Genet 34: 77–137, 2000
Imbalzano AN, Kwon H, Green MR, Kingston RE: Facilitated binding of TATA-binding protein to nucleosomal DNA. Nature 370: 481–485, 1994
Sherr CJ: Cancer cell cycles. Science 274: 1672–1677, 1996
Sylvester AM, Chen D, Krasinski K, Andres V: Role of c-fos and E2F in the induction of cyclin A transcription and vascular smooth muscle cell proliferation. J Clin Invest 101: 940–948, 1998
Lang SE, McMahon SB, Cole MD, Hearing P: E2F transcriptional activation requires TRRAP and GCN5 cofactors. J Biol Chem 276: 32627–32634, 2001
Denis GV, Vaziri C, Guo N, Faller DV: RING3 kinase transactivates promoters of cell cycle regulatory genes through E2F. Cell Grow Diff 11: 417–424, 2000
Guo N, Faller DV, Denis GV: Activation-induced nuclear translocation of RING3. J Cell Sci 113: 3085–3091, 2000
Greenwald RJ, Tumang JR, Sinha A, Currier N, Cardiff RD, Rothstein TL, Faller DV, Denis GV: Eμ-BRD2 transgenic mice develop B-cell lymphoma and leukemia. Blood 103: 1475–1484, 2004
Chua P, Roeder GS: BDF1, a yeast chromosomal protein required for sporulation. Mol Cell Biol 15: 3685–3696, 1995
Matangkasombut O, Buratowski RM, Swilling NW, Buratowski S: Bromodomain factor 1 corresponds to a missing piece of yeast TFIID. Genes Dev 14: 951–962, 2000
Sinha A, Faller DV, Denis GV: Bromodomain analysis of Brd2-dependent transcriptional activation of cyclin A. Biochem J 387: 257–269, 2005
Hudson BP, Martinez-Yamout MA, Dyson HJ, Wright PE: Solution structure and acetyl-lysine binding activity of the GCN5 bromodomain. J Mol Biol 304: 355–370, 2000
Dhalluin C, Carlson J, Zeng L, He C, Aggarwal AK, Zhou MM: Structure and ligand of a histone acetyltransferase bromodomain. Nature 399: 491–496, 1999
Bannister AJ, Kouzarides T: The CBP co-activator is a histone acetyltransferase. Nature 384: 641–643, 1996
Mizzen CA, Yang XJ, Kokubo T, Brownell JE, Bannister AJ, Owen-Hughes T, Workman J, Wang L, Berger SL, Kouzarides T, Nakatani Y, Allis D: The TAFII250 subunit of TFIID has histone acetyltransferase activity. Cell 87: 1261–1270, 1996
Ruppert S, Wang EH, Tjian R: Cloning and expression of human TAFII250: a TBP-associated factor implicated in cell-cycle regulation. Nature 362: 175–179, 1993
Tamkun JW, Deuring R, Scott MP, Kissinger M, Pattatucci AM, Kaufman TC, Kennison JA: A regulator of Drosophila homeotic genes structurally related to the yeast transcriptional activator SNF2/SWI2. Cell 68: 561–572, 1992
Winston F, Allis CD: The bromodomain: a chromatin-targeting module? Nat Struct Biol 6: 601–604, 1999
Crowley TE, Kaine EM, Yoshida M, Nandi A, Wolgemuth DJ: Reproductive cycle regulation of nuclear import, euchromatic localization, and association with components of Pol II mediator of a mammalian double-bromodomain protein. Mol Endocrinol 16: 1727–1737, 2002
Sterner DE, Grant PA, Roberts SM, Duggan LJ, Belotserkovskaya R, Pacella LA, Winston F, Workman JL, Berger SL: Functional organization of the yeast SAGA complex: distinct components involved in structural integrity, nucleosome acetylation, and TATA-Binding Protein interaction. Mol Cell Biol 19: 86–98, 1999
Ornaghi P, Ballario P, Lena AM, Gonzalez A, Filetici P: The bromodomain of GCN5p interacts in vitro with specific residues in the N terminus of histone H4. J Mol Biol 287: 1–7, 1999
Poux AN, Marmorstein R: Molecular basis for GCN5/PCAF histone acetyltransferase selectivity for histone and nonhistone substrates. Biochemistry 42: 14366–14374, 2003
Kimura A, Matsubara K, Horikoshi M: A decade of histone acetylation: marking eukaryotic chromosomes with specific codes. J Biochem 138: 647–662, 2005
Biswas D, Imbalzano AN, Eriksson P, Yu Y, Stillman DJ: Role for Nhp6, GCN5, and the Swi/Snf complex in stimulating formation of the TATA-Binding Protein-TFIIA-DNA complex. Mol Cell Biol 24: 8312–8321, 2004
Denis GV, McComb ME, Faller DV, Sinha A, Romesser PB, Costello CE: Identification of transcription complexes that contain the double bromodomain protein Brd2 and chromatin remodeling machines. J Proteome Res 5: 502–511, 2006
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peng, J., Dong, W., Chen, L. et al. Brd2 is a TBP-associated protein and recruits TBP into E2F-1 transcriptional complex in response to serum stimulation. Mol Cell Biochem 294, 45–54 (2007). https://doi.org/10.1007/s11010-006-9223-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-006-9223-6