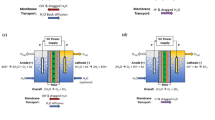
Protein electrochemistry offers a direct method to identify and characterize biological electron transfer processes, potentially leading to commercial applications such as biosensors and diagnostic tools. However, establishing a biocompatible electrode interface that maintains the native state of the redox protein involves several challenges. In general, membrane proteins require the presence of a phospholipid bilayer to maintain their biological activity. Synthetic `biomimetic’ membranes are widely used to characterize membrane proteins, however they have seldom been applied to measurements of protein redox activity in electrochemical cells due to their inherent insulating property. In this study we demonstrate the use of the phospholipids: PC, PC/PG and PC/PG/cholesterol membrane mixtures on chemically modified (supported) gold electrode surfaces for direct protein electrochemistry. We compare the electrochemical activity of a relatively small, redox active “test protein”, cytochrome c, in the presence and absence of phospholipid on a gold electrode modified with thiol self assembled monolayers, to explore the effect of chain length and composition of the thiol on the charge coupling. Three thiols were investigated as self assembled monolayers on a gold electrode: octanethiol, mercaptopropionic and mercaptoundecanoic acid. We demonstrate here that the charge transfer efficiency of cytochrome c is better in the presence of the membrane and in addition, a superior redox response is obtained with surfaces modified with a thiol functionalised with a carboxylic acid.





Similar content being viewed by others
Abbreviations
- MPA:
-
mercaptopropionic acid
- MUA:
-
mercaptoundecanoic acid
- SAM:
-
self-assembled monolayer
- PBS:
-
phosphate buffered saline
- DMPC:
-
1,2-dimyristoyl-l-α-phosphatidylcholine
- DMPG:
-
1,2-dimyristoyl-l-α-phosphatidyl-dl-glycerol
References
Armstrong F. A., Bond A. M., Hill H. A. O., Oliver B. N., Psalti I. S. M., 1989a J. Am. Chem. Soc. 111: 9185–9189
Armstrong F. A., Bond A. M., Hill H. A. O., Psalti I. S. M., Zoski C. G., 1989b J. Phys. Chem. 93: 6485–6493
Armstrong F. A., Hill H. A. O., Walton N. J., 1988 Acc. Chem. Res.21: 407–413
Bianco P., 2002Rev. Mol. Biotechnol. 82: 393–409
Bond, A. M., Fleming, B. D. and Martin, L. L.: 2006, in A. Sigel et al. (eds.), The Ubiquitous Roles of Cytochrome P450 Proteins, Wiley & Sons, Chichester (invited book chapter)
Bond A. M., 1994 Inorg. Chim. Acta 226: 293–340
Bond A. M., Hill H. A. O., Lovric S. K., Lovric M., Mc Carthy M. E., Psalti I. S. M., Walton N. J., 1992 J. Phys. Chem. 96: 8100–8105
Cooper M. A., Try A. C., Carroll J., Ellar D. J., Williams D. H., 1998 Biochim. Biophys. Acta 1373: 101–111
Fedurco M., 2000 Coord. Chem. Rev. 209: 263–331
Feng Z. Q., Imabayashi S., Kakiuchi T., Niki K., 1997 J. Chem. Soc., Faraday Trans. 93: 1367–1370
Fleming B. D., Tan Y., Bell S. G., Wong L. L., Urlacher V., Hill H. A. O., 2003 Eur. J. Biochem. 27: 4082–4088
Honeychurch M. J., Rechnitz G. A., 1997 J. Phys. Chem. B 101: 7472–7479
Hook F., Ray A., Norden B., Kasemo B., 2001 Langmuir 17: 8305–8312
Hu N., 2001 Pure Appl. Chem. 73: 1979–1991
Johnson D. L., Martin L. L., 2005 J. Am. Chem. Soc. 127: 2018–2019
Johnson D. L., Lewis B. C., Elliot D. J., Miners J. O., Martin L. L., 2005 Biochem. Pharmacol. 69: 1533–1541
Johnson, D. L.: 2004, PhD Thesis, Flinders University of South Australia, Australia
Johnson D. L., Thompson J. L., Brinkmann S. M., Schuller K. A., Martin L. L., 2003a Biochemistry 42: 10,229–10,237
Johnson D., Norman S., Tuckey C., Martin L. L., 2003b Bioelectrochemistry 59: 41–47
Johnson D. L., Polyak S. W., Wallace J. C., Martin, L. L., 2003c Lett. Pept. Sci. 10(2): 495–500
Johnson D. L., Maxwell C. J., Losic D., Shapter J. G., Martin L. L., 2002, Bioelectrochemistry 58(2): 137–147
Kaim W., Schewederski B., 1994Bioinorganic Chemistry: Inorganic Elements in the Chemistry of Life, An Introduction and Guide John Wiley & Sons Ltd England
Kanazawa K. K., Gordon J. G., 1985 Anal. Chem.57: 1770–1771
Kamimori H., Hall K., Craik D. J., Aguilar M. I., 2004 Anal. Biochem. 337: 149–153
Keller C. A., Kasemo B., 1998 Biophys. J. 75: 1397–1402
Kern W., Puotinen D. A., 1970 RCA Rev. 31(2): 187–206
Lojou E., Bianco P., 2000 J. Electroanal. Chem. 485: 71–80
Mozsolits H., Wirth H. J., Werkmeister J., Aguilar M. I., 2001 Biochim. Biophys. Acta 1512: 64–76
Muller D. J., Hand G. M., Engel A., Sosinsky G. E.: 2002, Eur. Mol. Biol. Organ. J. 21, 3598–3607
Plant A. L., Gueguetchkeri M., Yap W., 1994 Biophys. J. 67: 1126–1133
Plant A. L., 1993 Langmuir 9: 2764–2767
Richter R., Mukhopadhyay A., Brisson A., 2003 Biophys. J. 85: 3035–3047
Rusling J. F., 1998 Acc. Chem. Res. 37: 363–369
Rusling J. F., Nassar A. E., 1993 J. Am. Chem. Soc. 115: 11,891–11,897
Sauerbrey G., 1959 Z. Phys.155: 206–222
Scheller F. W., Wollenberger U., Lei C., Jin W., Ge B., Lehmann C., Lisdat F., Fridmann V., 2002 Rev. Mol. Biotech. 82: 411–424
Svedhem S., Dahlborg D., Ekeroth J., Kelly J., Hook F., Gold J., 2003 Langmuir 19: 6730–6736
Tarlov M. J., Bowden E. F., 1991 J. Am. Chem. Soc. 113: 1847–1849
Thimm J., Mechler A., Lin H., Rhee S., Lal R., 2005. J. Biol. Chem. 280(11): 10,646–10,654
Zhang W., Li G., 2004 Anal. Sci. 20: 603–609
Zhang Z., Rusling J. M., 1997 Biophys. Chem.63: 133–146
Acknowledgments
This work was supported by a grant from the National Health and Medical Research Council. The Australian Research Council Discovery and LIEF Grants, Monash University and Victorian Institute for Chemical Sciences are also acknowledged for financial support for purchase of a Q-sense QCM and electrochemical equipment. We thank Dr John Lee for technical assistance with the Scheme.
Author information
Authors and Affiliations
Corresponding author
Additional information
On leave from: Research Group on Laser Physics of the Hungarian Academy of Sciences, University of Szeged, Szeged, Hungary.
Australian Peptide Conference Issue.
Rights and permissions
About this article
Cite this article
Mechler, A., Nawaratna, G., Aguilar, MI. et al. A Study of Protein Electrochemistry on a Supported Membrane Electrode. Int J Pept Res Ther 12, 217–224 (2006). https://doi.org/10.1007/s10989-006-9029-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-006-9029-0