Abstract
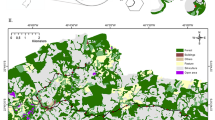
Habitat fragmentation is a major cause for species loss, but its effect on invertebrates with low active dispersal power, like terrestrial gastropods, has rarely been studied. Such species can not cross a hostile habitat matrix, for which the predictions of island theory, such as positive relations between species richness and patch size, should apply. In order to test this prediction, we studied gastropod species diversity by assessing gastropod assemblage characteristics from 35 sites in 19 fragments of deciduous old-growth forests in the Lower Rhine Embayment, Germany. Assemblages differed between larger (≥700 ha) and smaller forests (<400 ha), those of large forests held a higher percentage of forest species. Although α-diversity was similar between the two forest size classes, small forests often comprised matrix species, resulting in a higher β-diversity. Edge effects on the species richness of matrix species were noticeable up to 250 m into the forest. Hierarchical partitioning revealed that distance to disturbances (external edge, internal edges like roads) explained most assemblage variables, whereas forest size and woodland cover within a 1 km radius from the sites explained only a few assemblage variables. Densities of two forest-associated species, Discus rotundatus and Arion fuscus, decreased with forest size. Yet, forest size was positively correlated with richness of typical forest species and densities of Limax cinereoniger. The latter species seems to need forests of >1,000 ha, i.e., well above the size of most fragments. In conclusion, the prediction is valid only for forest species. The response to fragmentation is species specific and seems to depend on habitat specialization and macroclimatic conditions.




Similar content being viewed by others
References
Bailey S (2007) Increasing connectivity in fragmented landscapes: an investigation of evidence for biodiversity gain in woodlands. For Ecol Manage 238:7–23. doi:10.1016/j.foreco.2006.09.049
Berndes G, Hoogwijk M, van den Broek R (2003) The contribution of biomass in the future global energy supply: a review of 17 studies. Biomass Bioenergy 25:1–28. doi:10.1016/S0961-9534(02)00185-X
Brouwers NC, Newton AC (2009) The influence of habitat availability and landscape structure on the distribution of wood cricket (Nemobius sylvestris) on the Isle of Wight, UK. Landscape Ecol 24:199–212. doi:10.1007/s10980-008-9298-4
Bruinderink GG, Van Der Sluis T, Lammertsma D, Opdam P, Pouwels R (2003) Designing a coherent ecological network for large mammals in Northwestern Europe. Conserv Biol 17:549–557. doi:10.1046/j.1523-1739.2003.01137.x
Cameron RAD, Pokryszko BM (2004) Land mollusc faunas of Białowieža forest (Poland) and the character and survival of forest faunas in the north European plain. J Moll Stud 70:149–164. doi:10.1093/mollus/70.2.149
Cameron RAD, Pokryszko BM, Long DC (2006) Snail faunas in southern English calcareous woodlands: rich and uniform, but geographically differentiated. J Conch 39:13–40
Chen J, Franklin JF, Spies TA (1995) Growing season microclimatic gradients from clearcut edges into old Douglas-fir forest. Ecol Appl 5:74–86. doi:10.2307/1942053
Chevan A, Sutherland M (1991) Hierarchical partitioning. Am Stat 45:90–96. doi:10.2307/2684366
Cook WM, Lane KT, Foster BL, Holt RD (2002) Island theory, matrix effects and species richness patterns in habitat fragments. Ecol Lett 5:619–623. doi:10.1046/j.1461-0248.2002.00366.x
Davies-Colley RJ, Payne GW, van Elswijk M (2000) Microclimate gradients across a forest edge. N Z J Ecol 24:111–121
Dörge N, Walther C, Beinlich B, Plachter H (1999) The significance of passive transport for dispersal in terrestrial snails. Z Ökol Tiersch 8:1–10
Dunger W, Fiedler HJ (1997) Methoden der Bodenbiologie. Fischer, Jena
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev Camb Philos Soc 81:117–142. doi:10.1017/S1464793105006949
Falkner G, Ripken TEJ, Falkner M (2002) Mollusques continenteaux de France. Liste de référence annotée et bibliographie. Muséum nationale d’histoire naturelle, Paris
Forys EA, Allen CR, Wojcik DP (2002) Influence of the proximity and amount of human development and roads on the occurrence of the red imported fire ant in the lower Florida keys. Biol Conserv 108:27–33. doi:10.1016/S0006-3207(02)00086-1
Gehlhausen SM, Schwartz MW, Augspurger CK (2000) Vegetation and microclimatic edge effects in two mixed-mesophytic forest fragments. Plant Ecol 147:21–35. doi:10.1023/A:1009846507652
Götmark F, von Proschwitz T, Franc N (2008) Are small sedentary species affected by habitat fragmentation? Local vs. landscape factors predicting species richness and composition of land molluscs in Swedish conservation forests. J Biogeogr 35:1062–1076. doi:10.1111/j.1365-2699.2008.01882.x
Grimbacher PS, Catterall CP, Kitching RL (2006) Beetle species’ responses suggest that microclimate mediates fragmentation effects in tropical Australian rainforest. Austral Ecol 31:458–470. doi:10.1111/j.1442-9993.2006.01606.x
Hakkila P (2006) Factors driving the development of forest energy in Finland. Biomass Bioenergy 30:281–288. doi:10.1016/j.biombioe.2005.07.003
Harrison S, Bruna E (1999) Habitat fragmentation and large-scale conservation: what do we know for sure? Ecography 22:225–232. doi:10.1111/j.1600-0587.1999.tb00496.x
Hewitt G (2000) The genetic legacy of the Quaternary ice ages. Nature 405:907–913. doi:10.1038/35016000
Hilden HD (1988) Geologie am Niederrhein, 4th edn. Geologisches Landesamt Nordrhein-Westfalen, Krefeld
Hylander K, Nilsson C, Jonsson BG, Göthner T (2005) Differences in habitat quality explain nestedness in a land snail meta-community. Oikos 108:351–361. doi:10.1111/j.0030-1299.2005.13400.x
Kappes H (2006) Relations between forest management and slug assemblages (Gastropoda) of deciduous regrowth forests. For Ecol Manage 237:450–457. doi:10.1016/j.foreco.2006.09.067
Kerney MP, Cameron RAD, Jungbluth JH (1983) Die Landschnecken Nord- und Mitteleuropas. Verlag Paul Parey, Hamburg
Keyghobadi N (2007) The genetic implications of habitat fragmentation for animals. Can J Zool 85:1049–1064. doi:10.1139/Z07-095
Koivula MJ, Vermeulen HJW (2005) Highways and forest fragmentation—effects on carabid beetles (Coleoptera, Carabidae). Landscape Ecol 20:911–926. doi:10.1007/s10980-005-7301-x
Kuźnik-Kowalska E (2006) Life cycle and population dynamics of Discus ruderatus (Férussac, 1821) (Gastropoda: Pulmonata: Endodontidae). Folia Malacologica 14:35–46
Kuźnik-Kowalska E, Pokryszko BM (2007) Incipient parental care in Discus—a plesiomorphic state of a truly endodontid character? J Conch 39:467–468
Legendre P, Legendre R (1998) Numerical ecology. Elsevier Scientific, Oxford
Lisický MJ (1991) Mollusca Slovenska. VEDA, Bratislava
Löns H (1891) Die Gastropodenfauna des Münsterlandes. Malakozool Bl NF 11:121–157
López-Barrera F, Armesto JJ, Williams-Linera G, Smith-Ramírez C, Manson RH (2007) Fragmentation and edge effects on plant–animal interactions, ecological processes and biodiversity. In: Newton AC (ed) Biodiversity loss and conservation in fragmented forest landscapes: the forests of montane Mexico and temperate South America. CAB International, Wallingford, pp 69–101
Ložek V (1964) Quartärmollusken der Tschechoslowakei. Tschechoslovakische Akademie der Wissenschaften, Praha
Mac Nally R (1996) Hierarchical partitioning as an interpretative tool in multivariate inference. Aust J Ecol 21:224–228
MacArthur R, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton
MacKenzie DI, Nichols JD, Royle JA, Pollock KP, Bailey LL, Hines JE (2006) Occupancy estimation and modeling: inferring patterns and dynamics of species occurrence. Academic Press, San Diego
Matlack GR (1993) Microenvironment variation within and among forest edge sites in the eastern United States. Biol Conserv 66:185–194. doi:10.1016/0006-3207(93)90004-K
Meyrick RA (2001) The development of terrestrial mollusc faunas in the ‘Rheinland region’ (western Germany and Luxembourg) during the Lateglacial and Holocene. Quat Sci Rev 20:1667–1675. doi:10.1016/S0277-3791(01)00031-2
MURL (1989) Klimaatlas NRW. Ministerium für Umwelt. Raumordnung und Landwirtschaft des Landes Nordrhein-Westfalen, Düsseldorf
Nitare J, Norén M (1992) Nyckelbiotoper kartläggs i nytt projekt vid Skogsstyrelsen. Svensk Bot Tidskr 86:219–226
Noble LR, Jones CS (1996) A molecular and ecological investigation of the large arionid slugs of North-West Europe: the potential for new pests. In: Symondson WOC, Liddell JE (eds) The ecology of agricultural pests. Chapman & Hall, London, pp 93–131
Norén M, Hultgren B, Nitare J, Bergengren I (1995) Instruktion för Datainsamling vid inventering av nyckelbiotoper. National Board of Forestry, Jönköping
Noss RF (2001) Beyond Kyoto: forest management in a time of rapid climate change. Conserv Biol 15:578–590. doi:10.1046/j.1523-1739.2001.015003578.x
Pinceel J, Jordaens K, Van Houtte N, De Winter AJ, Backeljau T (2004) Molecular and morphological data reveal cryptic taxonomic diversity in the terrestrial slug complex Arion subfuscus/fuscus (Mollusca, Pulmonata, Arionidae) in continental north-west Europe. Biol J Linn Soc Lond 83:23–38. doi:10.1111/j.1095-8312.2004.00368.x
Prior DJ (1985) Water-regulatory behaviour in terrestrial gastropods. Biol Rev Camb Philos Soc 60:403–424. doi:10.1111/j.1469-185X.1985.tb00423.x
R Development Core Team (2008) R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. http://www.R-project.org
Ranius T (2000) Minimum viable metapopulation size of a beetle, Osmoderma eremita, living in tree hollows. Anim Conserv 3:37–43. doi:10.1111/j.1469-1795.2000.tb00085.x
Rosenzweig ML (1995) Species diversity in space and time. University of Cambridge Press, Cambridge
Saunders DA, Hobbs RJ, Margules CR (1991) Biological consequences of ecosystem fragmentation: a review. Conserv Biol 5:18–32. doi:10.1111/j.1523-1739.1991.tb00384.x
Shmida A, Wilson MV (1985) Biological determinants in species diversity. J Biogeogr 12:1–12. doi:10.2307/2845026
Stupak I, Asikainen A, Jonsell M et al (2007) Sustainable utilisation of forest biomass for energy—possibilities and problems: policy, legislation, certification, and recommendations and guidelines in the Nordic, Baltic, and other European countries. Biomass Bioenergy 31:666–684. doi:10.1016/j.biombioe.2007.06.012
Tews J, Brose U, Grimm V, Tielboerger K, Wichmann MC, Schwager M, Jeltsch F (2004) Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. J Biogeogr 31:79–92
Thomas CD, Cameron A, Green RE et al (2004) Extinction risk from climate change. Nature 427:145–148. doi:10.1038/nature02121
Tilman D, May RM, Lehman CL, Nowak MA (1994) Habitat destruction and the extinction debt. Nature 371:65–66. doi:10.1038/371065a0
Tilman D, Lehnman CL, Yin C (1997) Habitat destruction, dispersal and deterministic extinction in competitive communities. Am Nat 149:407–435. doi:10.1086/285998
Walsh C, Mac Nally R (2008) hier.part: hierarchical partitioning. R package version 1.0-3
Whittaker RJ (1998) Island biogeography. Ecology, evolution and conservation. Oxford University Press, Oxford
Wiese V (1985) Zur Verbreitungssituation der Land-Nacktschnecken in Schleswig-Holstein (Gastropoda: Arionidae, Milacidae, Limacidae, Agriolimacidae, Boettgerillidae). Faun-Ökol Mitt 5:305–311
Wiklund K (2004) Establishment, growth and population dynamics in two mosses of old-growth forests. Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 996, Uppsala. ISBN: 91-554-6006-2. 47 pp
Acknowledgments
We acknowledge the financial support from the FWO (project G.0202.06). HK received funding by a FWO visiting postdoctoral fellowship, KJ is a postdoctoral fellow of the FWO. This study was conducted within the framework of the European Distributed Institute of Taxonomy (EDIT). We also would like to thank Robert Cameron, Thomas Crist, and an unknown reviewer for their valuable comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jean-Pierre Maelfait: Deceased.
Rights and permissions
About this article
Cite this article
Kappes, H., Jordaens, K., Hendrickx, F. et al. Response of snails and slugs to fragmentation of lowland forests in NW Germany. Landscape Ecol 24, 685–697 (2009). https://doi.org/10.1007/s10980-009-9342-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-009-9342-z