Abstract
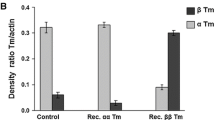
Tropomyosin (TM) plays a central role in calcium mediated striated muscle contraction. There are three muscle TM isoforms: α-TM, β-TM, and γ-TM. α-TM is the predominant cardiac and skeletal muscle isoform. β-TM is expressed in skeletal and embryonic cardiac muscle. γ-TM is expressed in slow-twitch musculature, but is not found in the heart. Our previous work established that muscle TM isoforms confer different physiological properties to the cardiac sarcomere. To determine whether one of these isoforms is dominant in dictating its functional properties, we generated single and double transgenic mice expressing β-TM and/or γ-TM in the heart, in addition to the endogenously expressed α-TM. Results show significant TM protein expression in the βγ-DTG hearts: α-TM: 36%, β-TM: 32%, and γ-TM: 32%. These βγ-DTG mice do not develop pathological abnormalities; however, they exhibit a hyper contractile phenotype with decreased myofilament calcium sensitivity, similar to γ-TM transgenic hearts. Biophysical studies indicate that γ-TM is more rigid than either α-TM or β-TM. This is the first report showing that with approximately equivalent levels of expression within the same tissue, there is a functional dominance of γ-TM over α-TM or β-TM in regulating physiological performance of the striated muscle sarcomere. In addition to the effect expression of γ-TM has on Ca2+ activation of the cardiac myofilaments, our data demonstrates an effect on cooperative activation of the thin filament by strongly bound rigor cross-bridges. This is significant in relation to current ideas on the control mechanism of the steep relation between Ca2+ and tension.







Similar content being viewed by others
References
Brown HR, Schachat FH (1985) Renaturation of skeletal muscle tropomyosin: implications for in vivo assembly. Proc Natl Acad Sci USA 82:2359–2363
Conway JF, Parry DA (1990) Structural features in the heptad substructure and longer range repeats of two-stranded alpha-fibrous proteins. Int J Biol Macromol 12(5):328–334
Corbett MA, Akkari A, Domazetovska A, Cooper ST, North KN, Laing NG, Gunning PW, Hardeman EC (2005) An atropomysoin mutation alters dimer preference in nemaline myopathy. Ann Neurol 57:42–49
Coutu P, Bennett CN, Favre EG, Day SM, Metzger JM (2004) Parvalbumin corrects slowed relaxation in adult cardiac myocytes expressing hypertrophic cardiomyopathy-linked alpha-tropomyosin mutations. Circ Res 94(9):1235–1241
del Monte F, Hajjar RJ (2003) Targeting calcium cycling proteins in heart failure through gene transfer. J Physiol 546(Pt 1):49–61
Denz CR, Narshi A, Zajdel RW, Dube DK (2004) Expression of a novel cardiac-specific tropomyosin isoform in humans. Biochem Biophys Res Commun 320(4):1291–1297
Gimona M (2008) Dimerization of tropomyosins. Adv Exp Med Biol 644:73–84
Jagatheesan G, Rajan S, Petrashevskaya N, Schwartz A, Boivin G, Vahebi S, DeTombe P, Solaro RJ, Labitzke E, Hilliard G, Wieczorek DF (2003) Functional importance of the carboxyl-terminal region of striated muscle tropomyosin. J Biol Chem 278(25):23204–23211
Jagatheesan G, Rajan S, Petrashevskaya N, Schwartz A, Boivin G, Arteaga G, de Tombe PP, Solaro RJ, Wieczorek DF (2004) Physiological significance of troponin t binding domains in striated muscle tropomyosin. Am J Physiol Heart Circ Physiol 287(4):H1484–H1494
Jagatheesan G, Rajan S, Petrashevskaya N, Schwartz A, Boivin G, Arteaga GM, Solaro RJ, Liggett SB, Wieczorek DF (2007) Rescue of tropomyosin-induced familial hypertrophic cardiomyopathy mice by transgenesis. Am J Physiol Heart Circ Physiol 293(2):H949–H958
Jagatheesan G, Rajan S, Schulz EM, Ahmed RP, Petrashevskaya N, Schwartz A, Boivin GP, Arteaga GM, Wang T, Wang YG, Ashraf M, Liggett SB, Lorenz J, Solaro RJ, Wieczorek DF (2009) An internal domain of beta-tropomyosin increases myofilament Ca2+ sensitivity. Am J Physiol Heart Circ Physiol 297(1):H181–H190
Jagatheesan G, Rajan S, Wieczorek DF (2010) Investigations into tropomyosin function using mouse models. J Mol Cell Cardiol 48(5):893–898
Lehrer SS, Qian Y, Hvidt S (1989) Assembly of the native heterodimer of Rana esculanta tropomyosin by chain exchange. Science 246:926–928
Monteiro PB, Lataro RC, Ferro JA, de Castro Reinach F (1994) Functional alpha-tropomyosin produced in escherichia coli. A dipeptide extension can substitute the amino-terminal acetyl group. J Biol Chem 269(14):10461–10466
Muthuchamy M, Pajak L, Howles P, Doetschman T, Wieczorek DF (1993) Developmental analysis of tropomyosin gene expression in embryonic stem cells and mouse embryos. Mol Cell Biol 13(6):3311–3323
Muthuchamy M, Grupp IL, Grupp G, O’Toole BA, Kier AB, Boivin GP, Neumann J, Wieczorek DF (1995) Molecular and physiological effects of overexpressing striated muscle beta-tropomyosin in the adult murine heart. J Biol Chem 270(51):30593–30603
Muthuchamy M, Rethinasamy P, Wieczorek DF (1997) Tropomyosin structure and function: new insights. Trends Cardiovasc Med 7:124–128
Olson TM, Kishimoto NY, Whitby FG, Michels VV (2001) Mutations that alter the surface charge of alpha-tropomyosin are associated with dilated cardiomyopathy. J Mol Cell Cardiol 33(4):723–732
Palmiter KA, Kitada Y, Muthuchamy M, Wieczorek DF, Solaro RJ (1996) Exchange of beta- for alpha-tropomyosin in hearts of transgenic mice induces changes in thin filament response to Ca2+, strong cross-bridge binding, and protein phosphorylation. J Biol Chem 271(20):11611–11614
Patel JR, Fitzsimons DP, Buck SH, Muthuchamy M, Wieczorek DF, Moss RL (2001) Pka accelerates rate of force development in murine skinned myocardium expressing alpha- or beta-tropomyosin. Am J Physiol Heart Circ Physiol 280(6):H2732–H2739
Pena JR, Goldspink PH, Prabhakar R, del Monte F, Hajjar RJ, Wieczorek DF, Wolska BM (2003) Neonatal gene transfer of serca2a improves the response to ß-adrenergic stimulation in the α-tropomyosin (glu180gly) mouse model of familial hypertrophic cardiomyopathy. In: Scientific conference on molecular mechanisms of growth, death, and regeneration in the myocardium: basic biology and insights into ischemic heart disease and heart failure, Snowbird Conference Center, Snowbird, Utah, August 14, 2003. American Heart Association’s Council on Basic Cardiovascular Sciences
Pieples K, Wieczorek DF (2000) Tropomyosin 3 increases striated muscle isoform diversity. Biochemistry 39(28):8291–8297
Pieples K, Arteaga G, Solaro RJ, Grupp I, Lorenz JN, Boivin GP, Jagatheesan G, Labitzke E, DeTombe PP, Konhilas JP, Irving TC, Wieczorek DF (2002) Tropomyosin 3 expression leads to hypercontractility and attenuates myofilament length-dependent Ca(2+) activation. Am J Physiol Heart Circ Physiol 283(4):H1344–H1353
Rajan S, Ahmed RP, Jagatheesan G, Petrashevskaya N, Boivin GP, Urboniene D, Arteaga GM, Wolska BM, Solaro RJ, Liggett SB, Wieczorek DF (2007) Dilated cardiomyopathy mutant tropomyosin mice develop cardiac dysfunction with significantly decreased fractional shortening and myofilament calcium sensitivity. Circ Res 101(2):205–214
Rajan S, Jagatheesan G, Karam CN, Alves ML, Bodi I, Schwartz A, Bulcao CF, D’Souza KM, Akhter SA, Boivin GP, Dube DK, Petrashevskaya N, Herr AB, Hullin R, Liggett SB, Wolska BM, Solaro RJ, Wieczorek DF (2010) Molecular and functional characterization of a novel cardiac-specific human tropomyosin isoform. Circulation 121(3):410–418
Solaro RJ (2009) Maintaining cooperation among cardiac myofilament proteins through thick and thin. J Physiol 587(Pt 1):3
Sun YB, Lou F, Irving M (2009) Calcium- and myosin-dependent changes in troponin structure during activation of heart muscle. J Physiol 587(Pt 1):155–163
Vrhovski B, Theze N, Thiebaud P (2008) Structure and evolution of tropomyosin genes. Adv Exp Med Biol 644:6–26
Wieczorek DF, Jagatheesan G, Rajan S (2008) The role of tropomyosin in heart disease. Adv Exp Med Biol 644:132–142
Wolska BM, Keller RS, Evans CC, Palmiter KA, Phillips RM, Muthuchamy M, Oehlenschlager J, Wieczorek DF, de Tombe PP, Solaro RJ (1999) Correlation between myofilament response to Ca2+ and altered dynamics of contraction and relaxation in transgenic cardiac cells that express beta-tropomyosin. Circ Res 84(7):745–751
Acknowledgments
We thank Jon Neumann for production of the transgenic mice and Maureen Bender for her care of the animals. This work was supported in part by National Institutes of Health Grants HL71952 and HL081680 awarded to DFW, HL22231 and HL 62426 awarded to RJS, K01 HL67709 awarded to GMA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jagatheesan, G., Rajan, S., Ahmed, R.P.H. et al. Striated muscle tropomyosin isoforms differentially regulate cardiac performance and myofilament calcium sensitivity. J Muscle Res Cell Motil 31, 227–239 (2010). https://doi.org/10.1007/s10974-010-9228-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-010-9228-3