Abstract
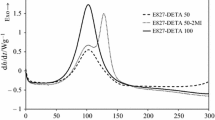
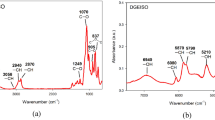
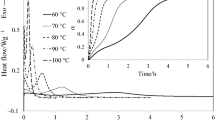
In the present work, we report the preparation and characterization of a new family of thermosets based on off-stoichiometric anhydride–epoxy formulations in the presence of an anionic initiator. Diglycidyl ether of bisphenol A (DGEBA) and hexahydro-4-methylphthalic anhydride (HHMPA) have been used as epoxy and anhydride comonomers, respectively, and 1-methylimidazole (1MI) has been used as anionic initiator. The isothermal curing kinetics and the thermal properties of the stoichiometric and the off-stoichiometric systems have been compared. The kinetics of the isothermal curing has been analyzed by differential scanning calorimetry (DSC) using an isoconversional method and the Šesták–Berggren equation to determine the activation energy, the frequency factor and the reaction orders. The materials obtained were characterized by DSC and dynamic mechanical analysis. Gelation during epoxy–anhydride condensation was determined by thermomechanical analysis. At the same curing temperature, the reaction is faster in the system with excess of epoxy groups. However, the glass transition temperatures of the partially cured stoichiometric system are greater. The gelation time of the off-stoichiometric system is shorter than that of the stoichiometric one. The results indicate that the dual-curing character of off-stoichiometric DGEBA/HHMPA thermosets with 1MI as anionic initiator makes them suitable for multistage curing processes with easy control of degree of cure and material properties in the intermediate stage and enhanced final material properties.







Similar content being viewed by others
References
Riew CK, Siebert AR, Smith RW, Fernando M, Kinloch AJ. Toughened epoxy resins: performed particles as tougheners for adhesives and matrices. In: Riew CK, Kinloch AJ, editors. Toughened plastics II novel approaches in science and engineering, vol. 252., Advances in chemical seriesWashington: American Chemical Society; 1996. p. 33–44.
Saiki N, Yamazaki O, Ebe K. UV/heat dual-curable adhesive tapes for fabricating stacked packages of semiconductors. J Appl Polym Sci. 2008;108:1178–83.
Kang B-U. Interfacial fracture behavior of epoxy adhesives for electronic components. J Korea Acad Ind Cooper Soc. 2011;12:1479–87.
May CA, Tanaka GY. Epoxy resins. In: May CA, editor. Chemistry and technology, chap 1. New York: Marcel Dekker; 1988.
Petrie EM. Epoxy adhesive formulations. New York: McGraw-Hill; 2006.
Pascault JP, Williams RJJ. Epoxy polymers: new materials and innovations. Weinheim: Wiley-VCH; 2010.
Kinloch AJ, Shaw SJ, Tod DA, Hunston DL. Deformation and fracture behavior of a rubber-toughened epoxy: 1. Microstructure and fracture studies. Polymer. 1983;24:1341–54.
Ho T-H, Wang C-S. Toughening of epoxy resins by modification with dispersed acrylate rubber for electronic packaging. J Appl Polym Sci. 1993;50:477–83.
Mezzenga R, Boogh L, Månson JAE. A review of dendritic hyperbranched polymer as modifiers in epoxy composites. Compos Sci Technol. 2001;61:787–95.
Guo QP, Habrard A, Park Y, Halley PJ, Simon GP. Phase separation, porous structure, and cure kinetics in aliphatic epoxy resin containing hyperbranched polyester. J Polym Sci B. 2006;44:889–99.
Ratna D, Varley R, Simon GP. Toughening of trifunctional epoxy using an epoxy-functionalized hyperbranched polymer. J Appl Polym Sci. 2003;89:2339–45.
He S, Shi K, Bai J, Zhang Z, Li L, Du Z, Zhang B. Studies on the properties of epoxy resins modified with chain-extended ureas. Polymer. 2001;42:9641–7.
Zhou L, Zhang G, Li J, Jing Z, Qin J, Feng Y. The flame retardancy and thermal stability properties of flame-retarded epoxy resins based on α-hydroxyphosphonate cyclotriphosphazene. J Therm Anal Calorim. 2017;129:1667–78.
Mao W, Li S, Yang X, Cao S, Li M, Huang K, Xia J. Preparation of a flame-retardant epoxy curing agent based on castor oil and study on the curing reaction kinetics. J Therm Anal Calorim. 2017;130:2113–21.
Evans D, Canfer SJ. Radiation stable, low viscosity impregnating resin systems for cryogenic applications. Adv Cryog Eng. 2000;46:361–8.
Ueki T, Nishijima S, Izumi Y. Designing of epoxy resin systems for cryogenic use. Cryogenics. 2005;45:141–8.
Nishijima S, Honda Y, Okada T. Application of the positron annihilation method for evaluation of organic materials for cryogenic use. Cryogenics. 1995;35:779–81.
Pascault JP, Sautereau H, Verdu J, Williams RJJ. Thermosetting polymers. 1st ed. New York: Marcel Dekker, Inc.; 2002.
Thanki JD, Parsania PH. Dynamic DSC curing kinetics and thermogravimetric study of epoxy resin of 9,9′-bis-(4-hydroxyphenyl)anthrone-10. J Therm Anal Calorim. 2017;130:2145–56.
Foix D, Yu Y, Serra À, Ramis X, Salla JM. Study on the chemical modification of epoxy/anhydride thermosets using a hydroxyl terminated hyperbranched polymer. Eur Polym J. 2009;45:1454–66.
Rocks J, Rintoul L, Vohwinkel F, George G. The kinetics and mechanism of cure of an amino-glycidyl epoxy resin by a co-anhydride as studied by FT-Raman spectroscopy. Polymer. 2004;45:6799–811.
Fernández-Francos X, Santiago D, Ferrando F, Ramis X, Salla JM, Serra À, Sangermano M. Network structure and thermomechanical properties of hybrid DGEBA networks cured with 1-methylimidazole and hyperbranched poly(ethyleneimine)s. J Polym Sci Part B Polym Phys. 2012;50:1489–503.
Leena K, Soumyamol PB, Baby M, Suraj S, Rajeev R, Mohan DS. Non-isothermal cure and decomposition kinetics of epoxy–imidazole systems. J Therm Anal Calorim. 2017;130:1053–61.
Ramis X, Fernández-Francos X, De La Flor S, Ferrando F, Serra À. Click-based dual-curing thermosets and their applications. In: Guo Q, editor. Thermosets: structure, properties and applications, chapter 16. 2nd ed. Amsterdam: Elsevier; 2017.
Fernández-Francos X, Konuray AO, Belmonte A, De la Flor S, Serra À, Ramis X. Sequential curing of off-stoichiometric thiol-epoxy thermosets with a custom-tailored structure. Polym Chem. 2016;7:2280–90.
Konuray O, Areny N, Morancho JM, Fernández-Francos X, Serra À, Ramis X. Preparation and characterization of dual-curable off-stoichiometric amine-epoxy thermosets with latent reactivity. Polymer. 2018;146:42–52.
Vyazovkin S, Burnham AK, Criado JM, Pérez-Maqueda LA, Popescu C, Sbirrazzuoli N. ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta. 2011;520:1–19.
Heise MS, Martin GC. Curing mechanism and thermal properties of epoxy-imidazole systems. Macromolecules. 1989;22:99–104.
Flores M, Fernández-Francos X, Ramis X, Serra À. Novel epoxy-anhydride thermosets modified with a hyperbranched polyester as toughness enhancer. Thermochim Acta. 2012;544:17–26.
Ivin KJ. Polymer handbook. Brandrup J, Immergut EH, editors. New York: Wiley; 1975
Montserrat S, Flaqué C, Calafell M, Andreu G, Málek J. Influence of the accelerator concentration on the curing reaction of an epoxy-anhydride system. Thermochim Acta. 1995;269(270):213–29.
Santiago D, Fernández-Francos X, Ramis X, Salla JM, Sangermano M. Comparative curing kinetics and thermal-mechanical properties of DGEBA thermosets cured with a hyperbranched poly(ethylenimine) and an aliphatic triamine. Thermochim Acta. 2011;526:9–21.
Morancho JM, Ramis X, Fernández-Francos X, Salla JM, Konuray AO, Serra À. Curing of off-stoichiometric amine-epoxy thermosets. J Therm Anal Calorim. 2018;133:519–27.
Morancho JM, Salla JM. Influence of a carboxyl-terminated modifier (CTBN) on the cure of an epoxy resin. J Non Cryst Solids. 1994;172–174:656–60.
Fernàndez-Francos X, Ramis X, Serra À. From curing kinetics to network structure: a novel approach to the modeling of the network buildup of epoxy-anhydride thermosets. J Polym Sci Part A Polym Chem. 2014;52:61–75.
Tanaka Y, Stanford JL, Stepto R. Interpretation of gel points of an epoxy-amine system including ring formation and unequal reactivity: measurements of gel points and analyses on ring structures. Macromolecules. 2012;45:7197–205.
Mauri AN, Galego N, Riccardi CC, Williams RJJ. Kinetic model for gelation in the diepoxide–cyclic anhydride copolymerization initiated by tertiary amines. Macromolecules. 1997;30:1616–20.
Acknowledgements
The authors would like to thank MINECO (Ministerio de Economía y Competividad) and FEDER (Fondo Europeo de Desarrollo Regional) (MAT2017-82849-C2-1-R and MAT2017-82849-C2-2-R) and Generalitat de Catalunya (2017-SGR-77 and Serra Húnter program) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Morancho, J.M., Ramis, X., Fernández-Francos, X. et al. Curing and thermomechanical properties of off-stoichiometric anhydride–epoxy thermosets. J Therm Anal Calorim 138, 2865–2872 (2019). https://doi.org/10.1007/s10973-019-08681-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-019-08681-z