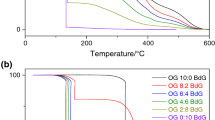
Abstract
This work aims to determine if thermogravimetry could be used to determine the degree of conversion of soybean oil to biodiesel without knowing the amount of ester in the sample, comparing the results with the values obtained by the standard method, gas chromatography. The syntheses were made using homogeneous and heterogeneous catalysts, and the measures were made in triplicate on both pieces of equipment. The average values (%) obtained were 74.59 ± 0.58, 66.5 ± 1.62, 13.54 ± 0.32 by thermogravimetry and 74.09 ± 0.46, 62.08 ± 2.19, 17.07 ± 0.31 by gas chromatography. These data were statistically compared using F-test, and the result indicated that the thermogravimetry could be used as analytical technique to determine the degree of conversion of biodiesel, with the same reliability as gas chromatography. The thermogravimetry shows more advantages than gas chromatography, because it does not require organic solvent, following the Green Chemistry Principles, and it is already used to determine other thermal properties of biodiesel.





Similar content being viewed by others
References
Knothe G, Van Gerpen J, Krahl J, Ramos LP. Biodiesel manual. São Paulo: Edgard Blücher; 2006.
Baird C, Cann M. Environmental chemistry. 4th ed. New York: W. F. Freeman and Company; 2008.
OECD/Food and Agriculture Organization of the United Nations. OECD-FAO Agricultural outlook 2015, OECD publishing; 2015.
ASSOCIAÇÃO BRASILEIRA DE NORMAS TÉCNICAS. NBR 15764: Biodiesel—Determinação do teor total de ésteres por cromatografia gasosa; 2015.
DIN EN14103 Fat and oil derivatives- Fatty acid methyl esters (FAME)—Determination of ester and linolenic acid methyl esters content. Brussels: European Commitee for Standartization, Management Centre; 2011.
Henderson RK, Jímenez-González C, Constable DJC, Alston SR, Inglis GGA, Fisher G, Sherwood J, Binks SP, Curzons AD. Expanding GSK’s solvent selection guide—embedding sustainability into solvent selection starting at medicinal chemistry. Green Chem. 2011;13:854–62.
Alfonsi K, Colberg J, Dunn PT, Fevig T, Jennings S, Johnson TA, Kleine HP, Knight C, Nagy MA, Perry DA, Stefaniak M. Green chemistry tools to influence a medicinal chemistry and research chemistry based organization. Green Chem. 2008;10:31–6.
Jain S, Sharma MP. Application of Thermogravimetric analysis for thermal stability of Jatropha Curcas biodiesel. Fuel. 2012;93:252–7.
Chien YC, Lu M, Chai M, Boreo FJ. Characterization of biodiesel and biodiesel particulate matter by TG, TG–MS and FTIR. Energy Fuels. 2009;23:202–6.
Dantas MB, Albuquerque AR, Barros AK, Rodrigues-Filho MG, Antoniosi-Filho NR, Sinfrônio FSM, Rosenhaim R, Soledade LEB, Santos IMG, Souza AG. Evaluation of the oxidative stability of corn biodiesel. Fuel. 2011;90:773–8.
Santos AGD, Souza LD, Caldeira VPS, Farias MF, Fernandes VJ Jr, Araújo AS. Kinetic study and thermoxidative degradation of palm oil biodiesel. Thermochim Acta. 2014;592:18–22.
Conceição MM, Fernandes VJ Jr, Araújo AS, Farias MF, Santos IMG, Souza AG. Thermal and oxidative degradation of castor oil biodiesel. Energy Fuels. 2007;21:1522–7.
Zhao H, Cao Y, Orndorff W, Cheng YH, Pan WP. Thermal behaviors of soy biodiesel. J Therm Anal Calorim. 2012;109:1145–50.
Jain S, Sharma MP. Thermal stability of biodiesel and its blends: a review. Renew Sustain Energy Rev. 2011;15:438–48.
Santos NA, Tavares MLA, Rosenhaim R, Fernandes Silva FC, Jr VJ, Santos IMG, Souza AG. Thermogravimetric and calorimetric evaluation of babassu biodiesel obtained by the methanol route. J Therm Anal Calorim. 2007;87:649–52.
Lima JRO, da Silva RB, de Moura EM, de Moura CVR. Biodiesel of tucum oil, synthesized by methanolic and ethanolic routes. Fuel. 2008;87:1718–23.
Chand P, Reddy CV, Verkade JG, Wang T, Grewell D. Termogravimetric quantification of biodiesel produced via alkali catalyzed transesterification of soybean oil. Energy Fuels. 2009;23:989–92.
Souza FP, Luciano MA, Pasa VMD. Termogravimetry and viscometry for assessing the ester content (FAME and FAEE). Fuel Process Technol. 2013;109:133–40.
Gaglieri C, Caires FJ, da Silva DJ, Bertholdo R, Mendes RA. Synthesis, thermal behaviour and catalytic study of Ni-chitosan in transesterification reaction using soybean oil. Braz J Therm Anal. 2017;6:1–6.
Hartman L, Lago RCA. Rapid preparation of fatty acid methyl esters from lipids. Lab Pract. 1973;22:475–6.
Menezes ML, Lopes LFS, Passaretti Filho J. Síntese de biodiesel empregando óleo de abacate. Rev Anal. 2010;44:68–78.
Miller JN, Miller JC. Statistics and chemometrics for analytical chemistry. 6th ed. Harlow: Pearson Education Limited; 2010.
Acknowledgements
The authors wish to thank CAPES (Proc. 024/2012 Pro-equipment), CNPq (Proc. No. 421469/2016-1), POSMAT/UNESP and FAPESP (Proc. No. 2017/14936-9 and No. 2017/08820-8) for financial support and the Ph.D. Manoel Lima de Menezes for help.
Author information
Authors and Affiliations
Corresponding author
Additional information
The present article is based on the lecture presented at SiAT VIII conference in Punta Grossa - Brazil on August 13-15, 2017.
Rights and permissions
About this article
Cite this article
Gaglieri, C., Alarcon, R.T., de Moura, A. et al. Is Thermogravimetry an efficient alternative to gas chromatography in degree of biodiesel conversion?. J Therm Anal Calorim 135, 2591–2597 (2019). https://doi.org/10.1007/s10973-018-7364-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-018-7364-y