Abstract
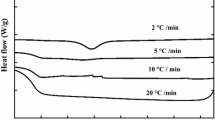
Polyvinyl alcohol (PVA) is a hydrophilic, biodegradable, semicrystalline polymer with a wide array of commercial uses ranging from textiles and packaging to medicine. Film samples of PVA were investigated to assess crystallization and melting behavior during self-nucleation experiments and thermal degradation, using differential scanning calorimetry (DSC) and thermogravimetric (TG) analysis, respectively. TG results show that degradation occurred at temperatures in excess of 200 °C which is close to the observed peak melting temperature of 223 °C. PVA was heated to various self-nucleation temperatures, T s, within its melting range, and then cooled and reheated. Three distinct crystallization regimes were observed upon cooling, depending upon the self-nucleation temperature (T s) selected. At low values of T s, T s < 227 °C, PVA only partially melts, and upon cooling the residual crystals anneal and become more stable. At intermediate values of T s, 228 °C < T s < 234 °C, PVA was found to crystallize exclusively from self-nucleation. For T s > 235 °C, the PVA melts completely and absence of self-nucleation sites causes crystallization to occur at lower temperatures.







Similar content being viewed by others
References
Kane TG, Robinson WD, inventors; Polyvinyl alcohol adhesives. United States 1972.
Sumi M, Suenaga J, Takenaka M, Murano I, Tanabe M, Hirai K, inventors; Cold-set adhesive compositions comprising polyvinyl alcohol for paper and paper board. United States 1984.
Wolff EA, inventor; Method of making a waterproof polyvinyl alcohol coated paper. United States 1970.
Han JH. Chapter 4—Antimicrobial food packaging. In: Ahvenainen R, editor. Novel food packaging techniques. Cambridge: Woodhead Publishing; 2003. p. 50–70.
Musetti A, Paderni K, Fabbri P, Pulvirenti A, Al-Moghazy M, Fava P. Poly(vinyl alcohol)-based film potentially suitable for antimicrobial packaging applications. J Food Sci. 2014;79(4):E577–82.
de Azeredo HMC. Nanocomposites for food packaging applications. Food Res Int. 2009;42(9):1240–53.
Chiellini E, Corti A, D’Antone S, Solaro R. Biodegradation of poly (vinyl alcohol) based materials. Prog Polym Sci. 2003;28(6):963–1014.
Huang H, Gu L, Ozaki Y. Non-isothermal crystallization and thermal transitions of a biodegradable, partially hydrolyzed poly(vinyl alcohol). Polymer. 2006;47(11):3935–45.
Arvanitoyannis I, Kolokuris I, Nakayama A, Yamamoto N, Aiba S-I. Physico-chemical studies of chitosan-poly(vinyl alcohol) blends plasticized with sorbitol and sucrose. Carbohydr Polym. 1997;34(1–2):9–19.
Zeng J, Aigner A, Czubayko F, Kissel T, Wendorff JH, Greiner A. Poly(vinyl alcohol) nanofibers by electrospinning as a protein delivery system and the retardation of enzyme release by additional polymer coatings. Biomacromolecules. 2005;6(3):1484–8.
Kenawy E-R, Abdel-Hay FI, El-Newehy MH, Wnek GE. Controlled release of ketoprofen from electrospun poly(vinyl alcohol) nanofibers. Mater Sci Eng A. 2007;459(1–2):390–6.
Khanna PK, Singh N, Charan S, Subbarao VVVS, Gokhale R, Mulik UP. Synthesis and characterization of Ag/PVA nanocomposite by chemical reduction method. Mater Chem Phys. 2005;93(1):117–21.
Hong KH. Preparation and properties of electrospun poly(vinyl alcohol)/silver fiber web as wound dressings. Polym Eng Sci. 2007;47(1):43–9.
Zhou Y, Yang D, Chen X, Xu Q, Lu F, Nie J. Electrospun water-soluble carboxyethyl chitosan/poly(vinyl alcohol) nanofibrous membrane as potential wound dressing for skin regeneration. Biomacromolecules. 2008;9(1):349–54.
Vaidya S, Tozer KR, Chen J. An overview of embolic agents. Semin Interv Radiol. 2008;25(03):204–15.
Baker MI, Walsh SP, Schwartz Z, Boyan BD. A review of polyvinyl alcohol and its uses in cartilage and orthopedic applications. J Biomed Mater Res B. 2012;100B(5):1451–7.
Hyon S-H, Cha W-I, Ikada Y, Kita M, Ogura Y, Honda Y. Poly(vinyl alcohol) hydrogels as soft contact lens material. J Biomater Sci Polym E. 1994;5(5):397–406.
Li CP, Vongsvivut J, She XD, Li YZ, She FH, Kong LX. New insight into non-isothermal crystallization of PVA-graphene composites. Phys Chem Chem Phys. 2014;16(40):22145–58.
Li CP, Hou TT, Vongsvivut J, Li YZ, She XD, She FH, et al. Simultaneous crystallization and decomposition of PVA/MMT composites during non-isothermal process. Thermochim Acta. 2015;618:26–35.
Li C, She M, She X, Dai J, Kong L. Functionalization of polyvinyl alcohol hydrogels with graphene oxide for potential dye removal. J Appl Polym Sci. 2014;131(3):39872.
Li CP, Hou TT, She XD, Wei XY, She FH, Gao WM, et al. Decomposition properties of PVA/graphene composites during melting-crystallization. Polym Degrad Stab. 2015;119:178–89.
Probst O, Moore EM, Resasco DE, Grady BP. Nucleation of polyvinyl alcohol crystallization by single-walled carbon nanotubes. Polymer. 2004;45(13):4437–43.
Holland BJ, Hay JN. The thermal degradation of poly(vinyl alcohol). Polymer. 2001;42(16):6775–83.
Endo R, Amiya S, Hikosaka M. Conditions for melt crystallization without thermal degradation and equilibrium melting temperature of atactic poly(Vinyl Alcohol). J Macromol Sci B. 2003;42(3–4):793–820.
Tubbs RK. Melting point and heat of fusion of poly(vinyl alcohol). J Polym Sci Part A. 1965;3(12):4181–9.
Peppas NA, Hansen PJ. Crystallization kinetics of poly(vinyl alcohol). J Appl Polym Sci. 1982;27(12):4787–97.
Peppas NA, Merrill EW. Differential scanning calorimetry of crystallized PVA hydrogels. J Appl Polym Sci. 1976;20(6):1457–65.
Wunderlich B. Macromolecular physics, volume 3—crystal melting. New York: Academic Press; 1980.
Koski A, Yim K, Shivkumar S. Effect of molecular weight on fibrous PVA produced by electrospinning. Mater Lett. 2004;58(3–4):493–7.
Minoo N, Tong L, Mark PS, Liming D, Xungai W. Electrospun single-walled carbon nanotube/polyvinyl alcohol composite nanofibers: structure–property relationships. Nanotechnology. 2008;19(30):305702.
Azuma Y, Yoshie N, Sakurai M, Inoue Y, Chûjô R. Thermal behaviour and miscibility of poly(3-hydroxybutyrate)/poly(vinyl alcohol) blends. Polymer. 1992;33(22):4763–7.
El-Zaher NA, Osiris WG. Thermal and structural properties of poly(vinyl alcohol) doped with hydroxypropyl cellulose. J Appl Polym Sci. 2005;96(5):1914–23.
Guirguis OW, Moselhey MTH. Thermal and structural studies of poly (vinyl alcohol) and hydroxypropyl cellulose blends. Nat Sci. 2012;04(01):11.
Jang J, Lee DK. Plasticizer effect on the melting and crystallization behavior of polyvinyl alcohol. Polymer. 2003;44(26):8139–46.
Minus ML, Chae HG, Kumar S. Single wall carbon nanotube templated oriented crystallization of poly(vinyl alcohol). Polymer. 2006;47(11):3705–10.
Fillon B, Lotz B, Thierry A, Wittmann JC. Self-nucleation and enhanced nucleation of polymers—definition of a convenient calorimetric efficiency scale and evaluation of nucleating additives in isotactic polypropylene (alpha-phase). J Polym Sci Pol Phys. 1993;31(10):1395–405.
Fillon B, Thierry A, Wittmann JC, Lotz B. Self-nucleation and recrystallization of polymers—isotactic polypropylene, beta-phase–beta-alpha conversion and beta-alpha-growth transitions. J Polym Sci Pol Phys. 1993;31(10):1407–24.
Fillon B, Wittmann JC, Lotz B, Thierry A. Self-nucleation and recrystallization of isotactic polypropylene (alpha-phase) investigated by differential scanning calorimetry. J Polym Sci Pol Phys. 1993;31(10):1383–93.
Müller AJ, Arnal ML. Thermal fractionation of polymers. Prog Polym Sci. 2005;30(5):559–603.
Lorenzo AT, Arnal ML, Sánchez JJ, Müller AJ. Effect of annealing time on the self-nucleation behavior of semicrystalline polymers. J Polym Sci Pol Phys. 2006;44(12):1738–50.
Mamun A, Umemoto S, Okui N, Ishihara N. Self-seeding effect on primary nucleation of isotactic polystyrene. Macromolecules. 2007;40(17):6296–303.
Arandia I, Mugica A, Zubitur M, Arbe A, Liu G, Wang D, et al. How composition determines the properties of isodimorphic poly(butylene succinate-ran-butylene azelate) random biobased copolymers: from single to double crystalline random copolymers. Macromolecules. 2015;48(1):43–57.
Jalali A, Huneault MA, Elkoun S. Effect of thermal history on nucleation and crystallization of poly(lactic acid). J Mater Sci. 2016;51(16):7768–79.
Ren MQ, Chen XJ, Sang Y, Alamo RG. Effect of heterogeneous short chain branching distribution on acceleration or retardation of the rate of crystallization from melts of ethylene copolymers synthesized with ziegler-natta catalysts. Macromol Symp. 2015;356(1):131–41.
Chen XJ, Mamun A, Alamo RG. Effect of level of crystallinity on melt memory above the equilibrium melting temperature in a random ethylene 1-butene copolymer. Macromol Chem Phys. 2015;216(11):1220–6.
Horváth Z, Menyhárd A, Doshev P, Gahleitner M, Varga J, Tranninger C, et al. Chain regularity of isotactic polypropylene determined by different thermal fractionation methods. J Therm Anal Calorim. 2014;118(1):235–45.
Reid BO, Vadlamudi M, Mamun A, Janani H, Gao HH, Hu WB, et al. Strong memory effect of crystallization above the equilibrium melting point of random copolymers. Macromolecules. 2013;46(16):6485–97.
Chau J, Teh J. Successive self-nucleation and annealing in the solvated state of ethylene copolymers. J Therm Anal Calorim. 2005;81(1):217–23.
Marco C, Ellis G, Gómez MA, Arribas JM. Analysis of the dynamic crystallisation of isotactic polypropylene/α-nucleating agent systems by DSC. J Therm Anal Calorim. 2002;68(1):61–74.
Arnal ML, Balsamo V, Ronca G, Sánchez A, Müller AJ, Cañizales E, et al. Applications of successive self-nucleation and annealing (SSA) to polymer characterization. J Therm Anal Calorim. 2000;59(1):451–70.
Gray AP. Polymer crystallinity determinations by DSC. Thermochim Acta. 1970;1(6):563–79.
Pyda M. Polyvinyl alcohol (PVA) heat capacity, enthalpy, entropy, gibbs energy. In: Advanced thermal analysis system (ATHAS) database. Springer-Verlag GmbH, SpringerMaterials. 2014. http://materials.springer.com/polymerthermodynamics/docs/athas_0147. Accessed 27 Oct 2015.
MATLAB R2015b. Version 8.6.0 ed. Natick: The Mathworks, Inc; 2015.
Cebe P, Hu X, Kaplan DL, Zhuravlev E, Wurm A, Arbeiter D, Schick C. Beating the heat—fast scanning melts silk beta sheet crystals. Sci Rep. 2013;3:7.
Cebe P, Partlow BP, Kaplan DL, Wurm A, Zhuravlev E, Schick C. Using flash DSC for determining the liquid state heat capacity of silk fibroin. Thermochim Acta. 2015;615:8–14.
Cebe P, Partlow BP, Kaplan D, Wurm A, Zhuravlev E, Schick C. Chapter 5: Fast scanning calorimetry of silk fibroin protein: sample mass and specific heat capacity determination. In: Schick C, Mathot VBF, editors. Fast scanning methods. New York: Springer International Publishing; 2016. p. 187–203.
Mileva D, Androsch R, Zhuravlev E, Schick C, Wunderlich B. Homogeneous nucleation and mesophase formation in glassy isotactic polypropylene. Polymer. 2012;53(2):277–82.
Androsch R, Schick C, Schmelzer JWP. Sequence of enthalpy relaxation, homogeneous crystal nucleation and crystal growth in glassy polyamide 6. Eur Polym J. 2014;53:100–8.
Acknowledgements
Support for this research was provided by the National Science Foundation, Polymers Program of the Division of Materials Research, under DMR-1206010, and through the MRI Program under DMR-0520655 which provided thermal analysis instrumentation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thomas, D., Cebe, P. Self-nucleation and crystallization of polyvinyl alcohol. J Therm Anal Calorim 127, 885–894 (2017). https://doi.org/10.1007/s10973-016-5811-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5811-1