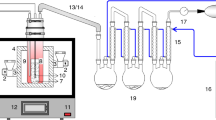
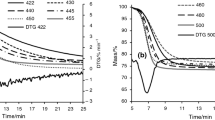
Abstract
Factors influencing the pyrolysis process of Longkou oil shale, such as nitrogen flow rate, pyrolysis temperature, particle size, and heating rate, were investigated in order to optimize the pyrolysis condition and reduce the secondary reactions. Oil yield reached the maximum value, 17.46 %, higher than Fischer assay oil yield, at heating rate 15 °C min−1 up to 550 °C under 0.6 L min−1 nitrogen flow rate for 2- to 3-mm oil shale particles. Compared with the retorting gas obtained from Fischer assay, the retorting gas under optimized condition had a higher organic/inorganic gas ratio and alkanes/alkenes (C1–C5), due to the inhibition of the gas cracking or secondary gas phase reactions. And the shale oil obtained from optimized condition had a lower IBP and higher distilled mass percent when reaching the same end temperature, 750 °C. The content of total alkanes and alkenes in the shale oil under optimized condition was 401 and 181 mg g −1oil , which were higher than those from Fischer assay, but the C8–C10 alkenes were lower, proving that optimizing pyrolysis condition was beneficial to reducing the secondary reactions.







Similar content being viewed by others
References
Jiang XM, Han XX, Cui ZG. New technology for the comprehensive utilization of Chinese oil shale resources. Energy. 2007;32(5):772–7.
Kok MV, Senguler I. Geological and thermal characterization of Eskisehir region oil shales. J Therm Anal Calorim. 2014;116:367–72.
Ma Y, He L, Li SY, Teng JS. Heat transfer of oil shale in a small-scale fixed bed. J Therm Anal Calorim. 2016;124:461–9.
Jiang XM, Han XX, Cui ZG. Progress and recent utilization trends in combustion of Chinese oil shale. Prog Energy Combust Sci. 2007;33:552–79.
Tiwari P, Deo M. Compositional and kinetic analysis of oil shale pyrolysis using TGA–MS. Fuel. 2012;94(1):333–41.
Wang W, Li SY, Yue CT, Ma Y. Multistep pyrolysis kinetics of North Korean oil shale. J Therm Anal Calorim. 2015;119:643–9.
Williams PT, Ahmad N. Investigation of oil-shale pyrolysis processing conditions using thermo gravimetric analysis. Appl Energy. 2000;66(2):113–33.
Li SY, Yue CT. Study of pyrolysis kinetics of oil shale. Fuel. 2003;82:337–42.
Liu QQ, Han XX, Li QY, Huang YR, Jian XM. TG–DSC analysis of pyrolysis process of two Chinese oil shales. J Therm Anal Calorim. 2014;116:511–7.
Al-Ayed OS, Matouq M, Anbar Z, Khaleela AM, Abu-Nameh E. Oil shale pyrolysis kinetics and variable activation energy principle. Appl Energy. 2010;87(4):1269–72.
Johannes I, Kruusement K, Veski R. Evaluation of oil potential and pyrolysis kinetics of renewable fuel and shale samples by Rock-Eval analyzer. J Anal Appl Pyrol. 2007;79(1–2):183–90.
Wang S, Liu JX, Jiang XM, Han XX, Tong JH. Effect of heating rate on products yield and characteristics of non-condensable gases and shale oil obtained by retorting Dachengzi oil shale. Oil Shale. 2013;30(1):27–47.
Wang S, Jiang XM, Han XX, Tong JH. Effect of residence time on products yield and characteristics of shale oil and gases products by low-temperature retorting of Dachengzi oil shale. Oil Shale. 2013;30(1):27–47.
Wang S, Jiang XM, Han XX, Tong JH. Effect of retorting temperature on product yield and characteristics of non-condensable gases and shale oil obtained by retorting Huadian oil shales. Fuel Process Technol. 2014;121:9–15.
Jeong GN, Cheol HI, Soo HC. Effect of oil shale retorting temperature on shale oil yield and properties. Fuel. 2012;95(1):131–5.
Dung NV. Factors affecting product yields and oil quality during retorting of Stuart oil shale with recycled shale: a screening study. Fuel. 1995;74(4):623–7.
Jaber JO, Probert SD. Non-isothermal thermogravimetry and decomposition kinetics of two Jordanian oil shales under different processing conditions. Fuel Process Technol. 2000;63(1):57–70.
Yue CT, Liu Y, Ma Y, Li SY, He JL, Qiu DK. Influence of retorting conditions on the pyrolysis of Yaojie oil shale. Oil Shale. 2014;31(1):66–78.
Nazzal JM. Influence of heating rate on the pyrolysis of Jordan oil shale. J Anal Appl Pyrol. 2002;62(2):225–38.
Campbell JH, Koskinas GJ, Gallegos G, Gregg M. Gas evolution during oil shale pyrolysis. 1. Nonisothermal rate measurements. Fuel. 1980;59(10):718–26.
Campbell JH, Gallegos G, Gregg M. Gas evolution during oil shale pyrolysis. 2. Kinetic and stoichiometric analysis. Fuel. 1980;59(10):727–32.
Al-Ayed OS, Al-Harahsheh A, Khaleel AM. Oil shale pyrolysis in fixed-bed retort with different heating rates. Oil Shale. 2009;26(2):139–47.
Al-Harahsheh A, Al-Ayed O, Al-Harahsheh M, Abu-El-Halawah R. Heating rate effect on fractional yield and composition of oil retorted from El-lajjun oil shale. J Anal Appl Pyrol. 2010;89(2):239–43.
Degirmenci L, Durusoy T. Effect of heating rate and particle size on the pyrolysis of Gönyük oil shale. Energy Sources. 2005;27(9):787–95.
Khalil AM. Oli shale pyrolysis and effect of particle size on the composition of shale oil. Oil Shale. 2013;30(2):136–46.
Jamal MN. The influence of grain size on the products yield and shale oil composition from the pyrolysis of Sultani oil shale. Energy Convers Manag. 2008;49:3278–86.
Nasir A, Paul TW. Influence of particle grain size on the yield and composition of products from the pyrolysis of oil shales. J Anal Appl Pyrol. 1998;46(1):31–49.
Williams P, Nazzal J. Pyrolysis of oil shales: influence of particle grain size on polycyclic aromatic compounds in the derived shale oil. J Inst Energy. 1999;72(491):48–55.
Carter SD, Citiroglu M, Gallacher J, Snape CE, Mitchell S, Lafferty CJ. Secondary coking and cracking of shale oil vapors from pyrolysis or hydropyrolysis of a Kentucky Cleveland oil shale in a two-stage reactor. Fuel. 1994;73(9):1455–8.
Fainberg V, Garbar A, Hetsroni G. Secondary pyrolysis of the products of the thermal destruction of high-sulfur oil shale. Energy Fuel. 1997;11(4):915–9.
Lai DG, Chen ZH, Lin LX, Zhang YM, Gao SQ, Xu GW. Secondary cracking and upgrading of shale oil from pyrolyzing oil shale over shale ash. Energy Fuel. 2015;29(4):2219–26.
Han XX, Jiang XM, Cui ZG. Studies of the effect of retorting factors on the yield of shale oil for a new comprehensive utilization technology of oil shale. Appl Energy. 2009;86(11):2381–5.
Dung VN. Yields and chemical characteristics of products from fluidized bed steam retorting of Condor and Stuart oil shales: effect of pyrolysis temperature. Fuel. 1990;69(3):368–76.
Burnham AK, Ward RL. A possible mechanism of alkene/alkane produc-tion in oil shale retorting. ACS Div Fuel Chem. 1980;25:82–8.
Raley JH. Monitoring oil shale retorts by off-gas alkenealkane ratios. Fuel. 1980;59(6):419–24.
Williams PT, Nazzal JM. Polycyclic aromatic compounds in oils derived from the fluidised bed pyrolysis of oil shale. J Anal Appl Pyrol. 1995;35(2):181–97.
Williams PT, Nazzal JM. Polycyclic aromatic compounds in shale oils: influence of process conditions. Environ Technol. 1998;19(8):775–87.
Burnham AK. Chemistry of shale oil cracking. American Chemical Society Symposium Series. 1981;163(Chap. 4):39–60.
Li SY, Qian JL, Qin KZ, Zhu YJ. Study of the kinetics of oil shale pyrolysis with bitumen as an intermediate product. Fuel Chem Technol. 1987;15(2):118–23.
Williams P. Thermogravimetry and decomposition kinetics of British Kimmeridge Clay oil shale. Fuel. 1985;64(4):540–5.
Sonoyama N, Nobuta K, Kimura T, Hosokai S, Hayashi J, Tago T, Masuda T. Production of chemicals by cracking pyrolytic tar from Loy Yang coal over iron oxide catalysts in a steam atmosphere. Fuel Process Technol. 2011;92(4SI):771–5.
Acknowledgements
This work was supported by the National Basic Research Program of China (973 programs, No. 2014CB744302), Taishan Scholar Constructive Engineering Foundation of Shandong province, China (No. ts20120518), and talents start scientific research funds of China University of Petroleum, Beijing (No. 2462015YJRC002). We are grateful to Prof. Q. Shi from China University of Petroleum, Beijing, for GC–MS analysis support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, W., Ma, Y., Li, S. et al. Pyrolysis characteristics of Longkou oil shale under optimized condition. J Therm Anal Calorim 125, 983–990 (2016). https://doi.org/10.1007/s10973-016-5452-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5452-4