Abstract
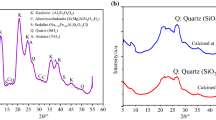
In this study, we have synthesized and performed the thermal decomposition kinetics of anhydrous calcium phosphate monetite CaHPO4 under air atmosphere by TG/DTG and DTA techniques using non-isothermal experiments. The prepared and calcined products at 500 °C were characterized by powder X-ray diffraction, infrared spectroscopy, transmission electron microscopy and X-ray microanalysis. The degradation of CaHPO4 was observed between [425–490 °C] and leads to the formation of calcium pyrophosphate by condensation of orthophosphate groups. The kinetic parameter results achieved by the isoconversional methods combined to Malek’s procedure showed that the thermal dehydroxylation is a single-step process which related to crystal nucleation and growth mechanism, according to JMA (n) model, with n = 1.37 and an activation energy of Friedman E α = 299.4 ± 2.1 kJ mol−1. The thermodynamic functions (ΔS*, ΔH* and ΔG*) of the thermal dehydroxylation step were calculated using the activated complex theory. The obtained values showed that the reaction is directly related to the introduction of heat and is non-spontaneous process.









Similar content being viewed by others
References
Dorozhkin SV. Calcium orthophosphates. J Mater Sci. 2007;42:1061–95.
Dorozhkin SV. Calcium orthophosphates in nature, biology and medicine. Materials. 2009;2:399–498.
Dorozhkin SV. Calcium orthophosphates: occurence, properties, biomineralization, pathological calcification and biomimetic applications. Biomaterials. 2011;1:121–64.
El Hamidi A, Mulongo Masamba R, Khachani M, Halim M, Arsalane S. Kinetics modeling in liquid phase sorption of copper ions on brushite di-calcium phosphate di-hydrate CaHPO4·2H2O (DCPD). Desalin Water Treat. 2015;56:779–91.
Da Silva Filho EC, da Silva OG, da Fonseca MG, Arakaki LNH, Airoldi C. Synthesis and thermal characterization of copper and calcium mixed phosphates. J Therm Anal Calorim. 2007;87:775–8.
Da Silva OG, da Fonseca MG, Arakaki LNH. Silylated calcium phosphates and their new behavior for copper retention from aqueous solution. Colloid Surf A. 2007;301:376–81.
Tortet L, Gavarri JR, Nihoul G, Dianoux AJ. Study of protonic mobility in CaHPO4·2H2O (Brushite) and CaHPO4 (Monetite) by infrared spectroscopy and neutron scattering. J Solid State Chem. 1997;132:6–16.
Doat A, Pellé F, Lebugle A. Europium-doped calcium pyrophosphates: allotropic forms and photoluminescent properties. J Solid State Chem. 2005;178:2354–62.
Miyazaki T, Sivaprakasam K, Tantry J, Suryanarayanan R. Physical characterization of dibasic calcium phosphate dihydrate and anhydrate. J Pharm Sci. 2009;98:905–16.
Chen GG, Luo GS, Yang LM, Xu JH, Sun Y, Wang JD. Synthesis and size control of CaHPO4 particles in a two-liquid phase micro-mixing process. J Cryst Growth. 2005;279:501–7.
Eshtiagh H, Houssaindokht MR, Chahkandhi M, Youssefi A. Preparation of anhydrous dicalcium phosphate, DCPA, through sol–gel process, identification and phase transformation evaluation. J Non-Cryst Solids. 2008;354:3854–7.
Baradaran S, Basirun WJ, Mahmoudian M, Hamdi M, Alias Y. Synthesis and characterization of monetite prepared using a sonochemical method in a mixed solvent system of water/ethylene glycol/N,N-dimethylformamide. Metall Mater Trans A. 2013;44:2231–8.
Dickens B, Bowen JS, Brown WE. A refinement of the crystal structure of CaHPO4 (synthetic monetite). Acta Crystallogr B Struct Sci. 1972;28:797–806.
Wikholm NW, Beebe RA, Kittelberger JS. Kinetics of the conversion of monetite to calcium pyrophosphate. J Phys Chem. 1975;79:853–6.
Berend G, Hegedüs AJ. Thermoanalytical investigation of secondary calcium phosphate dehydrate. Thermochim Acta. 1975;11:367–79.
Hlel F, Kamoun S, Guidara K. Investigation of phosphorus site condensation in CaHPO4 by analysis of 31P MAS–NMR tensor and X-ray powder patterns. Z Naturforsch A. 2006;61:375–82.
Vyazovkin S, Chrissafis K, Di Lorenzo ML, Koga N, Pijolat M, Roduit B, Sbirrazzuoli N, Suñol JJ. ICTAC kinetics committee recommendations for collecting experimental thermal analysis data for kinetic computations. Thermochim Acta. 2014;590:1–23.
Vyazovkin S, Burnham AK, Criado JM, Pérez-Maqueda LA, Popescu C, Sbirrazzuoli N. ICTAC kinetics committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta. 2011;520:1–19.
Sbirrazzuoli N, Vincent L, Bouillard J, Elégant L. Isothermal and non-isothermal kinetics when mechanistic information available. J Therm Anal Calorim. 1999;56:783–92.
Brown ME, Gallagher PK. Handbook of thermal analysis and calorimetry: recent advances, techniques and applications. 1st ed. Amsterdam: Elsevier; 2008.
Friedman HL. Kinetics of thermal degradation of char-forming plastics from thermogravimetry. Application to a phenolic plastic. J Polym Sci Polym Symp. 1964;6:183–95.
Doyle CD. Estimating isothermal life from thermogravimetric data. J Appl Polym Sci. 1962;6:639–42.
Ozawa T. A new method of analyzing thermogravimetric data. Bull Chem Soc Jpn. 1965;38:1881–6.
Coats AW, Redfern JP. Kinetic parameters from thermogravimetric data. Nature. 1964;201:68–9.
Málek J. The kinetic analysis of non-isothermal data. Thermochim Acta. 1992;200:257–69.
Senum GI, Yang RT. Rational approximations of the integral of the Arrhenius function. J Therm Anal. 1977;11:445–7.
Flynn JH. The “temperature integral”—its use and abuse. Thermochim Acta. 1997;300:83–92.
Shannon RD. Activated complex theory applied to the thermal decomposition of solids. Trans Faraday Soc. 1964;60:1902–13.
Eyring H. The activated complex in chemical reactions. J Chem Phys. 1935;3:107–15.
Ma H, Yan B, Li J, Ren Y, Chen Y, Zhao F, Song J, Hu R. Molecular structure, thermal behavior and adiabatic time-to-explosion of 3,3-dinitroazetidinium picrate. J Mol Struct. 2010;981:103–10.
Petrov I, Šoptrajanov B, Fuson N, Lawson JR. Infra-red investigation of dicalcium phosphates. Spectrochim Acta A Mol Spectrosc. 1967;23:2637–46.
Šimon P, Thomas P, Dubaj T, Cibulková Z, Peller A, Veverka M. The mathematical incorrectness of the integral isoconversional methods in case of variable activation energy and the consequences. J Therm Anal Calorim. 2013;115:853–9.
Sbirrazzuoli N, Vincent L, Mija A, Guigo N. Integral, differential and advanced isoconversional methods: complex mechanisms and isothermal predicted conversion–time curves. Chemometr Intell Lab. 2009;96:219–26.
Koga N, Tanaka H. A physico-geometric approach to the kinetics of solid-state reactions as exemplified by the thermal dehydration and decomposition of inorganic solids. Thermochim Acta. 2002;388:41–61.
Koga N, Takemoto S, Tatsuya N, Haruhiko T. A kinetic study of the thermal decomposition of iron(III) oxide-hydroxides. Part 3. Shape control and thermal decomposition of α-FeO(OH). Thermochim Acta. 1996;282(283):81–90.
Acknowledgements
This research was financially supported by University of Mohammed V - Morocco under the Project No. SCH 04/09 and CNRST-Morocco.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mulongo-Masamba, R., El Kassri, T., Khachani, M. et al. Synthesis and thermal dehydroxylation kinetic of anhydrous calcium phosphate monetite CaHPO4 . J Therm Anal Calorim 124, 171–180 (2016). https://doi.org/10.1007/s10973-015-5130-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-015-5130-y