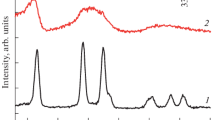
Abstract
Inclusion compounds (intercalates) of fluorinated graphite matrix with butanone (C2FxBrz·yCH3COC2H5, x = 0.49, 0.69, 0.87, 0.92, z ≈ 0.01) were prepared by guest substitution from acetonitrile to butanone. The kinetics of the thermal decomposition (the 1st stage of filling → the 2nd stage of filling) was studied under isothermal conditions at 294–313 K. The relationship of the host matrices structure with inclusion compounds’ thermal properties and kinetic parameters is discussed.







Similar content being viewed by others
References
Pinakov DV, Logvinenko VA. The relationship between properties of fluorinated graphite intercalates and matrix composition. 1. Intercalates with acetonitrile. J Therm Anal Calorim. 2006;86:173–8.
Pinakov DV, Logvinenko VA, Shubin YuV, Chekhova GN. The relationship between properties of fluorinated graphite intercalates and matrix composition. 2. Intercalates with chloroform. J Therm Anal Calorim. 2007;90:399–405.
Pinakov DV, Logvinenko VA, Shubin YuV, Chekhova GN. Relationship between properties of fluorinated graphite intercalates and matrix composition. Part III. Intercalates with 1,2-dichloroethane. J Therm Anal Calorim. 2009;96:501–5.
Pinakov DV, Logvinenko VA, Shubin YuV, Chekhova GN. The relationship between properties of fluorinated graphite intercalates and matrix composition. Part 4. Intercalates with methylene dichloride. J Therm Anal Calorim. 2010;100:163–9.
Pinakov DV, Logvinenko VA, Chekhova GN, Shubin YuV. The relationship between properties of fluorinated graphite intercalates and matrix composition. Part 5. Intercalates with acetone. J Therm Anal Calorim. 2011;104:1077–82.
Osipov OA, Minkin VI, Kletenik YuB. Dipole moments reference book (in Russian). Rostov: Vysshaya Shkola; 1965. p. 264.
Yudanov NF, Yakovlev II. Determination of fluorinated graphite chemical formula in its inclusion compounds (in Russian). Izvestiya Siberian Branch USSR Acad Sci. 1988;21(pt. 1):66–9.
Nikolsky BP (ed.). Chemical reference book, vol 1. Moscow-Leningrad: Chemistry; 1962. p. 694.
Chekhova GN, Ukraintseva EA, Ivanov IM, Yudanov NF, Shubin YuV, Logvinenko VA, Pinakov DV, Fadeeva VP, Alferova NI. Influence of the matrix composition on the properties of fluorinated graphite inclusion compounds with acetonitrile. J Inorg Chem. 2005;50(7):1055–61.
Shubin YuV, Pinakov DV, Chekhova GN, Alferova NI, Logvinenko VA. Phase transitions of intercalation inclusion compounds C2F0.92Br0.01·yCH3CN in the temperature range 20–260 °C. J Struct Chem. 2006;47:1141–54.
Pinakov DV. Different fluorination degree fluorinated graphite matrices intercalates synthesis and thermal stability. Ph.D. Thesis. Novosibirsk; 2007. p. 163.
Gordon AJ, Ford RA. The chemist’s companion. New York: Wiley; 1972. p. 541.
Erofeev BV. Generalized equation of chemical kinetics and its application to solid phase reactions. Rep USSR Acad Sci. 1946;52(6):515–8.
Sakovich GV. Notes about certain kinetic equations currently applied to solid-state reactions. Tomsk State Univ Sci Notes. 1955;26:103–10.
Patrilyak KI. Inclusion compounds and certain problems of heterogeneous equilibria (in Russian). Kiev: Naukova Dumka; 1987. p. 164.
Vlaev LT, Gospodinov GG, Genieva SD. The kinetics of isothermal decomposition of antimony and bismuth selenites. J Phys Chem. 2002;76(9):1437–40.
Stull D, Westrum E, Sinke G. Chemical thermodynamics of organic compounds. New York: Wiley; 1969.
Logvinenko VA. Thermal analysis of coordination compounds and clathrates (in Russian). Novosibirsk: Nauka, Siberian Department; 1982. p. 128.
Acknowledgments
The investigation was supported by the Russian Foundation for Basic Research under Project 09-03-00902. Special thanks to N. I. Alferova and V. P. Fadeeva for collaboration.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pinakov, D.V., Logvinenko, V.A. & Chekhova, G.N. The relationship between properties of fluorinated graphite intercalates and matrix composition. J Therm Anal Calorim 115, 503–509 (2014). https://doi.org/10.1007/s10973-013-3188-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-013-3188-y