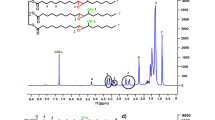
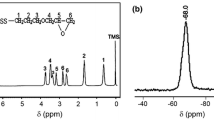
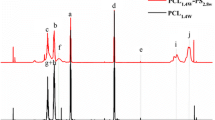
Abstract
The bulk crosslinking reaction kinetics of a novel two-component waterborne polyurethanes (2K-WPUs) composed of a bio-resin-based polyol dispersion and a hydrophilically modified hexamethylene diisocyanate tripolymer are investigated by freeze–drying and differential scanning calorimetry (DSC) technique at different heating rates. The data fit for the above two components is implemented with the nth-order kinetics equation and Málek’s mechanism function method, respectively. The kinetic parameters of crosslinking reaction are determined by the kinetic analysis of the data obtained from the thermal treatment, and then the kinetic model is built. The results indicate that the nth-order model deduced from Kissinger and Crane equation has great distinction with the experimental data, while the Málek analytic mechanism shows that the crosslinking process of the crosslinking reaction follows an autocatalytic reaction. The two-parameter (m and n) autocatalytic model (S–B model) can well describe the crosslinking reaction process of the studied 2K-WPU. The DSC curves derived from the experimental data show a good agreement with the theoretical calculation under 5–20 °C min−1 heating rate. The results provide theoretical basis for the choice of the manufacturing process and the optimization of processing window.








Similar content being viewed by others
References
Melchiors M, Sonntag M, Kobusch C, Jurgents E. Recent developments in aqueous two-component polyurethane (2K-PUR) coatings. Prog Org Coat. 2000;40:99–109.
Otts DB, Urban MW. Heterogeneous crosslinking of waterborne two-component polyurethanes; stratification processes and the role of water. Polymer. 2005;46:2699–709.
Otts DB, Pereira KJ, Arret WL, Urban MW. Dynamic colloidal processes in waterborne two-component polyurethanes and their effect on solution and film morphology. Polymer. 2005;46:4776–88.
Zhou X-H, Tu W-P, Hu J-Q. Preparation and characterization of two-component waterborne polyurethane comprised of water-soluble acrylic resin and HDI biuret. Chinese J Chem Eng. 2006;14:99–104.
Zhu W-Z, Liu X-Y. Study on curing kinetics of cast polyurethane elastomers by durometer. Polyurethane Ind. 1996;3:27–31.
Baker JW, Gaunt J. The mechanism of the reaction of aryl isocyanates with alcohols and amines. J Chem Soc. 1949;9–31.
Ren N–N, Yu X-H, Yu Y. Influences of catalysts on the film formation and performance of 2-K waterborne polyurethane. Paint Coat Ind. 2010;40:27–31.
Karger-Kocsis J. Thermoset polymers containing bio-based renewable resources. Exp Polym Lett. 2009;3:676.
Song Z-Q. Researches on pine chemicals in China. Chem Ind For Prod. 2004;24:7–11.
Wu G-M, Kong Z-W, Chu F-X. Synthesis of epoxy resin from hydrogenated terpinene- maleic anhydride. Chem Ind For Prod. 2007;27:57–62.
Wu G-M, Kong Z-W, Chu F-X. Study on curing reaction and mechanical properties of epoxy resin from hydrogenated terpinene-maleic anhydride. Chem Ind For Prod. 2007;27:21–6.
Wu G-M, Kong Z-W, Chen J, Jiang J-C. Preparation and properties of nonionic polyol dispersion from terpinene-maleic ester type epoxy resin. J App Polym Sci. 2011;120:579–85.
Wu G-M, Kong Z-W, Huang H, Chen J, Chu F-X. Synthesis, characterization and properties of polyols from hydrogenated terpinene-maleic ester type epoxy resin. J App Polym Sci. 2009;113:2894–901.
Mahendran AR, Wuzella G, Kandelbauer A, Aust N. Thermal cure kinetics of epoxidized linseed oil with anhydridehardener. J Therm Anal Calorim. 2011. doi:10.1007/s10973-011-1585-7.
Sheng X, Akinc M, Kessler MR. Cure kinetics of thermosetting bisphenol E cyanate ester. J Therm Anal Calorim. 2008;93:77–85.
Garcia SJ, Serra A, Ramis X. Influence of the addition of erbium and ytterbium triflates in the curing kinetics of a DGEBA/o-tolybiguanide powder mixture. J Therm Anal Calorim. 2007;89:223–31.
Sabzevari SM, Alavi-Soltani S, Minaie B. Effect of thermoplastic toughening agent on glass transition temperature and cure kinetics of an epoxy prepreg. J Therm Anal Calorim. 2011;106:905–11.
Yousefi A, Lafleur PG, Gauvin R. Kinetic studies of thermoset cure reactions: A review. Poly Compos. 1997;18:157–68.
Koreeda T, Matos J, Goncalves CS. Cure kinetics of epoxy composite applied on stator bars insulation. J Therm Anal Calorim. 2011;106:631–5.
Wan J-T, Fan H, Li B-G. Synthesis and nonisothermal reaction of a novelacrylonitrile-capped poly(propyleneimine) dendrimerwith epoxy resin. J Therm Anal Calorim. 2011;103:685–92.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29:1702–6.
Fu X-C, Shen W-X, Yao T-Y, Hou W-H. Physical chemistry Beijing. China: China Higher Education Press; 2006. p. 238.
Málek J. The kinetic analysis of non-isothermal data. Thermochim Acta. 1992;200:257–69.
Montserrat S, Málek J. A kinetic analysis of the curing reaction of an epoxy resin. Thermochim Acta. 1993;228:47–60.
Senum GI, Yang RT. Rational approximations of the integral of the Arrhenius function. J Therm Anal Calorim. 1977;11:445–7.
Vyazovkin S, Sbirrazzuoli N. Isoconversional kinetic analysis of thermally stimulated processes in polymers. Macromol Rapid Comm. 2006;27:1515–32.
Zhu L-M. Polyurethane synthetic material. Nanjing: Phoenix Science Press; 2002. p. 15.
Sestak J, Berggren G. Study of the kinetics of the mechanism of solid-state reactions at increasing temperatures. Thermochim Acta. 1971;3:1–12.
Acknowledgements
The authors gratefully acknowledge the financial support from the Chinese National Natural Science Foundation (contract Grant number: 30972320, 31100428).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wu, G.M., Kong, Z.W., Chen, C.F. et al. Kinetics of the crosslinking reaction of nonionic polyol dispersion from terpene-maleic ester-type epoxy resin. J Therm Anal Calorim 111, 735–741 (2013). https://doi.org/10.1007/s10973-012-2259-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-012-2259-9