Abstract
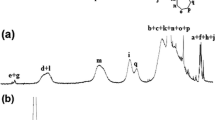
Samples of poly(l,l-lactide)-block-poly(ethylene glycol)-block-poly(l,l-lactide) (PLLA-PEG-PLLA) were synthesized from l,l-lactide polymerization using stannous 2-ethylhexanoate, Sn(Oct)2 as initiator and di-hydroxy-terminated poly(ethylene glycol) (PEG) (M n = 4000 g mol−1) as co-initiator. The chemical linkage between the PEG segment and the PLA segments was characterized by Fourier transform infrared spectroscopy (FTIR). Thermogravimetry analysis (TG) revealed the copolymers composition and was capable to show the deleterious effect of an excess of Sn(Oct)2 in the polymer thermal stability, while Differential Scanning Calorimetry (DSC) allowed the observation of the miscibility between the PLLA and PEG segments in the different copolymers.






Similar content being viewed by others
References
Langer R. Biomaterials in drug delivery and tissue engineering: one laboratory’s experience. Accounts Chem Res. 2000;33:94–101.
Hoffman AS. Hydrogels for biomedical applications. Adv Drug Deliv Rev. 2002;43:3–12.
Lee KS, Mooney DJ. Hydrogels for tissue engineering. Chem Rev. 2001;101:1869–80.
Riess G. Micellization of block copolymers. Prog Polym Sci. 2003;28:1107–70.
Deng C, Rong G, Tian H, Tang Z, Chen X, Jing X. Synthesis and characterization of poly(ethylene glycol)-b-poly(l-lactide)-b-poly(l-glutamic acid) triblock copolymer. Polymer. 2005;46:653–9.
Deng C, Chen X, Yu H, Sun J, Lu T, Jing X. A biodegradable triblock copolymer poly(ethylene glycol)-b-poly(l-lactide)-b-poly(l-lysine): synthesis, self-assembly and RGD peptide modification. Polymer. 2007;48:139–49.
Griffith LG. Polymeric biomaterials. Acta Mater. 2000;48:263–77.
Jacobsen S, Fritz HG, Degée Ph, Dubois Ph, Jérôme R. New developments on the ring opening polymerization of polylactide. Ind Crops Prod. 2000;11:265–75.
Zhang C, Liao L, Gong S. Synthesis of PLLA-MPEG diblock copolymers by microwave-assisted copolymerization of l-lactide and methoxy poly(ethylene glycol). Macromol Chem Phy. 2007;208:1122–8.
Kowalski A, Duda A, Penczek S. Kinetics and mechanism of cyclic esters polymerization initiated with Tin(II) octoate: polymerization of l,l-dilactide. Macromolecules. 2000;33:7359–70.
Ryner M, Stridsberg K, Albertsson AC, Schenck HV, Svensson M. Mechanism of ring-opening polymerization of 1, 5-dioxepan-2-one and l-lactide with stannous 2-ethylhexanoate: a theoretical study. Macromolecules. 2001;34:3877–81.
Storey RF, Sherman JW. Kinetics and mechanism of the stannous octoate-catalyzed bulk polymerization of ε-caprolactone. Macromolecules. 2002;35:1504–12.
Drumond WS, Mothé CG, Wang SH. Síntese e caracterização do copolímero poli(ácido lático-b-glicol etilênico). Polímeros: Ciência Tecnologia. 2004;14:74–9.
Drumond WS, Mothé CG, Wang SH. Quantitative analysis of biodegradable amphiphilic poly(l,l-lactide)-block-poly(ethylene glycol)-block-poly(l,l-lactide) by using TG, FTIR and NMR. J Therm Anal Cal. 2006;85:173–7.
Mothé CG, Drumond WS, Wang SH. Phase behaviour of biodegradable amphiphilic poly(l,l-lactide)-b-poly(ethylene glycol)-b-poly(l,l-lactide). Thermochim Acta. 2006;445:61–6.
Penco M, Bignotti F, Sartore S, D’Antone S, D’Amore A. Multiblock copolymers based on segments of poly (d,l-lactic-glycolic acid) and poly(ethylene glycol) or poly(#-caprolactone): a comparison of their thermal properties and degradation behaviour. J Appl Polym Sci. 2000;78:1721–8.
Martino VP, Ruseckaite RA, Jiménez A. Thermal and mechanical characterization of plasticized poly(l-lactide-co-d,l-lactide) films for food packaging. J Therm Anal Cal. 2006;86:707–12.
Acknowledgements
The authors thank Brazilian agencies CAPES, CNPq, and FAPESP for their financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mothé, C.G., Azevedo, A.D., Drumond, W.S. et al. Thermal properties of amphiphilic biodegradable triblock copolymer of l,l-lactide and ethylene glycol. J Therm Anal Calorim 101, 229–233 (2010). https://doi.org/10.1007/s10973-009-0589-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-009-0589-z