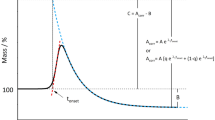
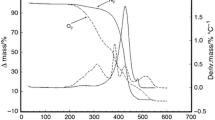
Abstract
Thermal decomposition of extra-virgin olive oil (EVOO) was investigated by thermogravimetry (TG) and derivative thermogravimetry (DTG) up to 550°C at different heating rates (from 5 to 12.5°C min−1). The thermal degradation study of four unsaturated or saturated esterified C18 fatty acids with glycerol (i.e., glyceryl-tristearate (C18:0),-trioleate (C18:1),-trilinoleate (C18:2) and-trilinolenate (C18:3)) was also carried out under the same experimental conditions.
A deconvolution procedure applied only to the first two overlapping steps of EVOO and C18:1 enabled the activation energy of decomposition to be determined both by the Kissinger and the Ozawa-Flynn-Wall isoconversional method for the two deconvoluted steps of EVOO and C18:1, as well as for the only single step of the other three C18 triglycerides. Practically constant activation energy for the first deconvoluted step of EVOO and C18:1 and for the single step of C18:0 was found in good agreement with the results obtained with the Kissinger method, while a similar increasing trend was observed for the second decomposition step of EVOO and C18:1 and for the single steps of C18:2 and C18:3 triglycerides.
Similar content being viewed by others
References
Kirk and Otmer, Encyclopedia of Chemical Technology: Fats and Fatty Acids, 3rd Ed., Wiley and Sons, Vol. 9, 1980, p. 808.
I. Buzás, J. Simon and J. Holló, J. Am. Oil Chem. Soc., 56 (1979) 685.
Y. H. Roos, J. Therm. Anal. Cal., 71 (2003) 197.
J. Magoshi, M. A. Becker, Z. Han and S. Nakamura, J. Therm. Anal. Cal., 70 (2002) 833.
B. Kowalski, Thermochim. Acta, 184 (1991) 49.
C. P. Tan and Y. B. Che Man, J. Am. Oil Chem. Soc., 77 (2000) 143.
L. Campanella, G. Favero, M. Pastorino and M. Tomassetti, Biosens. Bioelectron., 14 (1999) 179.
M. Angiuli, C. Ferrari, L. Lepori, E. Matteoli, G. Salvetti, E. Tombari, A. Banti and N. Minnaja, J. Therm. Anal. Cal., 84 (2006) 105.
M. Ollivon, G. Keller, C. Bourgaux, D. Kalnin, P. Villeneuve and P. Lesieur, J. Therm. Anal. Cal., 85 (2006) 219.
M. B. Dantas, M. M. Conceição, V. J. Fernandez Jr, N. A. Santos, R. Rosenhaim. A. L. B. Marques, I. M. G. Santos and A. G. Sousa, J. Therm. Anal. Cal., 87 (2007) 835.
J. Dweck and C. M. S. Sampaio, J. Therm. Anal. Cal., 75 (2004) 385.
J. C. O. Santos, I. M. G. Santos, M. M. Conceiçào, S. L. Porto, M. F. S. Trindade, A. G. Souza, S. Prasad, V. J. Fernandez Jr. and A. S. Araújo, J. Therm. Anal. Cal., 75 (2004) 419.
X. Gao, D. Chen and D. Dollimore, Thermochim. Acta, 223 (1993) 75.
H. E. Kissinger, Anal. Chem., 29 (1957) 1702.
J. H. Flynn and L. A. Wall, J. Polym. Sci. B: Polym. Lett., 4 (1966) 323.
T. Ozawa, Bull. Chem. Soc. Jpn., 38 (1965) 1881.
C. D. Doyle, J. Appl. Polym. Sci., 6 (1962) 639.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vecchio, S., Campanella, L., Nuccilli, A. et al. Kinetic study of thermal breakdown of triglycerides contained in extra-virgin olive oil. J Therm Anal Calorim 91, 51–56 (2008). https://doi.org/10.1007/s10973-007-8373-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-007-8373-4