Abstract
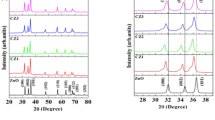
Carbon-doped zinc oxides were synthesized with different concentrations corresponding to PVA/Zn2+ molar ratios between 5 and 15% by the hydrothermal method. The XRD and SEM results show that the obtained materials have high crystallinity and a pill-like morphology. Carbon dopant significantly decreased band gap energy (EgCZ3 = 2.77 eV), improving the optical absorption of ZnO in the visible and ultraviolet regions. Generally, the catalyst with carbon doped 1.49% (CZ3), corresponding to the molar ratio of PVA/Zn2+ = 15%, showed the highest photocatalytic efficiency of ofloxacin (Ofx) and methylene blue (MB) and was higher than pure ZnO. This is possibly attributed to its morphology with good uniformity and porosity; the specific surface area of SCZ3 is 26 m2/g which is 1.5 times higher than ZnO. Moreover, it is also relative to the stable and highest saturation photocurrent density of CZ3, which is ∼6.4 µA.cm−2, 4.15 times higher than pure ZnO. In addition, trapping experiments showed that positive holes and hydroxyl radicals were the predominant active agents. Finally, COD and TOC analysis results suggested that the photodegradation progress of MB and Ofx formed organic compounds with smaller structures and proved that over 78% of MB and 66% of Ofx were converted entirely to simple inorganic substances such as CO2, H2O, etc.
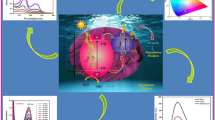
Graphical Abstract

Highlights
-
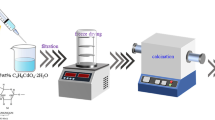
The pill-like nano-photocatalysts of C-doped ZnO (CZ) were prepared by the hydrothermal method with various PVA/Zn2+ molar ratios (5, 10, and 15%).
-
The C doping significantly decreased the band gap of pure ZnO (from 3.1 to 2.77 eV).
-
Enhancing the photoinduced charge separation, observing that photocurrent is about 5.5 times greater than pure ZnO.
-
Excellent performance in the decomposition of ofloxacin (Ofx) and methylene blue (MB) under visible light illumination and sunlight.
-
The role of photoactive species in the reaction process was ordered as follows: h+ > \(H{O}^{.} > {e}^{-}\).












Similar content being viewed by others
References
Gaur N, Dutta D, Singh A, et al. (2022) Recent advances in the elimination of persistent organic pollutants by photocatalysis. Front Environ Sci 10: https://doi.org/10.3389/fenvs.2022.872514
Alahmadi N (2022) Recent progress in photocatalytic removal of environmental pollution hazards in water using nanostructured materials. Separations 9: https://doi.org/10.3390/separations9100264
Nan M, Jin B, Chow CWK, Saint C (2010) Recent developments in photocatalytic water treatment technology: A review. Water Res. 44:2997–3027. https://doi.org/10.1016/j.watres.2010.02.039
Kolodziejczak-Radzimska A, Jesionowski T (2014) Zinc oxide-from synthesis to application: A review. Materials. 7:2833–2881. https://doi.org/10.3390/ma7042833
Lee JM, Pyun YB, Yi J et al. (2009) ZnO nanorod-graphene hybrid architectures for multifunctional conductors. J Phys Chem C. 113:19134–19138. https://doi.org/10.1021/jp9078713
Chen HM, Chen CK, Chang YC et al. (2010) Quantum dot monolayer sensitized ZnO nanowire-array photoelectrodes: True efficiency for water splitting. Angew Chem - Int Ed 49:5966–5969. https://doi.org/10.1002/anie.201001827
Lee KM, Lai CW, Ngai KS, Juan JC (2016) Recent developments of zinc oxide based photocatalyst in water treatment technology: A review. Water Res. 88:428–448. https://doi.org/10.1016/j.watres.2015.09.045
Mohamed KM, Benitto JJ, Vijaya JJ, Bououdina M (2023) Recent advances in ZnO-based nanostructures for the photocatalytic degradation of hazardous, non-biodegradable medicines. Crystals 13: https://doi.org/10.3390/cryst13020329
Kumari H, Sonia, Suman, et al. (2023) A review on photocatalysis used for wastewater treatment: dye degradation. Springer International Publishing.
Tanji K, Navio JA, Martín-Gómez AN et al. (2020) Role of Fe(III) in aqueous solution or deposited on ZnO surface in the photoassisted degradation of rhodamine B and caffeine. Chemosphere 241:125009. https://doi.org/10.1016/j.chemosphere.2019.125009
Tanji K, Navio JA, Naja J, et al (2019) Extraordinary visible photocatalytic activity of a Co0.2Zn0.8O system studied in the Remazol BB oxidation. J Photochem Photobiol A Chem 382: https://doi.org/10.1016/j.jphotochem.2019.111877
Belghiti M, Tanji K, El Mersly L et al. (2022) Fast and non-selective photodegradation of basic yellow 28, malachite green, tetracycline, and sulfamethazine using a nanosized ZnO synthesized from zinc ore. React Kinet, Mech Catal 135:2265–2278. https://doi.org/10.1007/s11144-022-02232-8
Yang Y, Li Y, Zhu L et al. (2013) Shape control of colloidal Mn doped ZnO nanocrystals and their visible light photocatalytic properties. Nanoscale. 5:10461–10471. https://doi.org/10.1039/c3nr03160h
Asahi R, Morikawa T, Ohwaki T et al. (2001) Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 293:269–271. https://doi.org/10.1126/science.1061051
Yu W, Zhang J, Peng T (2016) New insight into the enhanced photocatalytic activity of N-, C- and S-doped ZnO photocatalysts. Appl Catal B 181:220–227. https://doi.org/10.1016/j.apcatb.2015.07.031
Liu S, Li C, Yu J, Xiang Q (2011) Improved visible-light photocatalytic activity of porous carbon self-doped ZnO nanosheet-assembled flowers. CrystEngComm 13:2533–2541. https://doi.org/10.1039/c0ce00295j
Haibo O, Feng HJ, Cuiyan L et al. (2013) Synthesis of carbon doped ZnO with a porous structure and its solar-light photocatalytic properties. Mater Lett 111:217–220. https://doi.org/10.1016/j.matlet.2013.08.081
Alshammari AS, Chi L, Chen X et al. (2015) Visible-light photocatalysis on C-doped ZnO derived from polymer-assisted pyrolysis. RSC Adv 5:27690–27698. https://doi.org/10.1039/c4ra17227b
Ansari SA, Ansari SG, Foaud H, Cho MH (2017) Facile and sustainable synthesis of carbon-doped ZnO nanostructures towards the superior visible light photocatalytic performance. N. J Chem 41:9314–9320. https://doi.org/10.1039/c6nj04070e
Srinivasan N, Anbuchezhiyan M, Harish S, Ponnusamy S (2019) Applied Surface Science Hydrothermal synthesis of C doped ZnO nanoparticles coupled with BiVO 4 and their photocatalytic performance under the visible light irradiation. Appl Surf Sci 494:771–782. https://doi.org/10.1016/j.apsusc.2019.07.093
Perillo PM, Atia MN (2017) Nano-Structures & Nano-Objects C-doped ZnO nanorods for photocatalytic degradation of p-aminobenzoic acid under sunlight. Nano-Struct Nano-Objects 10:125–130. https://doi.org/10.1016/j.nanoso.2017.04.001
Bechambi O, Sayadi S, Najjar W (2015) Photocatalytic degradation of bisphenol A in the presence of C-doped ZnO: Effect of operational parameters and photodegradation mechanism. J Ind Eng Chem 32:201–210. https://doi.org/10.1016/j.jiec.2015.08.017
Matthews JA (2014) Chemical Oxygen Demand (Cod). Encyclopedia of Environmental Change 14–19 https://doi.org/10.4135/9781446247501.n623
Nezamzadeh-Ejhieh A, Ghanbari-Mobarakeh Z (2015) Heterogeneous photodegradation of 2,4-dichlorophenol using FeO doped onto nano-particles of zeolite P. J Ind Eng Chem 21:668–676. https://doi.org/10.1016/j.jiec.2014.03.035
Payrazm S, Baghshahi S, Sadeghian Z, Aliabadizadeh A (2022) Structural and optical characterization of ZnO-graphene nanocomposite quantum dots. Iran J Mater Sci Eng 19:1–13. https://doi.org/10.22068/ijmse.2603
Wazir AH, Kundi IW (2016) Synthesis of graphene nano sheets by the rapid reduction of electrochemically exfoliated graphene oxide induced by microwaves. J Chem Soc Pak 38:11–16
Güell F, Martínez-Alanis PR, Khachadorian S et al. (2016) Raman and photoluminescence properties of ZnO nanowires grown by a catalyst-free vapor-transport process using ZnO nanoparticle seeds. Phys Status Solidi B Basic Res 253:883–888. https://doi.org/10.1002/pssb.201552651
Silambarasan M, Saravanan S, Soga T (2014) Raman and photoluminescence studies of Ag and Fe-doped ZnO nanoparticles. Int J ChemTech Res 2014–2015 7(3):1644–1650
Beltrán JJ, Barrero CA, Punnoose A (2019) Relationship between ferromagnetism and formation of complex carbon bonds in carbon-doped ZnO powders. Phys Chem Chem Phys 21:8808–8819. https://doi.org/10.1039/c9cp01277j
Sun W, Meng S, Zhang S et al. (2018) Insight into the transfer mechanisms of photogenerated carriers for heterojunction photocatalysts with the analogous positions of valence band and conduction band: a case study of ZnO/TiO 2. J Phys Chem C 122:15409–15420. https://doi.org/10.1021/acs.jpcc.8b03753
Dargahi Z, Asgharzadeh H, Maleki-Ghaleh H (2018) Synthesis of Mo-doped TiO2/reduced graphene oxide nanocomposite for photoelectrocatalytic applications. Ceram Int 44:13015–13023. https://doi.org/10.1016/j.ceramint.2018.04.120
Maleki-Ghaleh H, Shahzadeh M, Hoseinizadeh SA et al. (2016) Evaluation of the photo-electro-catalytic behavior of nano-structured ZnO films fabricated by electrodeposition process. Mater Lett. 169:140–143. https://doi.org/10.1016/j.matlet.2016.01.090
Mu J, Shao C, Guo Z et al. (2011) High photocatalytic activity of ZnO-carbon nanofiber heteroarchitectures. ACS Appl Mater Interfaces 3:590–596. https://doi.org/10.1021/am101171a
Dillip GR, Banerjee AN, Anitha VC et al. (2016) Oxygen vacancy-induced structural, optical, and enhanced supercapacitive performance of zinc oxide anchored graphitic carbon nanofiber hybrid electrodes. ACS Appl Mater Interfaces 8:5025–5039. https://doi.org/10.1021/acsami.5b12322
Wang J, Wang Z, Huang B et al. (2012) Oxygen vacancy induced band-gap narrowing and enhanced visible light photocatalytic activity of ZnO. ACS Appl Mater Interfaces 4:4024–4030. https://doi.org/10.1021/am300835p
Mishra DK, Mohapatra J, Sharma MK et al. (2013) Carbon doped ZnO: Synthesis, characterization and interpretation. J Magn Magn Mater 329:146–152. https://doi.org/10.1016/j.jmmm.2012.09.058
Tayyebi A, Soltani T, Lee B-K et al. (2017) Novel visible light photocatalytic and Photoelectrochemical (PEC) activity of carbon-doped zinc oxide/reduced graphene oxide: supercritical methanol synthesis with enhanced photocorrosion suppression. J Alloy Compd 723:1001–1010. https://doi.org/10.1016/j.jallcom.2017.06.316
Fu F, Li L, Liu L et al. (2015) Construction of cellulose based ZnO nanocomposite films with antibacterial properties through one-step coagulation. ACS Appl Mater Interfaces 7:2597–2606. https://doi.org/10.1021/am507639b
Okeri HA, Arhewoh IM (2008) Analytical profile of the fluoroquinolone antibacterials. I. Ofloxacin. Afr J Biotechnol 7:670–680
Jaswal A, Kaur M, Singh S et al. (2021) Adsorptive removal of antibiotic ofloxacin in aqueous phase using rGO-MoS2 heterostructure. J Hazard Mater 417:125982. https://doi.org/10.1016/j.jhazmat.2021.125982
Sawant SY, Cho MH (2016) Facile and single-step route towards ZnO@C core-shell nanoparticles as an oxygen vacancy induced visible light active photocatalyst using the thermal decomposition of Zn(an)2(NO3)2. RSC Adv 6:70644–70652. https://doi.org/10.1039/c6ra14108k
Wang Y, Hu S, Li Q et al. (2018) One step synthesis of high-efficiency AgBr-Br-g-C3N4 composite catalysts for photocatalytic H2O2 production via two channel pathway. RSC Adv 8:36903–36909. https://doi.org/10.1039/c8ra07749e
Hinshelwood CN (1945) Physical chemistry. Nature 156:283–284. https://doi.org/10.1038/156283a0
Dung NT, Thu LM, Thuy UTD et al. (2022) Mechanism insight into the photocatalytic degradation of fluoroquinolone antibiotics by the ZIF-8@Bi2MoO6 heterojunction. Environ Sci Nano 9:3973–3991. https://doi.org/10.1039/d2en00219a
Orange M, Rodwihok C, Wongratanaphisan D, et al. (2020) Cerium-oxide-nanoparticle-decorated zinc oxide with enhanced photocatalytic degradation of methyl orange. https://doi.org/10.3390/app10051697
Author contributions
LTVH: Methodology, Writing - original draft, Formal analysis, Validation, Writing - review & editing, Project administration. NTMT: Investigation, Resources. TTTT: Investigation, Resources. LNT: Investigation, Resources. NTD: Data curation, Software. DHP: Writing - review & editing.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics
This study does not present research with ethical considerations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luu, T.V.H., Tho, N.T.M., Thuy, T.T.T. et al. Synthetization pill-like C-doped ZnO nano-photocatalyst for removing ofloxacin and methylene blue under visible light. J Sol-Gel Sci Technol 110, 204–220 (2024). https://doi.org/10.1007/s10971-024-06348-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-024-06348-2