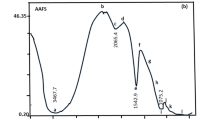
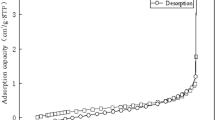
Abstract
In this study, humic substances—dry cowdung powder and humic acid were used for the removal of precious metal ion Ag(I) from aqueous solution. The effect of process parameters such as contact time, adsorbent dose, pH, and metal ion concentration on the adsorption process was estimated. These novel sorbents exhibited high percentage removal of Ag(I) as 95% and 77%, with a biosorption capacity of 19.0 mg g−1 and 3.88 mg g−1 for dry cowdung powder and humic acid respectively. High uptake percentages along with thermodynamic and kinetic calculations prove this process to be economical, practicable and the most eco-friendly amongst the available techniques.




Similar content being viewed by others
References
Semionov A (2018) Minamata disease: review. World J Neurosci 8:178–184
Mazumder D, Dasgupta UB (2011) Chronic arsenic toxicity: studies in West Bengal, India. Kaohsiung J Med Sci 27(9):360–370
Zeng H, Wu J (2013) Heavy metal pollution of lakes along the mid-lower reaches of the Yangtze River in China: intensity, sources and spatial patterns. Int J Environ Res Public Health 10(3):793–807
Jaishankar M, Tseten T, Anbalagan N, Mathew BB, Beeregowda KN (2014) Toxicity, mechanism and health effects of some heavy metals. Interdiscip Toxicol 7(2):60–72
Quiton KG, Doma B, Futalan CM, Wan MW (2018) Removal of chromium(VI) and zinc(II) from aqueous solution using kaolin-supported bacterial biofilms of Gram-negative E. coli and Gram-positive Staphylococcus epidermidis. Sustain Environ Res 28(5):206–213
Hefne JA, Mekhemer WK, Alandis NM, Aldayel OA, Alajyan T (2010) Removal of silver(I) from aqueous solutions by natural bentonite. JKAU Sci 22(1):155–176
Fomina M, Gadd GM (2014) Biosorption: current perspectives on concept, definition and application. Bioresour Technol 160:3–14
Hanzlik J, Jehlicka J, Sebek O, Weishauptova Z, Machovic V (2004) Multi-component adsorption of Ag(I), Cd(II) and Cu(II) by natural carbonaceous materials. Water Res 38(8):2178–2184
Jeon C (2015) Adsorption behavior of silver ions from industrial wastewater onto immobilized crab shell beads. J Ind Eng Chem 32:195–200
Herrera I, Tiemann KJ, Armendariz V (2003) Binding of silver(I) ions by alfalfa biomass (Medicago sativa): batch pH, time, temperature, and ionic strength studies. J Hazard Subst Res 4:1–16
Jeon C (2017) Adsorption of silver ions from industrial wastewater using waste coffee grounds. Korean J Chem Eng 34(2):384–391
Silva-medeiros FV, Consolin-filho N, Lima MX, Bazzo FP, Barros MA, Tavares CR (2016) Kinetics and thermodynamics studies of silver ions adsorption onto coconut shell activated carbon. Environ Technol 37(24):3087–3093
Sarı A, Tuzen M (2013) Adsorption of silver from aqueous solution onto raw vermiculite and manganese oxide-modified vermiculite. Microporous Mesoporous Mater 170:155–163
Cantuaria ML, Almeida Neto AF, Nascimento ES, Vieira MG (2016) Adsorption of silver from aqueous solution onto pre-treated bentonite clay: complete batch system evaluation. J Clean Prod 112:1112–1121
Barot NS, Bagla HK (2009) Extraction of humic acid from biological matrix: dry cow dung powder. Green Chem Lett Rev 2(4):217–221
Akinde SB, Obire O (2008) Aerobic heterotrophic bacteria and petroleum-utilizing bacteria from cow dung and poultry manure. World J Microbiol Biotechnol 24(9):1999–2002
Barot NS, Khilnani RP, Bagla HK (2014) Biosorptive profile of synthetic and natural humiresin for the remediation of metallic water pollutants. J Radioanal Nucl Chem 302(2):951–959
Boggs S, Livermore D, Seitz MG (1985) Humic substances in natural waters and their complexation with trace metals and radionuclides: a review. Geosci: Environ Sci. http://udspace.udel.edu/handle/19716/1567
Pettit RE (2004) Organic matter, humus, humate, humic acid, fulvic acid and humin: their importance in soil fertility and plant health. A & M University, Texas. http://www.humates.com/pdf/ORGANICMATTERPettit.pdf
Bhatt ND, Bagla HK (2017) Sustainable remediation of Hg(II) from wastewater by combo humiresin- dry cow dung powder. J Environ Biotechnol Res 6(1):168–178
Ambe F, Ambe S, Enomoto S (2003) Handbook of nuclear chemistry: chemical applications of nuclear reactions and radiations, vol 3. Kluwer Academic Publishers, Saitama
Lihareva N, Dimova L, Petrov O, Tzvetanova Y (2010) Ag+ sorption on natural and Na-exchanged clinoptilolite from Eastern Rhodopes. Microporous Mesoporous Mater Bulg 130(1–3):32–37
Lu X, Yin QF, Xin Z, Zhang ZQ (2010) Powerful adsorption of silver(I) onto thiol-functionalized polysilsesquioxane microspheres. Chem Eng Sci 65(24):6471–6477
Pearson RG (1963) Hard and soft acids and bases. J Am Chem Soc 85(22):3533–3539
Karthikeyan S, Balasubramanian R, Iyer CSP (2007) Evaluation of the marine algae Ulva fasciata and Sargassum sp for the biosorption of Cu(II) from aqueous solutions. Bioresour Technol 98:452–455
Horsfall M, Spiff AI (2005) Effects of temperature on the sorption of Pb2+ and Cd2+ from aqueous solution by Caladium bicolor (Wild Cocoyam) biomass. Electron J Biotechnol 8(2):162–169
King P, Namdeti R, Lahari BS, Yekula PK (2008) Biosorption of zinc onto Syzygium cumini L.: equilibrium and kinetic studies. Chem Eng J. 144(2):181–187
Tee TW, Khan MA (1988) Removal of lead, cadmium and zinc by waste tea leaves. Environ Technol Lett 9(1):1223–1232
Mishraa PC, Patel RK (2009) Removal of lead and zinc ions from water by low cost adsorbents. J Hazard Mater 168(1):31–325
Navarro RR, Wada S, Tatsumi K (2005) Heavy metal precipitation by polycation–polyanion complex of PEI and its phosphonomethylated derivative. J Hazard Mater 123(1–3):203–209
Morsy M, Ahmed A, Ten NM, Ibrahim H, Doaa H (2017) Heavy metals biosorption from aqueous solution by endophytic Drechslera hawaiiensis of Morus alba L. derived from Heavy Metals Habitats. Mycobiology 45(2):73–83
Nightingale ER (1959) Phenomenological theory of ion solvation: effective radii of hydrated ions. J Phys Chem 63(9):1381–1387
Bakouri HE, Usero J, Morillo J, Ouassini A (2009) Adsorptive features of acid-treated olive stones for drin pesticides: equilibrium, kinetic and thermodynamic modeling studies. Bioresour Technol 100(18):4147–4155
Lagergren S (1898) Zur theorie der sogenannten adsorption geloster stoffe. K. Sven. Vetenskapsakademiens, Handl. vol 24, pp 1–39
Ho YS, Mckay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465
Ho YS, Mckay G (2000) The kinetics of sorption of divalent metal ions onto Sphagnum moss peat. Water Res 34(3):735–742
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shaikh, S.A., Bagla, H.K. Comparative study of 110mAg(I) removal from aqueous media by humic substances. J Radioanal Nucl Chem 322, 225–230 (2019). https://doi.org/10.1007/s10967-019-06691-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-019-06691-7