Abstract
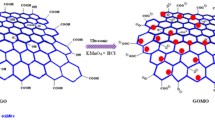
Functionalized magnetic graphene oxide nanoribbons (MGONRs) composite material was synthesized by hydrothermal treatment method using graphene oxide nanoribbons as raw material, which was formed by longitudinal unzipping of multi-walled carbon nanotubes in oxidizing environment. The morphology and structural properties of MGONRs were characterized by SEM, FT-IR, XRD and VSM and thorium adsorption behavior on MGONRs was investigated. The results showed that thorium adsorption on MGONRs was pH-dependent, endothermic and spontaneous. The adsorption process followed pseudo-second order and Freundlich isotherm model with rapid solid–liquid separation. MGONRs could have practical application in separation and recovery of thorium from aqueous solutions.










Similar content being viewed by others
References
García C, Rosales J, García L, Muñoz A, Rosales F, Brayner C, Pérez J (2013) Evaluation of uranium thorium and plutonium thorium fuel cycles in a very high temperature hybrid system. Prog Nucl Energy 66:61–72
Arogunjo AM, Höllriegl V, Giussani A, Leopold K, Gerstmann U, Veronese I, Oeh U (2009) Uranium and thorium in soils, mineral sands, water and food samples in a tin mining area in Nigeria with elevated activity. J Environ Radioact 99(3):232–240
Bacquart T, Bradshaw K, Frisbie S, Mitchell E, Springston G, Defelice J, Dustin H, Sarkar B (2012) A survey of arsenic, manganese, boron, thorium, and other toxic metals in the groundwater of a West Bengal, India neighbourhood. Metallomics 4(7):653–659
Wang L, Zhong B, Liang T, Xing B, Zhu Y (2016) Atmospheric thorium pollution and inhalation exposure in the largest rare earth mining and smelting area in China. Sci Total Environ 572:1–8
Wang M, Tao X, Song X (2011) Effect of pH, ionic strength and temperature on sorption characteristics of Th(IV) on oxidized multiwalled carbon nanotubes. J Radioanal Nucl Chem 288(3):859–865
Schierz A, Zänker H (2009) Aqueous suspensions of carbon nanotubes: surface oxidation, colloidal stability and uranium sorption. Environ Pollut 157(4):1088–1094
Romanchuk AY, Slesarev AS, Kalmykov SN, Kosynkin DV, Tour JM (2013) Graphene oxide for effective radionuclide removal. Phys Chem Chem Phys 15(7):2321–2327
Hirsch A (2009) Unzipping carbon nanotubes: a peeling method for the formation of graphene nanoribbons. Angew Chem Int Ed 48(36):6594–6596
Jiao L, Zhang L, Wang X, Diankov G, Dai H (2009) Narrow graphene nanoribbons from carbon nanotubes. Nature 458(7240):877–880
Camposdelgado J, Romoherrera JM, Jia X, Cullen DA, Muramatsu H, Kim YA, Hayashi T, Ren Z, Smith DJ, Yu O (2008) Bulk production of a new form of sp2 carbon: crystalline graphene nanoribbons. Nano Lett 8(9):2773–2778
Jiao L, Wang X, Diankov G, Wang H, Dai H (2010) Facile synthesis of high-quality graphene nanoribbons. Nat Nanotechnol 5(5):321–325
Li X, Wang X, Zhang L, Lee S, Dai H (2008) Chemically derived, ultrasmooth graphene nanoribbon semiconductors. Science 319(5867):1229–1232
Tapasztó L, Dobrik G, Lambin P, Biró LP (2008) Tailoring the atomic structure of graphene nanoribbons by scanning tunnelling microscope lithography. Nat Nanotechnol 3(7):397–401
Kosynkin DV, Higginbotham AL, Sinitskii A, Lomeda JR, Dimiev A, Price BK, Tour JM (2009) Longitudinal unzipping of carbon nanotubes to form graphene nanoribbons. Nature 458(7240):872–876
Higginbotham AL, Kosynkin DV, Sinitskii A, Sun Z, Tour JM (2010) Lower-defect graphene oxide nanoribbons from multiwalled carbon nanotubes. ACS Nano 4(4):2059–2069
Wang Y, Wang Z, Gu Z, Yang J, Liao J, Yang Y, Liu N, Tang J (2015) Uranium(VI) sorption on graphene oxide nanoribbons derived from unzipping of multiwalled carbon nanotubes. J Radioanal Nucl Chem 304(3):1–9
Wang Y, Wang Z, Ran R, Yang J, Liu N, Liao J, Yang Y, Tang J (2015) Synthesis of amidoximated graphene oxide nanoribbons from unzipping of multiwalled carbon nanotubes for selective separation of uranium(VI). RSC Adv 5(108):89309–89318
Gado MA (2018) Sorption of thorium using magnetic graphene oxide polypyrrole composite synthesized from natural source. Sep Sci Technol 53(13):2016–2033
Hritcu D, Humelnicu D, Dodi G, Popa MI (2012) Magnetic chitosan composite particles: evaluation of thorium and uranyl ion adsorption from aqueous solutions. Carbohydr Polym 87(2):1185–1191
Mirzabe GH, Keshtkar AR (2015) Application of response surface methodology for thorium adsorption on PVA/Fe3O4/SiO2/APTES nanohybrid adsorbent. J Ind Eng Chem 26:277–285
Wu L, Ye Y, Liu F, Tan C, Liu H, Wang S, Wang J, Yi W, Wu W (2013) Organo-bentonite-Fe3O4 poly(sodium acrylate) magnetic superabsorbent nanocomposite: synthesis, characterization, and Thorium(IV) adsorption. Appl Clay Sci 83–84:405–414
Liu P, Qi W, Du YF, Li Z, Wang J, Bi JJ, Wu WS (2014) Adsorption of thorium(IV) on magnetic multi-walled carbon nanotubes. Sci China Chem 57(11):1483–1490
Yang SK, Tan N, Yan XM, Chen F, Long W, Lin YC (2013) Thorium(IV) removal from aqueous medium by citric acid treated mangrove endophytic fungus Fusarium sp. #ZZF51. Mar Pollut Bull 74(1):213–219
Zhu HY, Fu YQ, Jiang R, Jiang JH, Xiao L, Zeng GM, Zhao SL, Wang Y (2011) Adsorption removal of congo red onto magnetic cellulose/Fe3O4/activated carbon composite: equilibrium, kinetic and thermodynamic studies. Chem Eng J 173(2):494–502
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (21601033, 21866006, 11875105, 21661003, 11705027), and Jiangxi Province Key Subjects Academy and Technique Leaders Funding Project (20172BCB22020), Natural Science Funds for Distinguished Young Scholar of Jiangxi Province (20171BCB23067), Open Project Foundation of Nuclear Technology Application Ministry of Education Engineering Research Center (East China University of Technology) (HJSJYB2016-6), Open Project Foundation of Stake key Laboratory of Nuclear Resources and Environment (East China University of Technology) (NRE1509).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, P., Wang, Y., Hu, X. et al. Synthesis of magnetic graphene oxide nanoribbons composite for the removal of Th(IV) from aqueous solutions. J Radioanal Nucl Chem 319, 1111–1118 (2019). https://doi.org/10.1007/s10967-018-6375-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-018-6375-2