Abstract
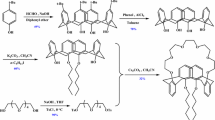
To separate Cs(I) from highly active liquid waste, a macroporous silica-based 25, 27-bis(iso-propyloxy)calix[4]arene-26,28-crown-6 (BiPCalix[4]C6) supramolecular recognition material, BiPCalix[4]C6/SiO2–P, was synthesized and characterized by SEM, FT-IR, and TG-DSC. The adsorption properties of BiPCalix[4]C6/SiO2–P and a macroporous polymer-based supramolecular recognition composite, BiPCalix[4]C6/XAD-7, were compared. It was found that BiPCalix[4]C6/SiO2–P exhibited better adsorption ability and faster adsorption dynamics than BiPCalix[4]C6/XAD-7. The adsorption isotherm of Cs(I) onto BiPCalix[4]C6/SiO2–P was studied at 298 K and it was well described by Langmuir isotherm model. The complex composition between BiPCalix[4]C6/SiO2–P and Cs(I) was determined as 1:1 type by investigating the effect of the concentrations of BiPCalix[4]C6, Cs(I), and H+ on the adsorption. Meanwhile, the selectivity of BiPCalix[4]C6/SiO2–P towards Na(I), K(I), Rb(I), Cs(I), Sr(II), Ba(II), Ru(III), Mo(VI), La(III), and Y(III) was investigated.













Similar content being viewed by others
References
Haverlock TJ, Bonnesen PV, Sachleben RA, Moyer BA (1997) Radiochim Acta 76:103
Gaur S (1996) J Chromatogr A 733:57
Haas PA (1993) Sep Sci Technol 28:2479
Yu B, Chen J, Song CL (2002) J Mater Sci Technol 18:206
Rao SVS, Lal KB, Narasimhan SV, Ahmed J (1999) J Radioanal Nucl Chem 240:269
Dwivedi C, Kumar A, Ajish JK, Singh KK, Kumar M, Wattal PK, Bajaj PN (2012) RSC Adv 2:5557
Dwivedi C, Kumar A, Juby KA, Kumar M, Wattal PK, Bajaj PN (2012) Chem Eng J 200:491
Whitney CD, Landsberger S (2009) J Radioanal Nucl Chem 280:281
Pedersen CJ (1967) J Am Chem Soc 89:7017
Schulz WW, Bray LA (1987) Sep Sci Technol 22:191
Mcdowell WJ (1988) Sep Sci Technol 23:1251
Romanovskiy VN, Smirnov IV, Babain VA, Todd TA, Herbst RS, Law JD, Brewer KN (2001) Solvent Extr Ion Exch 19:1
Law JD, Herbst RS, Todd TA, Romanovskiy VN, Babain VA, Esimantovskiy VM, Smirnov IV, Zaitsev BN (2001) Solvent Extr Ion Exch 19:23
Dozol JF, Simon N, Lamare V, Rouquette H, Eymard S, Tournois B, De Marc D, Macias RM (1999) Sep Sci Technol 34:877
Bonnesen PV, Delmau LH, Moyer BA, Leonard RA (2000) Solvent Extr Ion Exch 18:1079
Sachleben RA, Bryan JC, Engle NL, Haverlock TJ, Hay BP, Urvoas A, Moyer BA (2003) Eur J Org Chem 4862
Engle NL, Bonnesen PV, Tomkins BA, Haverlock TJ, Moyer BA (2004) Solvent Extr Ion Exch 22:611
Delmau LH, Haverlock TJ, Bazelaire E, Bonnesen PV, Ditto ME, Moyer BA (2009) Solvent Extr Ion Exch 27:172
Duncan NC, Roach BD, Williams NJ, Bonnesen PV, Rajbanshi A, Moyer BA (2012) Sep Sci Technol 47:2074
Arena G, Casnati A, Contino A, Mirone L, Sciotto D, Ungaro R (1996) Chem Commun 2277
Dietz ML, Ensor DD, Harmon B, Seekamp S (2006) Sep Sci Technol 41:2183
Zhang AY, Hu QH, Chai ZF (2009) Sep Sci Technol 44:2146
Zhang AY, Xiao CL, Chai ZF (2009) J Radioanal Nucl Chem 280:181
Zhang AY, Chen CM, Chat ZF, Kumagai M (2008) Adsorpt Sci Technol 26:705
Zhang AY, Chai ZF (2012) Ind Eng Chem Res 51:6196
Xiao CL, Zhang AY, Chai ZF (2012) Solvent Extr Ion Exch 30:17
Zhang AY, Wei YZ, Kumagai M (2004) React Funct Polym 61:191
Langmuir I (1918) J Am Chem Soc 40:1361
Freundlich H (1906) Z Phys Chem 57:385
Ungaro R, Casnati A, Ugozzoli F, Pochini A, Dozol JF, Hill C, Rouquette H (1994) Angew Chem Int Ed 33:1506
Casnati A, Pochini A, Ungaro R, Ugozzoli F, Arnaud F, Fanni S, Schwing MJ, Egberink RJM, Dejong F, Reinhoudt DN (1995) J Am Chem Soc 117:2767
Acknowledgments
The authors acknowledge financial support for the project sponsored by the National Natural Science Foundation of China under Contract Nos. 20671081 and 91126021, the Specialized Research Fund for the Doctoral Program of Higher Education under Contract No. 20070335183, and the Zhejiang Provincial Natural Science Foundation of China under Contract Nos. Y406022 and Y4110002.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xiao, C., Zhang, A. & Chai, Z. Synthesis and characterization of a novel organic–inorganic hybrid supramolecular recognition material and its selective adsorption for cesium. J Radioanal Nucl Chem 299, 699–708 (2014). https://doi.org/10.1007/s10967-013-2798-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-013-2798-y