Summary
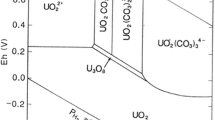
Understanding how uranium moves through the soil and groundwater is essential to determine the effectiveness of cleanup technologies. Uranium release and transport in the subsurface under oxic conditions have been reported to be mostly dependent on sorption onto Fe/Mn-oxide and complex interactions with organic substances. Available information in the literature, however, presents evidence of U retardation by natural sands. The aim of this investigation was to characterize U dissolution from a uraninite-containing rock (UO2-rock) in different waters under test conditions relevant to U transport from mine tailings.For this purpose, not shakenbatch experiments were conducted with a constant amount of anUO2-rockand different types of water (deionised, tap and mineral water). For comparison parallel experiments were conducted with 0.1M Na2CO3and 0.1M H2SO4. Furtherdissolution experiments using UO2-rock together with dolomite and pyritewere conducted. The results indicate that carbonate addition enhanced U solubilization, whereas pyrite addition essentially slowed the initial U solubilization. It is shown that SiO2and other rock constituents retard U transport.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Noubactep, C., Sonnefeld, J. & Sauter, M. Uranium release from a natural rock under near-natural oxidizing conditions. J Radioanal Nucl Chem 267, 591–602 (2006). https://doi.org/10.1007/s10967-006-0092-y
Issue Date:
DOI: https://doi.org/10.1007/s10967-006-0092-y